Review Article
Is Whole Body –Diffusion MRI an Alternative to PET-CT! A Review of Literature
Atul Kapur1*, Neeraj Jain2, Aprajita Kapur1 and Goldaa Mahajan1
1Department of Advanced Diagnostics & Institute of Imaging, Kennedy Avenue, India
*Corresponding author: Atul Kapur, Department of Advanced Diagnostics & Institute of Imaging, Kennedy Avenue, India
Published: 09 May 2017
Cite this article as: Kapur A, Jain N, Kapur A, Mahajan
G. Is Whole Body –Diffusion MRI an
Alternative to PET-CT! A Review of
Literature. Clin Oncol. 2017; 2: 1285.
Abstract
WB-DWMRI and PET-CT are the two functional tools available for detection and staging of cancer patients. PET-CT has been the established modality since last two decades however with the improvements in MRI technology it is now possible to perform a whole body functional and anatomical imaging. This article reviews the current literature to determine whether WB–DWMRI can be an alternative imaging to PET-CT for imaging cancer patients. The study reviewed 19 studies from 2009-2016 for all types of regional and metastatic cancers including lymphomas and myelo infiltrative disorders to determine the sensitivity and specificity of both the imaging modalities. The results of these studies are discussed and these showed a comparable sensitivities and specificities. The study concludes that due to inherent advantages of WB-DWMRI i.e. lack of radiation, time saving, no use of contrast and lower cost it can be considered as an alternative imaging tool in the evaluation of such patients.
Introduction
Diffusion Weighted Magnetic Resonance Imaging (DW-MRI) is a useful and attractive
technique for evaluation of cancer patients [1]. Recent technological advances have made it a
promising technique for staging disease in oncology practice [2]. Till date PET-CT is the established
modality for evaluation of oncology patients however it is associated with considerable cost and also
adds radiation burden to the patient and also to the medical personal.
The article reviews the current position of body these modalities in staging oncology patients to
find the answer to the question of whether WB-DWI MRI is an alternative to PET-CT!
Technique
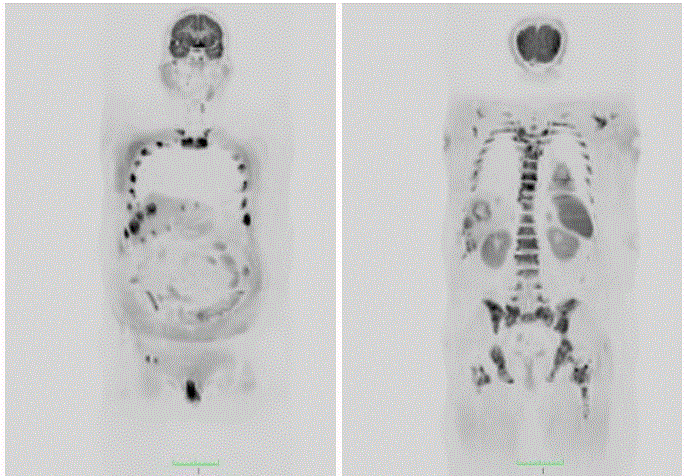
WB-DW MRI is done on both 1.5T and 3.0T MRI systems with the use of 4-5 station coils without the use of any contrast with images taken from head to mid tibia level and are acquired axially in free breathing state. Diffusion sequences are applied in X,Y and Z axis before and after 180 degree inversion pulse to have a fat saturation image along with diffusion sensitivity b values of 0,400,800mm2/sec. Data obtained is reformatted in multi planar manner as whole body images in coronal planes and inverted as black and white grey scale images (Figure1a and b) for analysis. ADC values for any region of interest are determined for estimation of tumor cellularity and expressed in units of 10mm2/s -3mm2/s. Another parameter used is lesion to spinal cord ratio determined on high b value image by using ROI technique.
Review of Literature
We searched the Pub med for studies done for correlation and comparison of PET-CT and WB-DWMRI and reviewed the results. 19 studies from current literature were found out which compared the results of both the modalities in patients of cancer for staging and suspected metastasis and also other myeloproliferative disorders (Table 1). All these studies were found to be having a common conclusion that WB- DWMRI can be used as a diagnostic test to diagnose and stage the disease and also to assess treatment response in such patients for which quantities DWMRI can be used as biomarker for early assessment of response to treatment.
Lung Cancer Detection and Staging
Mori “et al”. [3] performed prospectively 18F-FDG PET-CT and DWMRI on 104 patients with
pulmonary nodules of which 106 were proven malignant. SUV values and SUV CR values were
obtained to determine the nature of lesion on PET-CT and ADC min values are determined on DW MRI to determine the same. After using an optimal cut off PET-CT showed a sensitivity of 70% while it was 72% with ADC min Similarly
Specificities were 79% and 97% respectively. Authors concluded that
DWI can be used to distinguish between malignant and benign lung
nodules as the results of DWI were better than PET-CT Ohba “et al”.
[4] also studied 110 patients of NSCLC and showed similar results
of both DWI and PET-CT in Differentiating malignant from benign
nodules with sensitivity and specificity of 73%, 96% and 72% and
82%. However authors also suggested that DWI may not be able to
predict the aggressiveness of NSCLC.
However, Kanuchi “et al”. [5] in their study showed that DWI
had a sensitivity of 90% in predicting tumor aggressiveness in patients
of NSCLC with specificity of 81%. They also included the parameter
of lesion to spinal cord ratio along with ADC min to categories the
nature of lesion. Takenaka “et al”. [5]. Compared the results of staging
of lung cancer patients with both the modalities in 115 patients of
NSCLC. Results determined were on both per site and per-patient
basis and found that the results of WB-DWMRI and PET-CT were
similar. However when DWMRI was combined with routine MRI the
specificity improved on per site basis.
Table 1
Colorectal Cancer
Gong “et al”. [6] studied and compared WB-DWMRI and PETCT
in 29 patients of Gastro intestinal malignancies which included
predominantly patients of colorectal cancer. They showed a sensitivity
and specificity of WB –DWMRI 81.1% and 99.1% while of PETCT
95.1% and 99.8% with a similar overall accuracy of 98.9% and
95.9%. The sensitivity of PET-CT was higher due to detection of small
lung metastasis less than 6mm while two patients of liver metastasis
and brain metastasis were missed bt PET-CT. The study concluded
that WB-DWMRI is reliable in detecting gastro intestinal tumor
recurrence and metastasis and offers same diagnostic performance as
PET-CT and without radiation. The quantitative value of ADC also
provides extra information to determine metastasis.
Ono “et al”. [7] compared primary and metastatic lymph node
detection in patients with colorectal cancer.
A total of 25 patients of proven colorectal cancer were studied
by both PET-CT and WB-DWMRI. Out of the surgically proven
patients 23(85.2%) were true positive on both PET-CT and WBDWMRI.
PET-CT however detected only 3/10 patients with nodal
metastasis (30% sensitivity) while DWMRI detected 8/10 patients
(80% sensitivity). Ohio suggested that DWMRI was better to PET-CT
in nodal detection while inferior to it in primary colorectal cancer
detection.
Breast Cancer
Schmidt “et al”. [8]. Studied 33 patients with recurrent breast cancer and compared the diagnostic accuracy of WB-MRI and FDGPET- CT for the detection of tumor recurrence. WB-MRI and PETCT were both useful for the detection of tumor recurrence. WB-MRI was highly sensitive to detect distant metastatic disease. PET-CT was more sensitive in detecting lymph node involvement. Overall sensitivity was 91% for PET-CT and 90% for WB-MRI. Overall specificity was 90% for FDG-PET-CT and 86% for WB-MR. Heusner “et al”. [9]. Concluded in their study of 20 patients of breast cancer that WB-DWMRI had a sensitivity of 91% compared to 94% of PETCT for staging of breast cancer but had a low specificity of 72% while PET-CT had a specificity of 99% hence may not be suitable for staging the disease.
Figure 1a and b
Figure 1a and b
WB-DWMRI MIP images of the whole body with inverted MIP format showing multiple areas of focal restricted ADC in liver and in the skeletal
system consistent with metastasis in patient with bronchogenic carcinoma.
Multiple Myeloma
Whole body screening MRI is recommended for evaluation of any patient with plasmacytoma, asymptomatic myeloma for staging and lesion detection. Pawlyn “et al”. [10] evaluated 17 patients using WB-DWMRI and PET-CT for detection and quantification of lesions and showed that DWMRI detected far more statistically significant active diffuse disease than PET-CT. This is a important finding in the setting of new international myeloma working group consensus would lead to greater number of patients meeting criteria for active disease requirement. WB-DWMRI also outperformed PET-CT in assessment of iliac bone infiltration for good trephine results in such patients. In their study diffuse disease was seen in 37% (51/138) regions on WB-DWMRI compared to only 7% (10/140) on FDG PET-CT. Most of the regions reported with diffuse disease on DWMRI were reported as no disease on PET-CT. Overall 3/17 patients were reported as disease free on PET-CT had active diffuse disease on DWMRI.
Prostate Carcinoma
Prostate cancer is the most common cancer in males. Detection of disease is a challenge as is its staging. Most of the time there is early spread to regional nodes and bones. Shen “et al”. [11], did a meta analysis from 27 studies comparing PET-CT and DWMRI along with Bone Scan (BS) in detection and staging of prostate cancer. Their meta analysis showed on a per-patient basis, the pooled sensitivities by using choline PET-CT, MRI, and BS were 0.91, 0.97, 0.79 respectively. The pooled specificities for detection of bone metastases using choline PET-CT, MRI, and BS, were 0.99, 0.95 and 0.82 respectively. This meta-analysis indicated that MRI was better than choline PETCT and BS on a per-patient basis. Barchetti “et al”. [12] studied 152 males with recurrent prostate cancer after radical prostatectomy and showed a sensitivity and specificity of 99% and 98% with choline FDG PET-CT in evaluation of recurrent disease in such patients. Eschmann “et al”. [13] also compared both the modalities in 42 patients with prostate cancer and found high accuracy with similar results with the WB-DWMRI and PET-CT.
Lymphoma and Myeloproliferative Disorders
Staging of lymphoma is a important perspective before starting treatment of such patients. In routine clinical practice contrast enhanced CT is the commonly used modality because of its availability. WB-DWMRI becomes a attractive alternative due to lack of radiation and non use of contrast. Lin “et al”. [14] evaluated 17 patients of Diffuse large B cell lymphoma and compared results with PET-CT and showed 81% and 100% sensitivity of DWMRI in staging of disease. Stecco “et al”. [15] evaluated 17 patients with primary abdominal lymphomas and showed comparable results of DWMRI and PET-CT with sensitivity and specificity of 100%, 96% and 96% and 100% respectively. WB-DW-MRI was true-positive for 75 (100%) of the lymphomatous node groups and true-negative for 79 (96%) of the no metastatic node groups, while 18FFDG-PET-CT was true-positive for 71 (94%) of the lymphomatous node groups and true-negative for 83 (100%) of the non lymphomatous node groups. The study suggested like those of Abdulqadhr “et al”. [22] a role for WB-DW-MRI, combined with 18FFDG-PET-CT, in initial staging and provided that there is agreement between the two techniques as a standalone follow-up imaging technique. This would not only be safer for patients, but also improve cost-effectiveness and total examination time, both important limitations of 18F-FDG PET-CT, in the long term.
Any Metastasis
Gutzeit “et al”. [16] evaluated 37 patients with suspected bone
metastasis and compared WB-DWMRI with bone scintigraphy and
found a higher sensitivity of former of 97% with specificity of 99%
while bone scintigraphy had sensitivity of 91% and 87% respectively.
Fischer “et al”. [17] also showed in 68 patients higher detection rate of
WB-DWMRI combined with routine T2WI whole body MRI images
in detection of bony metastasis. The high performance of whole-body
MRI can in large part be ascribed to the emerging technique of WB
DW-MRI. Li “et al”. [18]. Showed WB DW-MRI in comparison to
PET/ CT had similar sensitivity (89.7% versus 89.5%) and specificity (95.4% versus 97.5%) for disease detection. A number of morphologic
criteria for bone disease progression and response were described.
Progression criteria include new focal/diffuse area(s) of metastatic
infiltration within normal marrow, increase in number/size of focal
lesions, evolution of focal lesions to a diffuse neoplastic pattern,
appearance of or increases in soft tissue associated with bone disease.
Stecco “et al”. [19] prospectively compared WB-DWMRI with
FDG PET-CT for staging 29 patients with malignancies to stage
and detect metastasis and showed an 89.1% sensitivity and 98.5%
specificity on region based detection of metastatic lesions using PETCT
as a reference standard and suggested a high predictive value of
DWI as a diagnostic tool.
Uterine Cervical Cancer
Choi “et al”. [20]. Retrospectively evaluated 236 patients of uterine cervical cancer with both WB-DWMRI and FDG PET-CT and the latter findings were used as standard of reference. Using ADC min value of 0.64 in nodes DWMRI showed a sensitivity of 86% and specificity of 80% in nodal metastasis detection also showed in 33 patients an inverse correlation of ADC min DWMRI and SUV max on PET-CT in Evaluation of uterine cervical cancer patients.
Head and Neck Cancer
Hassan “et al”. [21] did a metaanalysis of 6 studies in a search of 20 years of patients with Head and Neck Small Cell Carcinomas (HNSCC) and evaluated 329 patients with clinically N0 stage of nodal disease. Their results showed 100% sensitivity and 71% specificity of DWMRI in nodal detection while PET-CT showed a sensitivity and specificity of 68% and 84%. The evidence given in the study suggested that DWI was more sensitive than PET-CT in preoperative evaluation of C N0 disease in patients with HNSCC. PET-CT had a low sensitivity and a positive test would not help the clinician in the management of a patient with clinically N0 disease and therefore should not be routinely used in neck nodal status workups. The low sensitivity of PET-CT resulting in false negative results may be due to tumor necrosis FDG is not a cancer specific agent and its uptake is based on the metabolic rate of the cancer cell.
Conclusion
Review of the current literature in the present study highlights
the role of the WB-DWMRI as a functional and anatomical tool to
diagnose and stage different types of cancer patients. All most all the
studies have suggested comparable sensitivity and specificity of both
the modalities in detection of metastasis with PET-CT have a slight
edge in diagnosing small lung metastasis and sub cm nodal metastasis.
On the other hand DWMRI had a slight edge in detecting Liver, brain
and bony metastasis. The meta analysis done by Shen “et al”. [11]
showed that DWMRI had highest pool sensitivity for detection of
bony metastasis in prostate 0.97 followed by choline PET-CT 0.91
and SPECT bone scan was 0.71. For bone marrow assessment DW
MRI is considered to be the best as is sensitive to bone marrow
cell density, the relative proportions of fat and marrow and tumor
infiltrating cells, water content and bone marrow perfusion. The
information obtained can be quantified easily by ADC values and
can be used as robust parameter in assessing treatment response of
such patients. The strength of DWI MRI is also clearly established
in imaging of myeloma patients where whole body MR imaging is
being recommended by Myeloma working group guidance [25]. For
evaluation of lymphoma patients the evaluated studies in this paper
shows comparable accurate results of both modalities. Since normal
nodes in the body also have high cellularity and show reduced ADC
it is important to Determine ADC min values with cut off threshold
value to detect metastasis in nodes which are sub cm in size as was
shown in the studies discussed. However it would be more prudent
to have a combined DWI MRI and PET-CT base line study in the
first work up of such patients and then to follow up such patients
with WB DW MRI as a standalone technique. This would not only cut
down radiation dose in these patients but also be more convenient,
time saving and more importantly be cost effective [22]. Further
in monitoring of disease the use WB-DWMRI can also quantify
the tumor cellularity by use of ADC values and also obviates the
phenomenon of pseudo flare phenomenon seen of the plain MRI
T1W images.
To conclude review of the literature clearly shows that WBDWMRI
has a. important role in detection and staging of different
type of cancer patients and can be a viable alternative to PET-CT.
However one also needs to be aware of inherent pitfalls in the
technique DW-MRI like the false negatives in the presence of tumor
necrosis, cystic metastasis and imaging areas near to the heart.
References
- Koh DM, Collins DJ. Diffusion-weighted MRI in the body: applications and challenges in oncology. AJR Am J Roentgenol. 2007; 188(6): 1622-1635.
- Engelhard K, Hollenbach HP, Wohlfart K, von Imhoff E, Fellner FA. Comparison of whole-body MRI with automatic moving table technique and bone scintigraphy for screening for bone metastases in patients with breast cancer. Eur Radiol. 2004; 14(1): 99-105.
- Mori T, Nomori H, Ikeda K, Kawanaka K, Shiraishi S, Katahira K, et al. Diffusion-weighted magnetic resonance imaging for diagnosing malignant pulmonary nodules/masses: comparison with positron emission tomography. J Thorac Oncol. 2008; 3: 358-364.
- Ohba Y, Nomori H, Mori T, Ikeda K, Shibata H, Kobayashi H, et al. Is diffusion-weighted magnetic resonance imaging superior to positron emission tomography with fludeoxyglucose F 18 in imaging non-small cell lung cancer? J Thorac Cardiovasc Surg. 2009;138: 439-445.
- Takenaka D, Ohno Y, Matsumoto K, Aoyama N, Onishi Y, Koyama H, Nogami, et al. Detection of bone metastases in nonsmall cell lung cancer patients: comparison of whole-body diffusion-weighted imaging (DWI), whole-body MR imaging without and with DWI, wholebody FDG-PET-CT, and bone scintigraphy. J Magn Reson Imaging. 2009; 30: 298-308.
- Gong J, Cao W, Zhang Z, Deng Y, Kang L, Zhu P, et al. Diagnostic efficacy of whole-body diffusion-weighted imaging in the detection of tumor recurrence and metastasis by comparison with 18F-2-fluoro-2-deoxy-D-glucose positron emission tomography or computed tomography in patients with gastrointestinal cancer. Gastroenterol Rep. 2015; 128-135.
- Ono K, Ochiai R, Yoshida T, Kitagawa M, Omagari J, Kobayashi H, et al. Comparison of diffusion-weighted MRI and 2-[fluorine-18]-fluoro-2-deoxy-D-glucose positron emission tomography (FDGPET) for detecting primary colorectal cancer and regional lymph node metastases. J Magn Reson Imaging. 2009; 29: 336-340.
- Schmidt GP, Schmid R, Hahn K, Reiser MF. [Whole-body MRI and PET/CT in tumor diagnosis]. Radiologe. 2004; 44(11): 1079-1087.
- Heusner TA, Kuemmel S, Koeninger A, Hamami ME, Hahn S, Quinsten A, et al. Diagnostic value of diffusion-weighted magnetic resonance imaging (DWI) compared to FDG PET-CT for whole-body breast cancer staging. Eur J Nucl Med Mol Imaging. 2010; 37: 1077-1086.
- Pawlyn C, Fowkes L, Otero S, Jones J. Whole-body diffusion-weighted MRI: a new gold standard forassessing disease burden in patients with multiple myeloma? Leukemia. 2016; 30:1446-1448.
- Shen G, Deng H, Hu S, Jia Z. Comparison of choline-PET/CT, MRI, SPECT, and bone scintigraphy in the diagnosis of bone metastases in patients with prostate cancer: a meta-analysis. Skeletal Radiol. 2014; 43(11): 1503-1513.
- Barchetti F, Stagnitti A, Megna V, Ansari N, Marini A, Musio D, et al. Unenhanced whole-body MRI versus PET-CT for the detection of prostate cancer metastases after primary treatment. Eur Rev Med Pharmacol Sci. 2016; 20: 3770-3776.
- Eschmann SM, Pfannenberg AC, Rieger A, Aschoff P, Müller M, Paulsen F, et al. Comparison of 11C-choline-PET/CT and whole body-MRI for staging of prostate cancer. Nuklearmedizin. 2007; 46(5): 161-168.
- Lin C, Luciani A, Itti E, El-Gnaoui T, Vignaud A, Beaussart P, et al. Whole-body diffusion-weighted magnetic resonance imaging withapparent diffusion coefficient mapping for staging patients with diffuse large B-cell lymphoma. EurRadiol. 2010; 20: 2027-2038.
- Stecco A, BeumiF, QuagliozziM, Lombardi M. Staging of Primary Abdominal Lymphomas: Comparison of Whole-Body MRI with Diffusion-Weighted Imaging and 18F-FDG-PET-CT.Gastroenterology Research and Practice. 2015; 1-8.
- Gutzeit A, Doert A, Froehlich JM, Eckhardt BP, Meili A, Scherr P, et al. Comparison of diffusion-weighted whole body MRI and skeletal scintigraphy for the detection of bone metastases in patients with prostate or breast carcinoma. Skeletal Radiol. 2010; 39(4): 333-343.
- Fischer MA, Nanz D, Hany T, Reiner CS, Stolzmann P, Donati OF, et al. Diagnostic accuracy of whole-body MRI/DWI image fusion for detection of malignant tumours: a comparison with PET/CT. Eur Radiol. 2011; 21(2): 246-55.
- Li B, Li Q, Nie W, Liu S. Diagnostic Value of Whole-Body Diffusion-Weighted Magnetic Resonance Imaging for Detection of Primary and Metastatic Malignancies: A Meta-Analysis. Eur J Radiol. 2013; 83(2): 338-344.
- Stecco A, Romano G, Negru M. Whole-body diffusion-weighted magneticresonance imaging in the staging of oncological patients: comparison with positron emission tomography computed tomography (PET-CT) in a pilot study. Radiol Med (Torino). 2009; 114: 11-17.
- Choi EK, Kim JK, Choi HJ, Park SH, Park BW, Kim N, et al. Node-by-node correlation between MR and PET-CT in patients with uterine cervical cancer: diffusion-weighted imaging versus size-based criteria on T2WI. Eur Radiol. 2009; 19: 2024-2032.
- Hassan O, Taha S, Farag W. Diffusion-weighted MRI versus PET-CT in evaluation of clinically N0 neck in patients with HNSCC. Systematic review and meta-analysis study. Egyptian Journal of Ear, Nose, Throat and Allied Sciences. 2014; 15: 109-116.
- Abdulqadhr G, Molin D, Aström G, Suurküla M, Johansson L, Hagberg H, et al. Whole-body diffusion-weighted imaging compared with FDG-PET/CT in staging of lymphoma patients. Acta Radiol. 2011; 52(2): 173-180.
- Kanauchi N, Oizumi H, Honma T, Kato H, Endo M, Suzuki J, et al. Role of diffusion-weighted magnetic resonance imaging for predicting of tumor invasiveness for clinical stage IA non-small cell lung cancer. Eur J Cardiothorac Surg. 2009; 35: 706-710.
- Ho KC, Lin G, Wang JJ, Lai CH, Chang CJ, Yen TC. Correlation of apparent diffusion coefficients measured by 3T diffusion-weighted MRI and SUV from FDG PET/CT in primary cervical cancer. Eur J Nucl Med Mol Imaging. 2009; 36: 200-208.
- Dimopoulos MA, Hillengass J, Usmani S, Zamagni E, Lentzsch S, Davies FE, et al. Role of magnetic resonance imaging in the management of patients with multiple myeloma: a consensus statement. J Clin Oncol. 2015; 33(6): 657-64.