Short Communication
Detection of PDL-1 as of Biomarker in Checkpoint Blockade Therapy
Xiangyan Qiu, Haidong Tang and Mingyi Chen*
Department of Pathology, University of Texas Southwestern Medical Center, USA
*Corresponding author: Mingyi Chen MD, PhD, Associate Professor of Pathology, Division of Hematopathology, UT Southwestern Medical Center, BioCenter; 2330 Inwood Rd. EB3.234A, Dallas, Tx 75390
Published: 25 Nov, 2017
Cite this article as: Qiu X, Tang H, Chen M. Detection of
PDL-1 as of Biomarker in Checkpoint
Blockade Therapy. Clin Oncol. 2017;
2: 1368.
Short Communication
Immune checkpoints are a series of pathways which can regulate T cell activity as either coinhibitory or co-stimulatory signals [1]. Increasing evidences confirmed that Immune checkpoints play important roles in tumor evasion from antitumor immune responses (Figure 1) [2]. Clinical trials using antibodies targeting immune checkpoints, especially programmed cell death protein 1 (PD-1) and its ligandPD-1 ligand (PD-L1 or B7-H1), have shown revolutionized beneficial effects inmany patients with high clinical stages cancers which are usually resistant or refractory to conventional chemoradiation therapy. However, the objective responses are variable among each individual patient [3]. Thus, it has become a top priority to identify biomarkers that can predict patient responses to the immunotherapy.
PD-L1 Expression as Biomarker
It is widely accepted that tumor cells over express PD-L1 to inhibit T cell responses [4]. Higher
expression of PD-L1 on tumor cells has been shown a better prognosis after checkpoint blockade
intervention [5]. Thus, the expression of PD-L1 has been selected as companion diagnostics for
PD-1/PD-L1 blockade therapy (Table 1) [6]. It has been found that both tumor cells and tumor
associated stromal/immune cells express PD-L1. Due to the lack of conditional knockout mice,
the contributions of PD-L1 from different cell populations are still not well-defined. Most studies
analyze the PD-L1 protein level in tumor cellsto grade the expression. Some recent studies emphasize
the role of the PD-L1 expression in immune cells to predict the response rate of PD-L1 checkpoint
inhibitor treatment. (Table 1)(Figure 2) [7].
In spite of the fact that a higher potential response rate in patients with high level of positivity of
PD-L1 expression in tumor tissues, clinical responses were also observed in some patients that were
essentially negative for PD-L1 in tumor tissues [3]. The underlying mechanisms is still undetermined
for patients who respond to PD-L1 blockade therapy with negativePD-1 expression in tumor tissue.
Several potential hypothesis have been proposed. First, temporal and spatial heterogeneity of PDL1
expression may lead to false-negative result duet to the limitation of tumor sampling especially
in small core biopsy of clinical sample. Only a small fragment of the tumor tissues can be collected
at certain time points, which may not be representative of the entire tumor tissue [8]. Second, the
sensitivity of immunohistochemistry (IHC) is suboptimal to evaluate the PD-L1 expression. PD-L1
antigen may not be detected in the conventional IHC due to the tissue degeneration, fixing artifact
in formalin fixed paraffin embed tissue (FFPE) sample and different type of antigen retrieval process
in different labs. Furthermore, the lack of standardization as shown in Table 1, several different
antibody clones with different cut-off values have been utilized to quantify the PD-L1 expression
in different studies. One sample negative in one assay/study may turn out to be positive in others.
Third, the expression of PD-L1 can be somehow upregulated in adjacent to nonspecific chronic
inflammation responses, especially by interferon-gamma and TNF-alpha. It is possible that some
PD-L1 negative tumor tissue may turn out to be positive after prolonged therapies.
Biomarkers other Than PD-L1 Expression
Besides PD-L1 expression, several other biomarkers are also being developed as to predict patient responses to PD-1/PD-L1 blockade therapies. Defects in DNA repair mechanisms, such as microsatellite instability/mismatch repair deficiency (MSI/MRD), are associated with an increased somatic mutation loads. It has been shown that tumor mismatch repair status can also be used to predict the clinical responses to checkpoint blockade therapy [9]. Recently, FDA has approved Keytruda for the treatment of patients with microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) cancers. This is the first approval a cancer treatment based on a common biomarker rather than the location of the primary tumor. Other widely acceptedbiomarkeris the entire tumor mutation burden [10]. In fact, many tumors that respond well to checkpoint blockade therapy usually have higher mutation burdens [11]. The caveat still remains as 0a clear cut-off value for tumor mutation burden is not established. In addition, the requirement for high-throughput sequencing and relative high cost limit further development of the test.
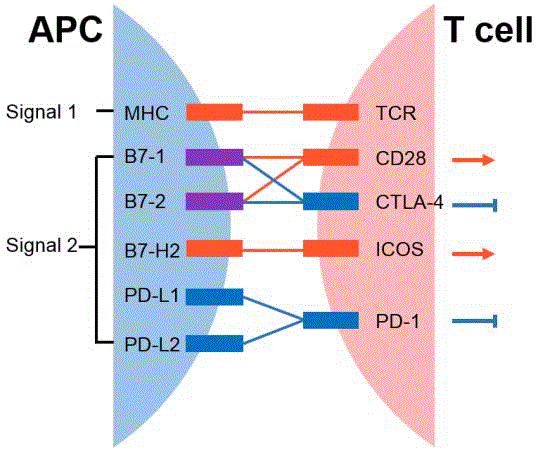
Figure 1
Figure 1
Immune checkpoints regulate T cell activation. Immune
checkpoints provide co-inhibitory or co-stimulatory signals, which lead to T
cell suppression or activation.
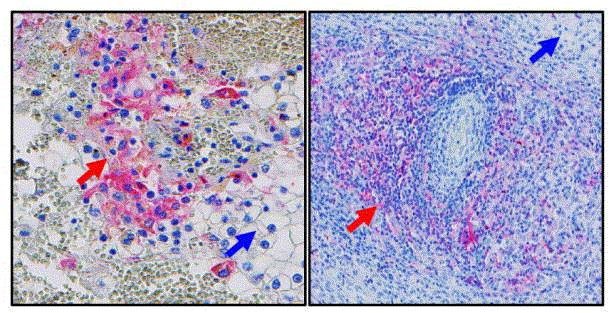
Figure 2
Figure 2
Typical immunohistochemical staining of PD-L1 in tissues from
renal cell cancer (RCC) patients. Blue arrow: tumor cells; red arrow: PD-L1
positive macrophages.
Table 1
Conclusion
With the explorative growing clinical trials on checkpoint blockade therapies in cancers, it has become an utmost need to identify reliable biomarkers to predict patient responses. In spite of the rapid progress of biomarker development of caner immunotherapy, the current assays are still not perfect due to the lack of selectivity, specificity, accuracy, and precision. Further investigations are needed in order to improve the current test or develop novel assays.
References
- Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nature reviews. Immunology, 2013; 13(4): p. 227.
- Shin DS, Ribas A. The evolution of checkpoint blockade as a cancer therapy: what's here, what's next? Curr Opin Immunol. 2015; 33: 23-35.
- Sunshine J, Taube JM. Taube, Pd-1/Pd-L1 Inhibitors. Curr Opin Pharmacol. 2015; 23: 32-8.
- Baumeister SH, Freeman GJ, Dranoff G, Sharpe AH6. Coinhibitory Pathways in Immunotherapy for Cancer. Annu Rev Immunol. 2016; 34: 539-73.
- Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Science translational medicine, Sci Transl Med. 2012; 4(127): 127ra37.
- Garber K. Predictive biomarkers for checkpoints, first tests approved. Nature biotechnology. 2015; 33(12): p. 1217-1219.
- Herbst RS, Soria JC, Kowanetz M, Fine GD, Hamid O, Gordon MS, et al., Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature, 2014; 515(7528): 563-567.
- Taube JM, Klein A, Brahmer JR, Xu H, Pan X, Kim JH, et al., Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti–PD-1 therapy. Clinical Cancer Research, 2014; 20(19): p. 5064-5074.
- Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015; 372(26): 2509-20.
- Yadav M, Jhunjhunwala S, Phung QT, Lupardus P, Tanguay J, Bumbaca S, et al. Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing. Nature. 2014; 515(7528): p. 572-576.
- Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SA, Behjati S, Biankin AV, et al., Signatures of mutational processes in human cancer. Nature, 2013; 500(7463): p. 415.