Case Presentation
Penile Metastasis as Unusual Presentation of Testicular Non-Seminomatous Germ Cell Tumor
Guida A1#, Pipitone S1*#, Noventa S2, Vitale MG3, Cascinu S1 and Sabbatini R3
1University of Modena and Reggio Emilia, Modena, Italy
2Operative Unit of Medical Oncology, Poliambulanza Foundation, Brescia, Italy
3Policlinico of Modena University Hospital of Modena, Modena, Italy
#Both authors are contributed equally
*Corresponding author: Annalisa Guida, University of Modena and Reggio Emilia, Modena, Italy
Published: 18 Jul, 2018
Cite this article as: Guida A, Pipitone S, Noventa S, Vitale
MG, Cascinu S, Sabbatini R. Penile
Metastasis as Unusual Presentation of
Testicular Non-Seminomatous Germ
Cell Tumor. Clin Oncol. 2018; 3: 1498.
Abstract
Penile metastasis from testicular cancer is a rare condition. To our knowledge, only 11 cases have been described in literature. Patients with non-pulmonary visceral metastases should be considered poor risk, based on the International Germ Cell Cancer Collaborative Group classification. It is debated whether penis lesion is a visceral site. With this clinical case we report our experience with a rare condition, focusing on its unusual clinical presentation, difficult prognostic assessment and identification of the best therapeutic approach.
Introduction
Penile Metastasis (PM) is an uncommon situation and only few data are available from case reports and reviews. In 1870 [1], described the first clinical case of a secondary cancer of the penis, as a consequence of colon-rectum adenocarcinoma [1]. In 2006 [2], published an exhaustive review describing clinical presentation and diagnosis of penile metastases. Among the 372 cases included in the review, only 11 cases of PM from testicular cancer were mentioned [2]. Secondary penile lesion from testicular Non-Seminomatous Germ Cell Tumors (NSGCT) is a rare condition with limited data about management. We describe an interesting case of a penile metastasis as first unusual presentation of a primitive testicular cancer.
Case Presentation
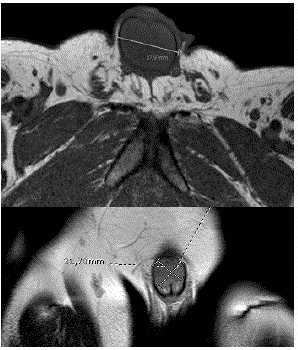
A Caucasian 45-year-old man presented to our clinic for a thickening at the base of his penis, appeared three months before. He had a negative familiar and personal history. Particularly no personal history of genitourinary pathology and of sexually transmitted disease was reported. He had no referred local surgery. Clinical examination revealed a fixed, hard, erythematous nodule, 2.5 cm in diameter, on the proximal third of the penis, without any other clinical signs or symptoms. Ultrasound scan of the penis showed diffusely thickened Corpora Cavernosas (CC). A Magnetic Resonance Imaging (MRI) revealed a single lesion measuring 35 mm x 38 mm of both CC (Figure 1: A, B). A detailed study with a total body computed Tomography (CT) revealed a single nodule in the right lung (17 mm). A fine-needle aspiration biopsy of the penile nodule indicated cohesive groups of pleomorphic epithelioid cells with prominent nucleoli, giving the concluding the diagnosis of metastasis due to germinal carcinoma. The patient underwent standard inguinal right orchiectomy. The tumor measured 1.5 cm, it was composed of embryonal carcinoma (60%), yolk sac tumor (30%) and teratoma (10%). Tunica albuginea infiltrates but does not invade, there is no vascular invasion. Most of the epididymis was replaced by moderately to poorly differentiated adeno carcinoma. Post-orchiectomy tumor markers were: AFP 267.5 ng/mL, βCGH 6529 mUI/ml, LDH 416 U/L. According to the American Joint Committee of Cancer/Union for International Cancer Control (AJCC/UICC), tumor staging was pT1a N0 M1b. Prognostic stratification for this patient, based on the International Germ Cell Cancer Collaborative Group (IGCCCG) the prognostic group was poor [3]. Patients with non-pulmonary visceral metastases should be considered poor risk. It is debated whether penis lesion is a visceral site. Anyway, four courses of BEP (cisplatin 20 mg/m2 on day 1–5, etoposide 100 mg/m2 on days 1–5, bleomicine 18 mg/m2 on days 2, 9 and 16) every three weeks represented the standard of care for both intermediate and poor risk patients. After the administration of only one cycle of BEP tumor markers were considerably reduced: AFP 10.9 ng/ mL, βCGH 105 mUI/ml, LDH 363 U/L. Only three cycles of BEP were administered, because of persistent penile infection. Prophylactics Granulocyte Colony-Stimulating Factors (G-CSF) were administrated after every cycle. At the end of systemic treatment, serum tumor markers were normalized. Penile MRI and ultrasound showed residual mass of the penis (19 mm x 23 mm x 17 mm) and CT scan described reduction of the lung lesion (5 mm). A multipledisciplinary discussion was held as to the option of surgical removal of the residual penis disease, as strangely recommended by National and International guidelines for residual mass >1 cm [4,5]. Our patient’s desire to preserve the penis was taken in account and they offered him a more strictly observation for the first one year: markers, CT scan and penile MRI every 3 months, than every 6 months. To date, the patient has been followed-up for 40 months and no progression of disease is evident.
Discussion
Penile metastasis from testicular cancer is uncommon. To our
knowledge, only 11 cases have been described in literature [2]. With
this clinical case we report our experience with a rare condition,
focusing on its unusual clinical presentation, difficult prognostic
assessment and identification of the best therapeutic approach.
Germ-cell tumor
Germ Cell Tumors (GCTs) are uncommon tumors and
represent for 1% of all male tumors [6,7]. GCTs are classified as
seminoma or non-seminoma. Non-seminomatous tumors often
comprehend multiple cell types, including embryonal cell carcinoma,
choriocarcinoma, yolk sac tumor, and teratoma. The 5-year survival
for testis cancer is 98% [6,7]. The most common site of metastasis in
testicular cancer is the lymph nodes in the abdomen; metastasis to the
lung, liver, bones, kidney and brain is also possible [8-10]. In 56% of
metastatic non-seminomatous GCTs disease there are normal tumors
markers are observed [11]. In their review [2], reported that in the
majority of cases Penis Metastases (PM) arise from pelvic primary
tumors. Prostate and bladder cancer are the most frequent primary
tumor presenting secondary penile lesions: respectively 34% and
30% of 372 penis metastases cases described in literature, followed by
recto-sigmoid neoplasm 13% and kidney 8%. As mentioned above,
only 11 cases (3%) arise from testicular cancer [2].
Clinical presentation and diagnosis
The most common site of secondary penile lesion is the CC (35%
of cases bilateral); most rarely are in Glans Penis (GP), Urethra Penile
(PU), Corpus Spongiosum (CS) [12-14]. Most commonly in duration
of the penis or a cutaneous lesion are the first presentation. Pain
or priapism is described, but less frequently. In 60% of metastatic
penile lesion there are multiple infiltrative nodules [15]. Differential
diagnosis of penile lesion includes mostly infectious disease, but also
pre-malignant conditions should be considered. Among malignant
lesions squamous cell carcinoma is the most common type of primary
penile cancer. Other epithelial forms of penis cancer, melanoma or
sarcoma are less frequent. Synchronous presentation of secondary
penile lesions was observed in around one third of patients, while
metachronous appearance of lesion occurs after a mean time of 18
months [16]. Ultrasonography and MRI are the gold standards
for accurate evaluation of involved anatomic structures of penis.
However, histological confirmation is necessary. In our clinical
case the penile localization was synchronous with testicular cancer,
while the other cases of penile metastasis were metachronous and
with a variable interval from the diagnosis of testicular tumor (from
2,5 to 24 month). Clinical presentation represents one of the main
points in our case, because about 4 months needed to discover the
primary tumor in the testis. Both synchronous and asymptomatic
presentations certainly contributed to delayed diagnosis.
Metastatic mechanism
It is difficult to understand the mechanism of metastatic
spreading to the penis. Indeed, despite the rich Vascularity of corpora
cavernosas, secondary lesions of the penis are rare. We briefly listed
several proposed mechanisms. Retrograde venous route: the venous
drainage of penis is via three main venous systems, composed by
superficial dorsal vein of penis, deep dorsal vein (retro pubic venous
plexus) and deep circumflex vein, retro chronal plexus and pudendal
plexus. Right testicular vein opens into inferior vena cava, where both
drainage venous systems are confluent. By a retrograde flow of blood,
testicular malignant cells may have spread into penis venous system
[14].
Retrograde lymphatic route: the lymphatic drainage of penis is
from the bladder base, the posterior surface of the prostate and the
lower rectum; drain into the external iliac nodes [14]. The lymphatic
drainage of testis is to lumbar and para-aortic nodes. By this way
malignant cells may arrive at the penile skin, then the corpora or
the glans penis [14]. Hematogenous spread: it is rare, but vascular
invasion is a known prognostic factor for NSGCT. Vascular invasion
was originally defined as the thrombotic occlusion in blood capillary
vessels [17]. The artery supply the penis originates from internal
pudendal arteries while testicular artery directly emerged from aorta
[14]. Direct invasion or instrumental seeding are less frequent and,
however, better recognized when occur [14]. Medical interview
is a crucial part in this setting. Surgical history for many common
conditions, such as inguinal hernia or cryptorchidism, may suggest
iatrogenic anatomic variant to understand spreading tumor way.
Management and treatment
According to IGCCCG prognostic classification, in good
prognosis group of patients the 5-years OS rate is 92% [3]. In
patients having intermediate prognosis features OS rate is 80% and
for the poor prognosis group patients’ OS rate is 48% [3]. In patients
with intermediate or poor prognosis, the standard treatment is
represented by chemotherapy, particularly four cycles of BEP [18].
In this subgroup of patients, complete response rate ranging from
55% to 82% [18]. VIP regimen (Cisplatin, Etoposide and Ifosfamide)
can be administrated in cases of contra-indications for the use to
bleomycin, such as high risk of lung injury [3,18]. The administration
of prophylactics G-CSF is indicated after every cycle of BEP [19]. In
2014 [19] published a phase III trial (GETUG 13) in patients with
poor-prognosis GCT. The patients with AFP and βHCG not declined
after one cycle of BEP, underwent to the administration of High-
Dose Chemo Therapy (HDCT) regimen. The data suggest that first
line HDCT may be effective in poor prognosis patients with slow
tumor markers decline after the administration of the first cycle of
BEP. HDCT consisting on the infusion of two cycles of paclitaxel (175
mg/m2 day 1) and BEP plus oxaliplatin (130 mg/m2 day 10), followed
by other two cycles of cisplatin (100 mg/m2 day 1), ifosfamide (2 g/
m2 on days 10, 12, 14 and mesna), and bleomycin (25 units/day, by
continuous infusion for 5 days on day 10 to 14), with G-CSF support.
The study met his primary endpoint: 5-year PFS rate was 60% in the
HDCT arm versus 47% in the BEP arm (HR 0.65; 95% CI 0.47–0.97;
p=0.037). Salvage chemotherapy rate was lower in HDCT treated
patients (17% vs. 8%; p=0.035) [20]. This approach is still not widely
accepted, cause of lacking similar data and the reproducibility of
tumor markers decline algorithm.
Surgical residual mass
After the administration of three or four cycles of cisplatinbased
chemotherapy, only 60% patients show radiological complete
response disease, more than 30% had partial response with the
presence of residual masses [21]. The prognosis of this patients
depended to serum tumour marker levelsdimensions of disease
before systemic treatment, and histological features of the resected
masses [22-24]. The presence of residual masses <1.5 cm, masses
reduction ≥ 90% after chemotherapy, and the absence of teratoma
component in the primitive testicular tumour are predictive
of necrosis or fibrosis [22-24]. Several trials showed histologic
discordance between Retro Peritoneal (RP) and Extra-Retro
Peritoneal (ERP) post-chemotherapy residual masses [22,25].
Histological features of residual mass were necrosis or fibrosis in 45%
to 50% of cases, teratoma in 30% to 40% of cases, variable malignant
cells (embryonal carcinoma, choriocarcinoma, yolk sac tumour,
undifferentiated carcinoma, undifferentiated teratoma, teratoma in
malignant transformation) in 10% to 20% of cases. Teratoma and
variable malignant cells tumor were related to poor outcomes [26-
28]. Removal of residual masses after chemotherapy in NSGCTs
remains the standard of care [29]. Resection of RP residual masses
has not only a curative objective but also a diagnostic rule [30,31].
The surgery can be done after four weeks at the end of chemotherapy
and in men with residual masses with normal serum tumour markers
levels [32]. When the primitive tumour shows components of variable
malignant cells or teratoma, the aim of surgery treatment is radical
excision of residual disease [32]. In fact, teratoma is chemo resistant;
it is very aggressive because it may infiltrate adjacent structures [25].
Devillè et al. retrospective analysed 51 men underwent surgery after
chemotherapy for metastatic NSGCT. Patients with elevated levels of
AFP at diagnosis, showed mature teratoma and/or viable malignant
cells in residual masses (p: 0.036). Worse outcome was reported in
poor prognosis group according to IGCCCG compared with good
and intermediate prognosis groups (p: 0.006) [29]. Steynberg et al.
suggested that the resection of RP residual disease was indicated
when tumoral mass reductions after chemotherapy was more than
70% and residual tumoral mass was more than 1 cm to CT-scan [33].
Carver et al. reported that poor prognostic risk according to IGCCCG
classification and dimension of residual masses were predictive of
disease recurrence in patients with residual teratoma at RP lymph
node dissection [34]. ERP metastasis is presents in 43% of patients
affected of GCT [35]. ERP residual disease is related to poor prognosis
if compared to retroperitoneal disease [25]. The most common
location of disease recurrence was in the lung, followed by the neck
and mediastinum [36-38]. In cases of surgical completely resection of
residual disease, mature teratoma or necrosis/fibrosis relapsed less of
10% patients; when in residual disease were found viable malignant
cells, the possibility of relapse was around 30% patients before the
first 2 years [29]. When residual disease was mature teratoma or
variable malignant cells, the administration of two cycles of cisplatinbased
chemotherapy (after the surgery) reduced the risks of relapse
and improved the PFS and OS [30,31,39].
Follow up
After treatments, men with metastatic NSGCT should be followed
for early detection and treatment of relapse. There are no a clear
management of this patients. According National and International
guidelines, it’s indicated a follow-up visit for at least the first 5 years
[4,5]. For non-semimoma stage I, clinical examination, serum markers
and chest-XR are recommended every 2-3 months for the first two
years and every 4-6 months from third and fifth year; abdomen-pelvic
CT scan are recommended every 6 months for the first two years and
once a year from third and fourth year as clinical indicated [4,5,40].
In metastatic setting, the follow-up schedule provides for a clinical
examination, serum markers, and every 2-3 months for the first two
year and every six months from third and fifth year. Chest-XR and CT
scan every six months for the first two years. CT scan should be done
once a year for the third year. Chest-XR should be done once a year
from third to fourth year [4,5,40].
Figure 1
Conclusion
To our knowledge, there are limited published data in the literature about non-seminomatous testicular cancer with metastatic localization to corpora cavernosas of penis. It’s interesting to observe that in our patient, penis metastases were the first evidence of neoplastic disease with any systemic symptoms. Despite the poor prognosis, after more than 3 years of the end of chemotherapy, he was asymptomatic without clinical evidence of neoplastic disease.
References
- CJE. Krehsmetastasen des corpus cavernosum penis. Virch Arch 51. 1870;145.
- Cherian J, Sreekumar Rajan, Ali Thwaini, Yaser Elmasry, Tariq Shah, Rajiv Puri. Secondary penile tumours revisited. Int Semin Surg Oncol. (2006);3:33.
- International Germ Cell Cancer Collaborative Group. Germ Cell Consensus Classification: a prognostic factor-based staging system for metastatic germ cell cancers. International Germ Cell Cancer Collaborative Group. J Clin Oncol. 1997;15(2):594-603.
- Linee guida AIOM Tumori del testicolo. 2013.
- NCCN clinical practice guidelines in oncology: testicular cancer. (2013). Siegel RL, Miller KD, J.A. Cancer statistics. CA Cancer J Clin. 2016.
- Bethesda. SEER Cancer Statistics Factsheets: Testis Cancer. Natl. Cancer Institute. 2016.
- Johnson DE, Appelt G, Samuels ML, Luna M. Metastases from testicular carcinoma Study of 78 autopsied cases. Urology. 1976;8(3):234-9.
- Donohue JP, Zachary JM, Maynard BR. Distribution of nodal metastases in nonseminomatous testis cancer. J Urol. 1982;128(2):315-20.
- Bredael JJ, Vugrin D, Whitmore WF. Autopsy findings in 154 patients with germ cell tumors of the testis. Cancer. 1982;50(3):548-51.
- Schmoll HJ, Souchon R, Krege S, Albers P, Beyer J, Kollmannsberger C, et al. European consensus on diagnosis and treatment of germ cell cancer: a report of the European Germ Cell Cancer Consensus Group (EGCCCG). Ann Oncol. 2004;15(9):1377-99.
- Abeshouse BS, Abeshouse GA. Metastatic tumors of the penis: a review of the literature and a report of two cases. J Urol. 1961;86:99-112.
- Mikuz G. Tumors of the Prostate Gland, Seminal Vesicles, Male Urethra, and Penis. Atlas of Tumor Pathology: Third Series, Fascicle 28. Young RH, Srigley JR, Amin MB, Ulbright TM Cubilla AL. Armed Forces Institute of Pathology, Washington, DC, 2000. J Pathol. 2001;195:638.
- Paquin AJ, Roland SI. Secondary carcinoma of the penis; A review of the literature and a report of nine new cases. Cancer. 1956;9(3):626-32.
- Osther PJ, Løntoft E. Metastasis to the penis. Case reports and review of the literature. Int Urol Nephrol. 1991;23(2):161-7.
- Pomara G, Pastina L, Simone M, Casale P, Marchetti G, Francesca F. Penile metastasis from primary transitional cell carcinoma of the renal pelvis: first manifestation of systemic spread. BMC Cancer. 2004;4:90.
- Howell TH. William Harvey’s report on Old Parr’s autopsy. Age Ageing. 1987;16(4):265-6.
- Feldman DR, Bosl GJ, Sheinfeld J, Motzer RJ. Medical treatment of advanced testicular cancer. JAMA 2008;299(6):672-84.
- Smith TJ, Khatcheressian J, Lyman GH, Ozer H, Armitage JO, Balducci L, et al. 2006 Update of Recommendations for the Use of White Blood Cell Growth Factors: an Evidence-Based Clinical Practice Guideline. J Clin Oncol. 2006;24(19):3187-205.
- Fizazi K, Pagliaro L, Laplanche A, Fléchon A, Mardiak J, Geoffrois L, et al. Personalised chemotherapy based on tumour marker decline in poor prognosis germ-cell tumours (GETUG 13): A phase 3, multicentre, randomised trial. Lancet Oncol. 2014;15(13):1442-50.
- Gerl A, Clemm C, Schmeller N, Dienemann H, Weiss M, Kriegmair M, et al. Sequential resection of residual abdominal and thoracic masses after chemotherapy for metastatic non-seminomatous germ cell tumours. Br J Cancer. 1994;70(5):960-5.
- Donohue JP, Rowland RG, Kopecky K, Steidle CP, Geier G, Ney KG, et al. Correlation of computerized tomographic changes and histological findings in 80 patients having radical retroperitoneal lymph node dissection after chemotherapy for testis cancer. J Urol. 1987;137(6):1176-9.
- Sheinfeld J, Bajorin D, Solomon M. Management of post chemotherapy residual masses in advanced germ cell tumors. AUA Updat Ser.1997;17:18-24.
- Bajorin DF, Herr H, Motzer RJ, Bosl GJ. Current perspectives on the role of adjunctive surgery in combined modality treatment for patients with germ cell tumors. Semin Oncol. 1992;19(2):148-58.
- Katz MH, McKiernan JM. Treatment of non-retroperitoneal residual germ cell tumor masses. Urol. Oncol. 2005;23(6):431-9.
- Fosså SD, Qvist H, Stenwig AE, Lien HH, Ous S, Giercksky KE. Is postchemotherapy retroperitoneal surgery necessary in patients with nonseminomatous testicular cancer and minimal residual tumor masses? J Clin Oncol. 1992;10(4):569-73.
- Stenning SP, Parkinson MC, Fisher C, Mead GM, Cook PA, Fossa SD, et al. Postchemotherapy residual masses in germ cell tumor patients: content, clinical features, and prognosis. Medical Research Council Testicular Tumour Working Party. Cancer. 1998;83(7):1409-19.
- Gerl A, Clemm C, Schmeller N, Dienemann H, Lamerz R, Kriegmai M, et al. Outcome analysis after post-chemotherapy surgery in patients with non-seminomatous germ cell tumours. Annals of oncology. 1995;6(5):483- 488.
- Deville JL, Gravis G, Salem N, Savoie PH, Esterni B, Walz J, et al. Resection of residual masses after chemotherapy for advanced nonseminomatous germ cell tumours, A monocentric analysis of pre-operative prognosticators. Eur J Cancer Care (Engl). 2010;19(6):827-32.
- Einhorn LH, Williams SD, Mandelbaum I, Donohue JP. Surgical resection in disseminated testicular cancer following chemotherapeutic cytoreduction. Cancer. 1981.
- Fox EP, Weathers TD, Williams SD, Loehrer PJ, Ulbright TM, Donohue JP, et al. Outcome analysis for patients with persistent nonteratomatous germ cell tumor in postchemotherapy retroperitoneal lymph node dissections. J Clin Oncol. 1993;11(7):1294-9.
- Melchior D, Müller SC, Albers P. Extensive surgery in metastatic testicular cancer. Aktuelle Urol. 2003;34(4):214-22.
- Steyerberg EW, Keizer HJ, Fosså SD, Sleijfer DT, Bajorin DF, Donohue JP, et al. Resection of residual retroperitoneal masses in testicular cancer: evaluation and improvement of selection criteria. The ReHiT study group. Re-analysis of histology in testicular cancer. Br J Cancer. 1996;74(9):1492-8.
- Carver BS, Shayegan B, Serio A, Motzer RJ, Bosl GJ, Sheinfeld J. Longterm clinical outcome after postchemotherapy retroperitoneal lymph node dissection in men with residual teratoma. J Clin Oncol. 2007;25(9):1033-7.
- Carver BS, Serio AM, Bajorin D, Motzer RJ, Stasi J, Bosl GJ, et al. Improved clinical outcome in recent years for men with metastatic nonseminomatous germ cell tumors. J Clin Oncol. 2007;25(35):5603-8.
- Stomper PC, Jochelson MS, Garnick MB, Richie JP. Residual abdominal masses after chemotherapy for nonseminomatous testicular cancer: Correlation of CT and histology. AJR Am J Roentgenol. 1985;145(4):743-6.
- Carter GE, Lieskovsky G, Skinner DG, Daniels JR. Reassessment of the role of adjunctive surgical therapy in the treatment of advanced germ cell tumors. J Urol. 1987;138(6):1397-401.
- Stephenson AJ, Sheinfeld J. The role of retroperitoneal lymph node dissection in the management of testicular cancer. Urologic Oncology: Seminars and Original Investigations. 2004;22(3):225-233.
- Fizazi K, Tjulandin S, Salvioni R, Germà-Lluch JR, Bouzy J, Ragan D, et al. Viable malignant cells after primary chemotherapy for disseminated nonseminomatous germ cell tumors: prognostic factors and role of postsurgery chemotherapy--results from an international study group. J Clin Oncol. 2001;19(10):2647-57.
- Van As NJ, Gilbert DC, Money-Kyrle J, Bloomfield D, Beesley S, Dearnaley DP, et al. Evidence-based pragmatic guidelines for the followup of testicular cancer: Optimising the detection of relapse. Br J Cancer. 2008;98(12):1894-902.