Review Article
Bacteriophages in Cancer Biology and Therapies
Swapnil Ganesh Sanmukh and Sérgio Luis Felisbino*
Laboratory of Extracellular Matrix Biology, Department of Morphology, Institute of Biosciences Botucatu, Sao Paulo State University, Sao Paulo, Brazil
*Corresponding author: Sérgio L Felisbino, Laboratory of Extracellular Matrix Biology, Department of Morphology, Institute of Biosciences Botucatu, Sao Paulo State University, Sao Paulo, Brazil
Published: 16 May, 2017
Cite this article as: Sanmukh SG, Felisbino SL.
Bacteriophages in Cancer Biology and
Therapies. Clin Oncol. 2017; 2: 1295.
Abstract
Cancer is one of the most challenging disease because of its unique individual identity and nonuniform
progression within the same host. This confronting situation demands individual
treatment therapies namely “personalized medicine”. Besides the improved success of available
cancer treatments in the majority of cancer cases, for many of them, they have proved ineffective as
the cancer cells evolve with treatment and show adaptive behavior. Bacteriophage (phage) therapy
and phage-based therapies have gained special attention as a promising approach for cancer therapy
beyond their action against multi-drug resistant bacterial infections. Considering the remarkable
development in the phage research, mainly in biomedical sciences, molecular biology and
nanobiotechnology; until now few conceptual reports about cancer and bacteriophage interactions
and/or detailed study for phage-based therapies in cancer are available. These kind of information
are valuable for reducing prejudices and to fundament discussions at regulatory agencies and
departments for its safe use in humans for cancer therapy. Here we have tried to summarize the
important works carried out about phage-cancer research and provide updated information about
its ongoing progress and possible future perspectives.
Keywords: Cancer; Bacteriophage; Nanobiotechnology; Regulatory agencies
Abbreviations
KGD Motifs: Tripeptide Lysine-Glycine-Asparagine Motifs; HAP1 Phage: A T4 Sub-Strain With A High Affinity To Melanoma Cells; IL: Interleukin; PMN: Polymorphonuclear Leukocytes; RGD: Tripeptide; Arginine-Glycine-Asparagine; HRP: Horseradish Peroxidase; PC3: Prostate Cancer Cell Line; Grp78: Glucose Regulated Protein; Hsp70: Heat Shock Protein 70; AAVP: Adeno-Associated Virus/Phage; IF: Interferon; USFDA: United States Food And Drug Administration; EMA: European Medicines Agency; IND: Investigational New Drug; CTA: Clinical Trial Application; Tsips: Tumor- Specific Internalizing Peptides; Thps: Tumor Homing Peptides
Introduction
Presently, various types of virus are used for broad range applications in biomedical
fields including many mammalian and bacterial viruses (bacteriophages) [1-3]. Among them,
bacteriophages have been explored for their range of applications since its discovery in 1915. Besides
being a “rosette stone” for molecular biology [4] they have shown various promising application
including phage therapy [5-8], phage display (expression of proteins or peptides on the phage
capsid) [9-13]| nanoparticles for practical applications in physical and material sciences [10], and
industrial applications [14]. The rise of antibiotic resistant bacteria has increased our interest to
focus on bacteriophages application as a potential treatment method for bacterial disinfection
through phage therapy and phage enzyme applications [15]. There is a need for intensive research
on bacteriophages and their role in cancer detection, treatment, diagnosis, etc. as reported recently
by researchers worldwide [16]. The phages have been identified as a genetically modifiable molecule
in various areas including medicine [17]..
Considering the interest exhibited by various researchers through direct or indirect approaches
for bacteriophage applications in medical sciences such as cancer research in last few decades,
it is very essential to explore it further to know in which way bacteriophages may prove useful.
The possible specificity of bacteriophage for certain cell and tissue components makes them a
crucial candidate against cancer. Irrespective to the wide use of bacteriophages for their potential
to kill many species of pathogenic bacteria through phage therapies; Nowadays, approaches for
using bacteriophages against cancer cells are emerging a lot and further understanding on how
mammalian cells responds in vivo to these phage agents with conjugates or through modification
requires serious attention [18].
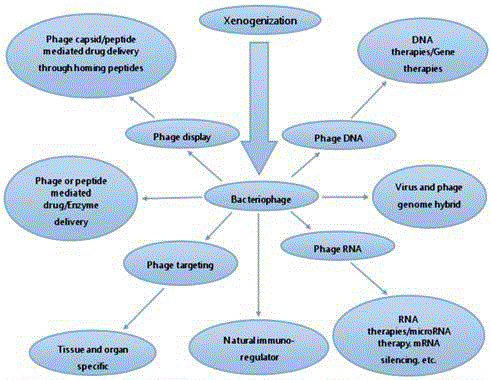
Figure 1
History of Bacteriophage in Cancer Research
Since, bacteriophages had proved to be one of the most important tools for various breakthroughs in molecular biology [19,20]. They are mostly studied for their derived applications worldwide along with their lytic life cycle which is well known and their lytic enzymes responsible for bacterial lyses events. The therapeutic potential of phages lies in their structure itself. Its minute and homogeneous size makes it the most capable nanoparticle for drug delivery as well as for various others purposes including phage display and targeting. Their capsid can be used directly to induce immune response naturally (due to their ubiquitous nature) or artificially (through genetic or protein engineering) in any organisms to express desired proteins on its surface to trigger immune response[21]. Due to the limitations of the current review article, we have restricted the broad applications of bacteriophages and just summarized the history of bacteriophage applications in cancer research and their possible outcome in Table 1.
Bacteriophage Interacts with Cancer Cell!
Although some studies have been conducted to know how the
bacteriophages can interact with cancer cells through in vivo and in
in vitro studies, their impact on influencing host immune response as
well as cancer cell physiology are less understood. In 1958, in-vivo and
in-vitro demonstration of bacteriophage binding to the cancer cells
was reported [22-24]. Similarly, the possible interactions between
phage capsid proteins (KGD motifs) as well as β-1 and β-3 integrin
receptors on target cancer cells was also confirmed along with the
binding mechanism of bacteriophage T4 to Melanoma cells [25,26].
The most puzzling aspect of cancer cell is some of their behaviors,
which are similar to a viral infected cell including apoptosis, cell cycle
deregulation, metastasis, etc. [27]. Moreover, the 'oncogene', the
genes which are attributed to most of the cancers have their origin
from viruses. It is by now well established concept that cancer is a
systemic biology disease and hence has a very deep root at the gene
level [28]. Taking into account the inert properties of phages due
to their non pathogenic nature for mammalian host, they can be
explored more extensively for further studies for their applications
in cancer research.
Current gold standard treatment for cancer includes surgery,
chemotherapy, radiation therapy and hormonal therapy. Other
advanced treatment methods have shown promising response
such as immunotherapy which includes tumor-targeting mAbs,
dendritic cell-based interventions, peptide-based vaccines, immunostimulatory
cytokines, immuno-modulatory monoclonal antibody,
pattern recognition receptor agonists, immunogenic cell death
inducers, etc. [29-33], are the emerging nanobiotechnologies recently
developed for the detection and treatment of different cancers. Even
though these methods are responsible for favorable response in cancer
patients, patients now have the option for a personalized medicine
approach [34], in which precise knowledge of the tumor molecular
signature is accessible making personalized medicine mandatory
[35]. In this scenario, bacteriophage, being a nanoparticle, can best fit
to be a unique component for various molecular therapies and can be
explored as a nanobiomedicine for cancer-treatment.
Why Bacteriophages for Cancer?
Bacteriophages are the most abundant entities in the biosphere that leads to its licensing for human use, mostly due to its non pathogenic nature and inert properties [36,37]. Being ubiquitous, we are frequently consuming large amount of phage populations through different sources [38]. Their detection in saliva, serum and stool samples confirms this finding and validates this observation [39-41]. Various results which have been published earlier showing wide applications of bacteriophages in cancer research and newly proposed applications seems to be very exciting (Figure 1). Some of the most preliminary and established bacteriophage applications are discussed below for getting an in depth idea about its current status and possible future applications.
Bacteriophage as a Natural and Involuntary Medicine
Bacteriophages seem to be an involuntary medicine as they are circulating in the mammalian body controlling invading pathogens and regulating our immune system naturally [42-44]. Furthermore, they can block β3 integrin activity on neoplastic cells thus preventing growth and metastasis formation [42]. Moreover, they also restrict angiogenesis in the developing cancerous tissues or organs and prevent metastasis by inhibiting the adhesion of platelets and T-cells to the fibrinogens [42,45,46]. As initially, it was noticed that phage phagocytes is mostly occurs in liver and spleen [47], hence it can be said that being antigenic in nature they naturally behave as an antigen presenting cells and constantly act as a source for antibody production. Moreover, some phages have been observed to induce the secretion of interleukins (eg. IL-12) and interferons (eg. IF-c) leading to the destruction of tumor tissues through cytokines; which initiate the cascade for neutrophil activation releasing reactive oxidative species (ROS) and other cytotoxic agents, ultimately leading to tissue damage [48]. In our ongoing studies, based on our preliminary results related to natural bacteriophage and Prostate Cancer cell (PC3) interactions it was observed that phages can stimulate internal cellular signaling and were responsible for gene expression alterations. The increased expressions for mRNAs corresponding to integrins (namely integrin αV, α5, β3 and β5) were detected significantly though quantitative PCR along with increase in AKT and PI3K signaling within these cells. Interestingly, we also got down-regulation for Hsp27 and Hsp90 genes after phage treatment, which are on contrary highly expressed in cancer cells and are responsible for migration, proliferation and prevention of apoptosis through survivin up regulation (unpublished). This broad phage binding characteristic as well as its role in specific gene expression mostly through their proteinaceous external coat represents a natural way of phage display.
Table 1
Bacteriophage for Gene Delivery, Gene Therapy, Cancer Detection, Targeting and Imaging
Recently, it has been showed that, bacteriophages can be
harnessed for their potential in gene delivery to eukaryotic cells
through transection of toxic genes to cancer cells. Such gene delivery
along with chemotherapeutic agents have great prospective in the
near future [49-51]. Similarly, phage display is useful in cancer
treatment because it is easy to modify the phage by gene insertion
to express desired anticancer proteins and/or peptides on its capsids
showing anticancer activities or induce immune response [52].
Similarly, various homing peptides have been reported against
cancer specific cells, tissues and organs. The M13/T7 phages were
used for tumor cell targeting through tumor homing peptides
identification (eg. RGD-4C Peptide) on the cancer cells and tissue
specific endothelial cells [53-58]. Moreover, organ specific targeting has been reported to be achieved by using organ homing peptides
and antigen detection approaches (eg. prostate-specific membrane
antigen) by using M13 as well as other phages [52,59-61]. It was also
demonstrated that anticancer drugs efficiency against breast cancer
was increased when homing peptides were used with integrins for
drug delivery without much toxic effect [62,63]. Identification of
tumour homing peptides and organ homing peptides can be helpful
for targeting cancer cells, antigen detection and drug delivery using
nanoparticles [61,62,64-66]. This approach facilitates the specific
targeting of cancer cells, which can be optimized for targeted
treatment or drug delivery through bacteriophages.
Nowadays, various databases and online tools are available
for predicted, experimentally validated and manually curated
comprehensive database for tumor homing and internalizing peptides
(Table 2). Many Tumor-Specific Internalizing Peptides (TSIPs) or
Tumor Homing Peptides (THPs) have been reported by using in
vitro and in vivo phage display technologies; which specifically target
tumor along with its microenvironments (eg., tumor vasculature)
through their systematic delivery. Such internalizing peptides are
of 3 to 15 amino acids length and are smaller than tumor homing
peptides. Screening of proteins or peptides for protein-protein
interactions can be carried out by displaying them on the surface of a
phage through phage coat proteins for identification of internalizing
or homing peptides [66].
Furthermore, promiscuous tumor targeting phage proteins have
been used for specifically targeting different cancer cells; It is reported
that intercellular accumulation of phage clones in cytoplasm,
mitochondria as well as nuclear region can be utilized as a potential
ligands for intracellular drug delivery and/or molecular imaging
techniques as they have few hours of pronuclear accumulation time
[67].
Gene therapy is a technique in which functional gene is delivered
or introduced into the target cells to restore, over express or inhibit
desired gene products [68], which is now used to treat different
cancers (e.g., peroxidases whose over expression can lead to the death
of cancer cells). Similarly, telomerase is extremely dynamic in most
of the cancer and fetal cells, but is almost undetectable in normal
tissues hence can be applicable as a good tool for targeting cancer cells
[69,70]. Furthermore, glucose regulated protein (Grp78) categorized
within the heat shock protein (Hsp70) family can be used as a biomarker
in stressed cells such as in tumors as a target for drug delivery
or gene therapy by phages [71-73]. The chimeric phage vector, named
Adeno-Associated Virus/Phage (AAVP) is a promising candidate
in targeted gene therapy [74]. It is a hybrid of eukaryotic adeno
associated virus and the filamentous M13 bacteriophage both having
single stranded DNA. It expresses 3-5 copies of the cyclic RGD-4C
ligand as in RGD-GC peptides on the phage pIII minor coat protein
targeting αvβ3-integrin receptor mostly associated with tumor cells
[75,76]. Considering this scenario and potential of bacteriophages,
they exhibit enormous prospectives in cancer detection, targeting
and imaging.
Table 2
Bacteriophage as an Immunomodulatory
The first study on interactions between bacteriophage and
immune response were conducted by Felix d’Herelle demonstrating
that phages have the potential to influence immune response [77].
Some phages like phi X174 are considered to be a standard antigen
and can trigger humoral responses when administered intravenously;
Since then, phage phi X174 have been extensively studied to
evaluate humoral immunity in immunosuppressed patients [78,79].
Moreover, due to the omnipresent nature of bacteriophages in the
biosphere, phage-neutralizing antibodies are reported to be present
in the serum even before phage administration and with systemic
administration can increase the phage neutralizing antibody titers
[80-83]. Interestingly, considering the antigenic nature of phages,
they can stimulate cellular response as whole phage particles and
can induce stronger sensitization than their conjugate counterparts
as reported in the case of phage sensitized lymphocytes in animals
[84]. T-cells do not inactivate phages in vivo, which seems to be a very
good indication for further exploring phages for their potential as an
immuno modulator [85].
Similarly, rodents show humoral response against phage coat
proteins when introduced through oral route [86]. It was also reported
that regardless of the route of phage administration, T4 phage particles
are cleared considerably from the mice by spleen [86,87]. However, in
vivo experimentation studies in mouse have led us to the observation
that, T4 phage can be cleared from mouse circulatory system due to
removal of Hoc protein (Highly immunogenic outer capsid protein)
or defect in their genes [88,89]. As the results seems quite selfcontradictory
due to lack of more detailed investigation, there is a
need for reevaluation and further in-depth studies for understanding
this mechanism. Nevertheless, the therapeutic potential of the phages
seems promising and demands urgent attention.
The most interesting thing to be explored is how phages modulate
immune system of the diseased host (in vivo) and also cancer cell
line responses (in vitro). The first report of phage demonstrating the
immune response in animals dated back to 1956, have shown their
potential as a new way of treatment methods in the near future [90].
The antibodies produced against phage T4 head proteins (i.e. Hoc and gp23) have been demonstrated to interfere in T4 phage activity [91].
Therefore, it is not a surprise to know that anti-phage antibodies are
always present in the healthy individuals [90,92-94]. The studies on
bacteriophages and their interaction in mammalian immune system
are not yet explored thoroughly. Very few researchers have shown
that phages can induce immune response due to their antigenic
nature and many more accomplishments are yet to be made [95].
Patents on Bacteriophage Mediated Cancer Treatment
Recent increase in therapeutic approach for cancer treatment
have facilitated the use of bacteriophages for cancer detection and
treatment as various patents have been granted in the last few decades
to phage based cancer technologies. The main aspect of developing
the phage based technologies is not just restricted to its nanostructure,
immunogenicity or ubiquitous nature but its flexibility, which can
be utilized for different applications and therapies. Filamentous
phages have been used as a modulator for angiogenesis (Patent:
20150110745) in which phages carrying bacterial lipopollysaccharide
endotoxin on its capside surface can be used for treating cancer by
modulating angiogenesis process, which can be upregulated or down
regulated depending upon the requirement for treating the cancer.
Similarly, bacteriophages and their prophage proteins can be used
in vitro to express poly-peptides having a proliferation inhibitory
activity in the mammalian cell lines as well as it can be used along
with pharmaceutical compositions and gene therapeutic approach
(Patents: US2009117084, US20050572260 and EP 1853291B1). A
patent (US8507445) explaining the methodology as well as targeting
peptide composition for diagnosis and therapy of human cancer was
also introduced in last decade. Furthermore, patent (WO2002020822
A2) for biopanning and rapid analysis of selective interactive ligands
(brasil) was also developed which helps for rapid and efficient
separation of specific target binding phages.
Recent patent (Patent: US9034312B2) on extracellular matrix
composition for the treatment of cancers looks very promising, as
there are various reports of different poly-peptides, mRNAs and
microRNAs interfering in cancer metabolic process at cellular and
genetic level [52]. Even though few cytotoxic suicide genes are known,
there is a requirement for an effective and efficient gene for target
oriented killing of cancer cells in a patient (Patent EP 1618886 A1). If
we are able to deliver a suicide gene within the specific cancer tissues
or organs, nanoparticles like bacteriophage can assist us to specifically
detect and target the cancer cells. Another example of combinatory
therapy is using miRNAs, which have been reported to modify tumor
phenotypes by regulating the genetic mechanism and also regulate cell
growth and apoptosis [95]. Considering this potential applicability
and possible approaches for targeting cancerous cells, bacteriophages
proves to be a good candidate for cancer research encompassing
various areas like cancer imaging, detection, targeting and treatment
by both in vivo and in vitro applications.
Regulatory Framework for Phage Therapy and Bacteriophage-based Therapy
The bacteriophage therapy is been used for the treatment of
various pathogenic bacteria as well as multi drug resistant bacterial
infections and they have also shown potential applications for
agricultural, animal and human use [96]. However, bacteriophagebased
technologies are emerging significantly against various
infectious diseases with the advancement in the biotechnological
applications and provide options through improved phage-based
therapies and diagnostics tools [97].
The main issue is in accepting phage based therapies or products
for human use, which are restricted due to regulatory frameworks
in different countries. In Poland, the clinical applications of
bacteriophages are accepted for treatment as per Polish Law Gazette
Number 28 of 1997 and Declaration of Helsinki [98]. Presently,
the commercial phage preparations are approved against bacterial
pathogens in food for human consumption as per USFDA and are
restricted to phage therapy [101,103]. As per standard industrial
production criteria, phage-therapy products have been recognized
for Investigational New Drug (IND) or Clinical Trial Application
(CTA) submission [99-102]. However, bacteriophage based therapies
are not yet fully permitted for human use by some important
regulatory bodies like United States Food and Drug Administration
(USFDA) and the European Medicines Agency (EMA) as very limited
randomized controlled clinical trials for bacteriophages have been
conducted till date. As an alternative, some US companies have tried
to develop phage based products for the decontamination purposes
in agricultural sectors [103]. If the possible recommendation of phage
based therapies are well accepted, it would be easier to have great
awareness for accepting phage based products not only limited for
use as an alternative for antibiotics, but also, for other phage derived
products for humans including those for cancer treatments.
Future Prospective
The bacteriophages are presently used as a vector for various applications and have been reported to be important constituents of mammalian body playing a vital role in influencing their host immune response. As we know that extracellular matrix composition plays an important role in suppressing cancerous tumor growth, bacteriophages can be used to intensify this effect through their direct or indirect applications [104-109]. The phages can be used to display ploy-peptides and modulate useful constituent of extracellular matrix for anti-cancerous activities by regulating different poly-peptides, mRNAs and microRNAs interfering in cancer mechanisms. Similarly, phage mediated naked eye counting by miRNA molecule in petri dish can be implemented for detection of cancer biomarkers [110-114], which can prove helpful in understanding the cancer cell cycle and pathways. Recently, Anti-bacterial antibody-M13-Dye-probe was used to detect bacterial infections in living host, similar applications can be implemented for detecting cancerous tissues or organs in humans [115]. If we are able to optimize the therapeutic potential of bacteriophages for their direct or indirect applications, we can possibly revolutionize a whole new field of cancer treatment therapy. Bacteriophages can be an efficient cancer research tool and provide a better option for cancer treatment. As observed from the previous research works and results, the applicability of phages in medical field is still not fully explored and hence, in future tremendous breathtaking achievements and technological innovations awaits.
Acknowledgment
This article comprises part of the Ph.D. thesis of SGS, supported by a CAPES funding (processes No. 963-14-2). We would also like to thank Dr. Krystyna Dabrowska from Institute of Immunology and Experimental Therapy at Polish Academy of Science, for providing important literatures for this review.
References
- Agrawal P, Bhalla S, Usmani SS, Singh S, Chaudhary K, Raghava GP, et al. CPPsite 2.0: a repository of experimentally validated cell penetrating peptides. Nucleic Acids Research. 2016; 44(D1): D1098-D1103.
- Arap W, Kolonin MG, Trepel M, Lahdenranta J, Cardo-Vila M, Giordano RJ, et al. Steps toward mapping the human vasculature by phage display. 2002; 8(2): 121-127.
- Arap W, Pasqualini R, Ruoslaht E. Cancer treatment by targeted drug delivery to tumor vasculature in a mouse model. Science. 1998; 279(5349): 377-380.
- Arter JA, Diaz JE, Donavan KC, Yuan T, Penner RM, Weiss GA. Virus- polymer hybrid nanowires tailored to detect prostate-specific membrane antigen. Anal Chem. 2012; 84(6): 2776-2783.
- Au GG1, Lincz LF, Enno A, Shafren DR. Oncolytic Coxsackievirus A21 as a novel therapy for multiple myeloma. Br J Haematol. 2007; 137(2): 133-141.
- Bachrach G, Leizerovici-Zigmond M, Zlotkin A, Naor R, Steinberg D. Bacteriophage isolation from human saliva. Lett Appl Microbiol. 2003; 36(1): 50-53.
- Bais S, Bartee E, Rahman MM, McFadden G, Cogle CR. Oncolytic Virotherapy for Hematological Malignancies. Adv Virol. 2012; 2(1): 1-5.
- Bardhan N M, Ghosh D, Belcher A M. M13 Virus based detection of Bacterial Infections in Living Hosts. J Biophotonics. 2014; 7(8): 617-623.
- Bearden CM, Agarwal A, Book BK, Vieira CA, Sidner RA, Ochs HD, et al. Rituximab inhibits the in vivo primary and secondary antibody responses to a neoantigen, bacteriophage phi X174. Am J Tranplant. 2005; 5(1): 50-57.
- Bloch H. Experimental investigation on the relationships between bacteriophages and malignant tumors. Arch Virol. 1940; 1(2): 481-496.
- BoratyĹ ski J, Syper D, Weber-Dabrowska B, Ĺusiak-Szelachowska M, PoĹşniak G, GĂłrski A. Preparation of endotoxin-free bacteriophages. Cell Mol Biol Lett. 2004; 9(2): 253-259.
- Broccoli D, Young JW, De Lange T. Telomerase activity in normal and malignant hematopoietic cells. Proc Natl Acad Sci USA. 1195; 92(20): 9082-9086.
- Burrows FJ and Thorpe PE. Vascular targeting a new approach to the therapy of solid tumors. Pharmac. Ther. 1994; 64: 155-174.
- Cao B, Yang M, and Mao C. Phage as a Genetically Modifiable Supramacromolecule in Chemistry, Materials and Medicine. Accounts of Chemical Research. 2016.
- Chen PY, Dang X, Klug MT, Qi J, Dorval Courchesne NM, Burpo FJ, et al. Versatile three-dimensional virus-based template for dye-sensitized solar cells with improved electron transport and light harvesting. 2013; 27(8): 6563-6574.
- Chen WR, Adams RL, Carubelli R, Nordquist RE. Laser-photosensitizer assisted immunotherapy: a novel modality for cancer treatment. Cancer Lett. 1997; 115(1): 25-30.
- Citorik RJ, Mimee M, Lu TK. Bacteriophage-based synthetic biology for the study of infectious diseases. Curr Opin Microbiol. 2014; 19: 59-69.
- Clark JR, March JB. Bacterial viruses as human vaccines? Expert Rev Vaccines. 2004; 3(4): 463-476.
- Cooper C J, Khan Mirzaei M, Nilsson, A S. Adapting drug approval pathways for bacteriophage-based therapeutics. Front. Microbiol. 2016; 7(7): 1209.
- D’Herelle F. Opsonic power of the lysins. In ‘‘Bacteriophage: It’s Role in Immunity’’, p. 125. Williams & Wilkins, Baltimore. 1922.
- Dabrowska K, Opolski A, Wietrzyk J, Switala-Jelen K, Boratynski J, Nasulewicz A, et al. Antitumour activity of bacteriophages in murine experimental cancer models caused possibly by inhibition of ß 3 integrin signaling pathway. Acta Virol. 2004; 48(4): 241-248.
- Dabrowska K, Opolski A, Wietrzyk J, Switala-Jelen K, Godlewska J, Boratynski J, Syper et al. Anticancer activity of bacteriophage T4 and its mutant HAP1 in mouse experimental tumour models. Anticancer Res. 2004; 24(6): 3991-3995.
- Dabrowska K, Switała-Jelen K, Opolski A, Weber-Dabrowska B, Gorski A. Bacteriophage penetration in vertebrates. J Appl Microbiol. 2005; 98(1): 7-13.
- Dabrowska Krystyna, Miernikiewicz Paulina, Piotrowicz Agnieszka, Hodyra Katarzyna, Owczarek Barbara, Lecion Dorota, et al. Immunogenicity studies of protein forming the T4 phage head surface. J Virol. 2014; 88(21): 12551-12557.
- Denekamp J. Angiogenesis, neovascular proliferation and vascular pathophysiology as targets for cancer therapy. Br J Radiol. 1193; 66(783): 181-196.
- DePorter SM. McNaughton BR. Engineered M13 Bacteriophage Nanocarriers for Intracellular Delivery of Exogenous Proteins to Human Prostate Cancer Cells. Bioconjugate Chem. 2014; 25(9): 1620-1625.
- Di Giovine M, Salone B, Martina Y, Amati V, Zambruno G, Cundari E. Saggio Binding properties, cell delivery, and gene transfer of adenoviral penton base displaying bacteriophage. Virology. 2001; 282(1): 102-112.
- Diamandis M, White NMA, Yousef GM. Personalized Medicine: Marking a New Epoch in Cancer Patient Management. 2010; 8(9): 1175-1187.
- Doll TAPF, Raman S, Dey R, Burkhard P. Nanoscale assemblies and their biomedical applications. J R Soc Interface. 2013; 10(80): 657.
- Drulis-Kawa Z, Majkowska-Skrobek G, Maciejewska B, Delattre AS, Lavigne, R. Learning from Bacteriophages - Advantages and Limitations of Phage and Phage-Encoded Protein Applications. Current Protein & Peptide Science. 2012; 13(8): 699-722.
- Duelli D1, Lazebnik Y. Cell-to-cell fusion as a link between viruses and cancer. Nat Rev Cancer. 2007; 7(12): 968-796.
- Edelstein ML, Abedi MR; Wixon J. Gene therapy clinical trials worldwide to 2007- An update. J Gene Med. 2007; 9: 833-842.
- Eriksson F, Tsagozis P, Lundberg K, Parsa R, Mangsbo SM, Persson MA, et al. Tumor-specific bacteriophages induce tumor destruction through activation of tumor-associated macrophages. J Immunol. 2009; 182(5): 3105-3111.
- Fossati G, Bucknall RC, Edwards SW. Insoluble and soluble immune complexes activate neutrophils by distinct activation mechanisms: changes in functional responses induced by priming with cytokines. Ann Rheum Dis.2002; 61(11): 13-19.
- Galluzzi L, Vacchelli E, Bravo-San Pedro JM, Buqué A, Senovilla L, Baracco EE, et al. Classification of current anticancer immunotherapies. Oncotarget. 2014; 5(24): 12472-12508.
- Gantzer C, Henny J, Schwartzbrod L. Bacterioides fragilis and Escherichia coli bacteriophages in human faeces. Int J Hyg Environ Health. 2002; 205(4): 324-328.
- Garzon R, Marcucci G, Croce CM. Targeting microRNAs in cancer: rationale, strategies and challenges. Nat Rev Drug Discov. 2010; 9(10): 775-789.
- Gautam A, Singh H, Tyagi A, Chaudhary K, Kumar R, Kapoor P, Raghava GP. CPPsite: a curated database of cell penetrating peptides. Database (Oxford). 2012.
- Geier MR, Trigg ME, Merril CR. Fate of bacteriophage lambda in non-immune germ-free mice. Nature. 1973; 246(5430): 221-223.
- Gorski A, Dabrowska K, Switala-Jelen´ K, Nowaczyk M, Weber- Dabrowska B, Boratynski J, et al. New insights into the possible role of bacteriophages in host defense and disease. Med Immunol. 2003; 14: 2.
- Gorski A, Kniotek M, Perkowska-Ptasinska A, Mroz A, Przerwa A, Gorczyca W. Bacteriophages and transplantation tolerance. Transplant Proc. 2006; 38(1): 331-333.
- Górski A, Wazna E, Dabrowska BW, Dabrowska K, Switała-Jeleń K, Miedzybrodzki R. Bacteriophage translocation. FEMS Immunol Med Microbiol. 2006; 46(3): 313-319.
- Gorski A, Weber-Dabrowska B. The potential role of endogenous bacteriophages in controlling invading pathogens. Cell Mol Life Sci. 2005; 62: 511-519.
- Goubran HA, Elemary M, Radosevich M, Seghatchian J, El-Ekiaby M, Burnouf T. Impact of Transfusion on Cancer Growth and Outcome. Cancer Growth and Metastasis. 2016.
- Gross AL, Gillespie JW, Petrenko VA. Promiscuous tumor targeting phage proteins. Protein Eng Des Sel. 2016; 29(3): 93-103
- Hajitou A, Rangel R, Trepel M, Soghomonyan S, Gelovani JG, Alauddin MM, et al. Design and construction of targeted aavp vectors for mammalian cell transduction. Nat Protoc. 2007; 2(3): 523-531.
- Hajitou A, Trepel M, Lilley CE, Soghomonyan S, Alauddin MM. Marini FC3rd, Rest el al. A hybrid vector for ligand-directed tumor targeting and molecular imaging. Cell. 2006; 125: 385-398.
- Hedstrom SA, Kamme C. Antibodies against staphylococcal bacteriophages in human sera. II. Assay of antibodies in exacerbation and regression of chronic staphylococcal osteomyelitis. Acta Path Microbiol Scand. 1973; 81(6): 749-752.
- Henry M, Debarbieux L. Tools from viruses: Bacteriophage successes and beyond. Virology. 2012; 434(2): 151-161.
- Hornberg JJ, Bruggeman FJ, Westerhoff HV, Lankelma J. Cancer: a Systems Biology disease. Biosystems. 2006; 83(2-3): 81-90.
- http://www.bioprocessintl.com/manufacturing/nonantibody-therapeutics/bacteriophages-an-alternative-to-antibiotics-challenges-and-possible-solutions-for-bringing-them-to-market.
- Ibrahim SA, Hassan H, Götte M. MicroRNA-dependent targeting of the extracellular matrix as a mechanism of regulating cell behavior. Biochim Biophys Acta. 2014; 1840(2): 2609-2620.
- Inchley CJ. The activity of mouse kupffer cells following intravenous injection of T4 bacteriophage. Clin Exp Immunol. 1969; 5(2):173-187.
- Jerne NK, Avegno P. The development of the phage-inactivating properties of serum during the course of specific immunization of an animal: reversible and irreversible inactivation. J Immunol. 1956; 76(3): 200-208.
- Jerne NK. The presence in normal serum of specific antibody against bacteriophage T4 and its increase during the earliest stages of immunization. J Immunol. 1956; 76(3): 209-216.
- Jin SE, Jin HE, Hong SS. Targeted delivery system of nanobiomaterials in anticancer therapy: from cells to clinics. Biomed Res Int. 2014; 814208(6): 1-23.
- Kamme C. Antibodies against staphylococcal bacteriophages in human sera. I. Assay of antibodies in healthy individuals and in patients with staphylococcal infections. Acta Pathol Microbiol Scand (B) Microbiol Immunol. 1973; 81(6): 741-748.
- Kantoch M, Mordarski M. Binding of bacterial viruses by cancer cells in vitro. Postepy Hig Med Dosw. 1958; 12(2): 191-192.
- Kapoor P, Singh H, Gautam A, Chaudhary K, Kumar R, Raghava GP. TumorHoPe: a database of tumor homing peptides. PLoS One. 2012; 7(4): e35187.
- Keen EC. A century of phage research: bacteriophages and the shaping of modern biology. Bioessays. 2015; 37(1): 6-9.
- Kim M, Madlambayan GJ, Rahman MM. Myxoma virus targets primary human leukemic stem and progenitor cells while sparing normal hematopoietic stem and progenitor cells. Leukemia. 2009; 23(3): 2313-2317.
- Kleinschmidt WJ, Douthart RJ, Murphy EB. Interferon production by T4 coliphage. Nature. 1970; 228(5266): 27-30.
- Kniotek M, Ahmed AMA, Dabrowska K, Switala-Jelen K, Opolski A, Gorski A. Bacteriophage interactions with T cells and platelets. In cytokine network, Regulatory, cells signalling, and apoptosis (immunology). Monduzzi Editors, Bologna. 2004; 89-192.
- Kucharzewicz-Krukowska A, Slopek S. Immunogenic effects of bacteriophage in patients subjected to phage therapy. Arch Immun Ther Exp. 1987; 35(7): 553-561.
- Kurzepa A, Dabrowska K, Skaradziński G, Górski A. Bacteriophage interactions with phagocytes and their potential significance in experimental therapy. Clin Exp Med. 2009; 9(2): 93-100.
- Kurzepa-Skaradzinska A, Lusiak-Szelachowska M, Skaradzinski G, Jonczyk-Matysiak E, Weber-Dabrowska B, Zaczek M, et al. Influence of bacteriophage preparations on intracellular killing of bacteria by human phagocytes in vitro. Viral Immunol. 2013; 26(2): 150-162.
- Larocca D, Burg MA, Jensen-Pergakes K, Ravey EP, Gonzalez AM, Baird A. Evolving phage vectors for cell targeted gene delivery. Curr Pharm Biotechnol. 2002; 3(1): 45-57.
- López MB, Teijeiro A, Rivas J. Magnetic nanoparticle-based hyperthermia for cancer treatment. Reports of Practical Oncology & Radiotherapy 2013; 18(6): 397-400.
- Mactier CE, Islam MS2. Haematopoietic stem cell transplantation as first-line treatment in myeloma: a global perspective of current concepts and future possibilities. Oncol Rev. 2012; 6(2): e14.
- Matsumoto M, Saeki S, Kajiwara K, Ishihara H, Amano T, Nishizaki T, et al. ERvelopment of brain tumor demarcation technique using two-color laser-induced fluorescence. 2014; 33(4): 260-278.
- Merabishvili M, Pirnay JP, Verbeken G, Chanishvili N, Tediashvili M, Lashkhi N, et al. Quality-controlled small-scale production of a well-defined bacteriophage cocktail for use in human clinical trials. PLoS One. 2009; 4(3): e4944.
- Merril CR, Friedman TB, Attallah AF, Geier MR, Krell K, Yarkin R. Isolation of bacteriophages from commercial sera. In Vitro. 1972; 8(2): 91-93.
- Mohan K, Weiss G A. Engineering chemically modified viruses for prostate cancer cell recognition. Mol. Biosyst. 2015; 11: 3264-3272.
- Mohan K, Weiss GA1. Chemically Modifying Viruses for Diverse Applications. ACS Chem Biol. 2016; 11(5): 1167-1179.
- Nilsson AS. Phage therapy--constraints and possibilities. Ups J Med Sci. 2014; 119(2): 192-198.
- Oliveira H, Sillankorva S, Merabishvili M, Kluskens LD, Azeredo J. Unexploited opportunities for phage therapy. Front Pharmacol. 2015; 6(2): 180.
- Pajtasz-Piasecka E, Rossowska J, Dum D, Weber-Dabrowska B, Zablocka A, Gorski A. Bacteriophages support anti-tumor response initiated by DC-based vaccine against murine transplantable colon carcinoma. Immunol Lett. 2008; 116: 24-32.
- Pasqualini R, Ruoslahti E. Tissue targeting with phage peptide libraries. Mol Psychiatry. 1996; 1: 423-423.
- Pasqualini R. Vascular targeting with phage peptide libraries. Q J Nucl Med. 1999; 43(2): 159-162.
- Pelfrene E, Willebrand E2, Cavaleiro Sanches A3, Sebris Z4, Cavaleri M2. Bacteriophage therapy: a regulatory perspective. J Antimicrob Chemother. 2016; 71(8): 2071-2074.
- Pirnay JP, Blasdel BG, Bretaudeau L, Buckling A, Chanishvili N, Clark JR, et al. Quality and Safety Requirements for Sustainable Phage Therapy. Products. Pharm. Res. 2015(7): 2173-2179.
- Pranjol Md, Zahidul I and Hajitou A. Bacteriophage-Derived Vectors for Targeted Cancer Gene Therapy. Viruses. 2015; (1): 268-284.
- Przerwa A, Kniotek M, Nowaczyk M, Weber-Dabrowska B, Switala-Jelen K, Dabrowska K,et al. Bacteriophages inhibit interleukin-2 production by human T lymphocytes. In ‘‘12th Congress of the European Society for Organ Transplantation, Geneva Switzerland. 2005.
- Przerwa A, Zimecki M, Switala-Jelen´ K, Dabrowska K, Krawczyk E, Luczak M. Effects of bacteriophages on free radical production and phagocytic functions. Med Microbiol Immunol. 2006; 195(6): 143-150.
- Rama AR, Prados J, Melguizo C, Alvarez PJ, Ortiz R, Madeddu R, et al. The phage gene transfection associated to chemotherapeutic agents increases apoptosis in lung and colon cancer cells. Bioeng Bugs. 2011; 2(2): 163-167.
- Rivinoja A, Laakkonen P. Identification of homing peptides using the in vivo phage display technology. Methods Mol Biol. 2011; 683(3): 401-415.
- Sathaliyawala T, Islam MZ, Li Q, Fokine A, Rossmann MG, Rao VB. Functional analysis of the highly antigenic outer capsid protein, Hoc, a virus decoration protein from T4-like bacteriophages. Mol Microbiol. 2010; 77(2): 444-455.
- Schilsky RL. Personalized medicine in oncology: the future is now. Nature Reviews Drug Discovery. 2010; 9(5): 363-366.
- Sharma A, Kapoor P, Gautam A, Chaudhary K, Kumar R, Chauhan JS, et al. Computational approach for designing tumor homing peptides. Sci Rep. 2013; 3: 1607.
- Shu D, Li H, Shu Y, Xiong G, Carson W E, Haque F, et al. Systemic Delivery of Utilizing RNA Nanotechnology Anti-miRNA for Suppression of Triple Negative Breast Cancer. ACS Nano; Ahead of printing. 2015.
- Sinkovics JG, Horvath JC. Natural and genetically engineered viral agents for oncolysis and gene therapy of human cancers. Arch Immunol Ther Exp (Warsz). 2008; 56(1): 3s-59s.
- Srivastava AS, Kaido T, Carrier E. Immunological factors that affect the in vivo fate of T7 phage in the mouse. J Virol Methods. 2004; 115(1): 99-104.
- Stone R. Stalin's forgotten cure. Science. 2002; 298(5594): 728-731.
- Sugahara KN, Teesalu T, Karmali PP, Kotamraju VR, Agemy L, Girard OM, et al. Tissue-penetrating delivery of compounds and nanoparticles into tumors. Cancer Cell. 2009; 16(6): 510-520.
- Sulakvelidze A, Alavidze Z, Morris JG Jr. Bacteriophage therapy. Antimicrob Agents Chemother. 2001; 45(3): 649-659.
- Srivastava AS, Kaido T, Carrier E. Badanie mechanizmu dziaiania przeciwnowotworowego preparatu fagowego BP T4 u myszy obarczonych przeszczepialnym czerniakiem B16 (In Polish). Dissertation, Polish Academy of Science, Institute of Immunology and Experimental Therapy, Wroclaw, Poland. 2004.
- Thiel K. Old dogma, new tricks--21st Century phage therapy. Nat Biotechnol. 2004; 22(1): 31-36.
- Thirukkumaran CM, Luider JM, Stewart DA, et al. Reovirus oncolysis as a novel purging strategy for autologous stem cell transplantation. Blood. 2003; 102(1): 377-387.
- Torring T, Helmig S, Ogilby PR, Gothelf KV. Singlet Oxygen in DNA Nanotechnology. Acc Chem Res. 2014; 47(2): 1799-1806.
- Twort F. An investigation on the nature of ultra-microscopic viruses. Lancet. 1915; 186(4): 4814.
- Verbeken G, De Vos D, Vaneechoutte M, Merabishvili M, Zizi M, Pirnay J P. European regulatory conundrum of phage therapy. Future Microbiology. 2007; 2(5): 485-491.
- Verbeken G, Pirnay J P, De Vos D, Jennes S, Zizi M, Lavigne R, et al. Call for a dedicated European legal framework for bacteriophage therapy. Arch. Immunol. Ther. Exp. 2014; 62(2): 117-129.
- Verbeken G, Pirnay JP, De Vos D, Jennes S, Zizi M, Lavigne R, et al. Optimizing the European Regulatory Framework for Sustainable Bacteriophage Therapy in Human Medicine. Arch Immunol Ther Exp. 2012; 60: 161.
- Wang G, Li X, Wang Z. APD2: The updated antimicrobial peptide database and its application in peptide design. Nucleic Acids Research. 2009.
- Wang, G Li, X Wang Z. APD3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Research. 2016; 44.
- Wang Z, Wang G. APD: the Antimicrobial Peptide Database. Nucleic Acids Res. 2004; 32: D590-592.
- Warner C, Barker N, Lee SW, Perkins E. M13 bacteriophage production for large-scale applications. Bioprocess Biosyst Eng. 2014; 37(10): 2067-2072.
- Weber-Dabrowska B, Mulczyk M, Górski A. Bacteriophage therapy of bacterial infections: an update of our institute's experience. Arch Immunol Ther Exp. 2000; 48(6): 547-551.
- Wedgwood R J, Ochs H D, Davis SD. The recognition and classification of immunodeficiency diseases with bacteriophage phi X174. Birth Defects Orig Artic Ser. 1975; 11(2): 331-338.
- Wenger SL, Turner JH, Petricciani JC. The cytogenetic, proliferative and viability effects of four bacteriophages on human lymphocytes. In Vitro. 1979; 14(6): 543-549.
- Yang XL, Fan CH, Zhu HS. Photo-induced cytotoxicity of malonic acid [C60] fullerene derivatives and its mechanism. Toxicology in Vitro. 2002; 16(1): 41-46.
- Zagar TM, Oleson JR, Vujaskovic Z, Dewhirst MW, Craciunescu OI, Blackwell KL, et al. Hyperthermia for locally advanced breast cancer. Int J Hyperthermia. 2010; 26(7): 618-624.
- Zhang LH, Zhang X. Roles of GRP78 in physiology and cancer. J Cell Biochem. 2010; 110(6): 1299-1305.
- Zheng Y, Yin G, Le V, Zhang A, Chen S, Liang X, Liu J. Photodynamic-therapy Activates Immune Response by disrupting Immunity Homeostasis of Tumor Cells, which Generates Vaccine for Cancer Therapy. Int J Biol Sci. 2016; 12(1): 120-132.
- Zhou X, Cao P, Zhu Y, Lu W, Gu N and Mao C. Phage-mediated counting by the naked eye of miRNA molecules at attomolar concentrations in a Petri dish. Nature Materials. 2015; 14(10): 1058-1064.