Review Article
Pivotal Role of Lymph Node Sinus Macrophages and Anti-Tumor Immune Responses in Patients with Malignant Tumors
Takuya Shiota, Yoshihiro Komohara*, Yoichi Saito, Koji Ohnishi and Motohiro Takeya
Department of Cell Pathology, Graduate School of Medical Sciences, Kumamoto University, Japan
*Corresponding author: Yoshihiro Komohara, Department of Cell Pathology, Graduate School of Medical Sciences, Kumamoto University, Honjo 1-1-1, Chuouku, Kumamoto, 860-8556, Japan
Published: 05 May, 2017
Cite this article as: Shiota T, Komohara Y, Saito Y, Ohnishi
K, Takeya M. Pivotal Role of Lymph
Node Sinus Macrophages and Anti-
Tumor Immune Responses in Patients
with Malignant Tumors. Clin Oncol.
2017; 2: 1279.
Abstract
Recent progress in anti-tumor therapy has revealed the significance of anti-tumor immune responses
in tumor progression and clinical course in several kinds of malignant tumor. The draining Lymph
Node (LN) is an important immune system in which a number of Antigen-Presenting Cells (APCs)
are present that induce rapid immune responses to foreign antigens. Recent studies have shown that
LN sinus macrophages are associated with the induction of anti-tumor immunity. In some malignant
tumors, a high density of CD169-positive macrophages in the draining LN sinus has been shown
to be a predictive factor for better clinical prognosis. The density of CD169-positive macrophages
in the LN sinus is also positively associated with the density of infiltrating T- or natural killer (NK)
cells in tumor tissues. Moreover, antigen delivery targeting LN macrophages is considered to be a
promising approach for vaccination. In this article, we have summarized the significance of CD169-
positive LN macrophages in anti-tumor immunity, and suggested the possible correlation between
macrophage phenotype and anti-tumor immunity in the draining LN.
Keywords: CD169; Lymph node; Anti-tumor immunity; Sinus macrophage
Introduction
The distribution of macrophages in LNs
In the spleen and LNs, immune responses are induced by the activation of lymphocytes and
Natural Killer (NK) cells, which in turn are dependent on the activity of Antigen Presenting Cells
(APCs) such as dendritic cells and macrophages. Draining LNs are located near the tumor nodule
in many kinds of malignant tumors, and the LN sinus is filled with lymphatic fluid in which a large
number of macrophages are found [1,2]. Under normal conditions, LN sinus macrophages capture pathogens, but they also capture the abnormal antigens and debris derived from tumor tissues that flow into the lymphatic fluid in patients with malignant tumors. As such, the LN sinus is proactively
associated with the induction of antigen-specific immune responses [3,4]. It is well established that,
together with dendritic cells, many macrophages are distributed in lymph reticular organs such as
the spleen and LNs, and that those detected in the sub capsular sinus and the medullary sinus of LNs
express CD169 [5,6]. CD169 is a Type I lectin, which specifically recognizes sialic acid-containing
sugar chains, and is involved in exosome capture and the immune response to exosomal antigens
[7].
LN macrophages and anti-tumor immunity in animal studies
In some malignant diseases, the LNs are thought tobe important for anti-tumor immune
responses because of the induction of Cytotoxic T-Lymphocytes (CTLs) into tumor tissue by APCs
[8,9]. Sinus macrophages in the LN engulf antigens via several receptors, including scavenger
receptors, and present antigen-derived peptides to T- and B-lymphocytes [10,11]. Asano “et al”. [12] demonstrated that CD169-positive sub capsular sinus macrophages in LNs are preferentially
involved in antigen-presentation and the induction of CTLs, and that these CD169-positive
macrophages are more significant for anti-tumor immune responses than the CD169 antigen itself
[9]. Benhard “et al”. [13]. Additionally found that macrophages generate CTLs that react to a broader
range of epitopes than dendritic cells [12]. CD169 has consequently been considered to be useful as a potential target for antigen delivery of vaccines [13-15]. On the other hand, Pucci “et al”. [16] demonstrated that sub capsular sinus macrophages engulf melanoma-derived extracellular vesicles and subsequently suppress the induction of proteome B-cells. They suggested that sub capsular sinus macrophages may act as a physical barrier to B-cell activation under specific circumstances.
LN macrophage-mediated B-lymphocyte activation might thus be a
novel target for anti-tumor immunotherapy.
LN macrophages and anti-tumor immunity in human malignant tumors
A high number of lymphocytes (especially CD8-positive cells)
infiltrating into tumor tissues or circulating in peripheral blood has
been associated with favorable clinical prognosis in several malignant
tumors [17-20]. However, it has not yet been ascertained how CTLs
are generated in tumor patients. In order to verify whether an antitumor
immune mechanism is linked to CD169-positive macrophages
in humans, we analyzed the correlation of CD169-positive
macrophages of regional LNs to anti-tumor immune reactions and
clinical prognoses in cases of human colorectal cancer [21]. CD169
expression was analyzed immunohistochemically, but interestingly,
it was found that the CD169-positive rate in LN sinus macrophages
differed widely from case to case. An increased density of CD169-
positive macrophages in the sinus area and higher percentages of
CD169-positive cells among CD68-positive sinus macrophages, were
significantly correlated with higher T-stages, non-LN metastasis
status, and notably, high CD8-positive lymphocyte infiltration into
primary tumor tissues. Patients with a higher density or percentage
of CD169-positive macrophages showed significantly better overall
survival. These observations suggested that CD169 expression in
sinus macrophages was closely involved with the induction of antitumor
immune responses and exerted a beneficial effect on the
clinical course. We subsequently carried out similar research using
resected samples from patients with melanoma, endometrial cancer,
or breast cancer [22-24]. Similar to the study of colorectal tumors,
higher expression of CD169 in LN macrophages was significantly
associated with better a clinical course in patients with melanoma or
endometrial cancer. In endometrial tumors, increased density and
percentage of CD169-positive LN macrophages correlated well with
higher density of infiltrating CD8-positive lymphocytes and CD57-
positive NK cells in primary tumor tissues, and with lower clinical
stage and non-LN metastasis. In breast cancers, increased density
and percentage of CD169-positive LN macrophages was associated
with small tumor size, early clinical stage and non-LN metastasis,
and significantly correlated with high CD8-positive lymphocyte
infiltration into primary tumor tissues in cases with high Ki-67 index.
LN microenvironment and CD169 expression
Our in vitro studies using human monocyte-derived macrophages,
found that CD169 expression was induced by type 1 interferon (IFN),
consistent with a previous report [21,25]. However, this observation was inconsistent with data from rodents in which CD169 expression
was shown not to be affected by IFN [26]. Therefore, we next examined
which cells in LNs express type 1 IFN. Immunostaing of IFN-alpha in
LN samples was performed. Positive IFN-alpha signals were detected
on CD303-positive plasmacytoid dendritic cells and a part of CD68-
positive macrophages. IFN-alpha signaling is known to be important
for anti-tumor responses, and single nucleotide polymorphisms in
type 1 IFN-alpha receptor genes have been associated with altered
overall survival of patients with glioma [27]. Therefore, genetic
background is also likely to influence IFN-alpha signaling and antitumor
responses in LNs.
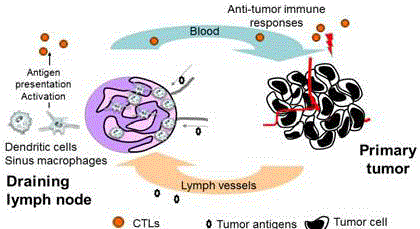
Figure 1
Figure 1
Schematic of the function of LN sinus macrophages. CD169-
positive LN macrophages are considered to cross-present tumor antigens
to effector cells. Proliferating cytotoxic T-lymphocytes (CTLs) migrate to the
tumor microenvironment via the blood stream. CTLs also attack tumor cells
in the primary tumor, blood, and metastatic sites.
Targeting Macrophages – A Novel and Promising Method for Vaccination
A number of methods have been investigated for targeting antigens to LN macrophages as part of the ongoing development of lymphatic-targeted vaccines. Shiku and colleagues reported that vaccination using cholesteryl pullulan (CHP) nanogels induced tumor regression in some patients with malignant tumors [28-34], and CD169-targeting liposomes with lipid antigen have been shown to induce activation of invariant NK T-cells (iNKTs) in a CD1d dependent manner [35]. Thus, lymphatic-targeting is considered to be a promising approach to improving vaccine efficacy, and CD169- positive macrophages are now of interest as APCs to which antigens might be efficiently delivered.
Conclusion
Unlike resident macrophages in organs outside of the lymph reticular system, sinus macrophages in draining LNs possess antitumor activity via the induction of anti-tumor CTLs and are thus important cells for tumor immunotherapy (Figure 1). Antigen delivery targeting LN macrophages is thus considered to be a promising approach for vaccination. Immunotherapy using immune checkpoint inhibitors is currently of great interest as a novel approach to anti-tumor therapy; however there are few biomarkers for evaluating anti-tumor immune responses. The evaluation of CD169- positive macrophages in LNs might thus be useful for evaluating the status of anti-tumor immune responses and for predicting the effect of chemotherapy and immunotherapy.
Acknowledgments
This work was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan (No.16H05162).
References
- Martinez-Pomares L, Gordon S. CD169+ macrophages at the crossroads of antigen presentation. Trends Immunol. 2012;33(2):66-70.
- Gasteiger G, Ataide M, Kastenmüller W. Lymph node - an organ for T-cell activation and pathogen defense. Immunol Rev. 2016;271(1):200-20.
- Marmey B, Boix C, Barbaroux JB, Dieu-Nosjean MC, Diebold J, Audouin J, et al. CD14 and CD169 expression in humanlymph nodes and spleen: specific expansion of CD14+CD169- monocyte-derivedcells in diffuse large B-cell lymphomas. Hum Pathol. 2006;37:68-77.
- Junt T, Moseman EA, Iannacone M, Massberg S, Lang PA, Boes M, et al. Subcapsular sinus macrophages in lymphnodes clear lymph-borne viruses and present them to antiviral B cells. Nature. 2007;450:110-114.
- O'Neill AS, van den Berg TK, Mullen GE. Sialoadhesin - a macrophage-restricted marker of immunoregulation and inflammation. Immunology. 2013;138(3):198-207.
- Hartnell A, Steel J, Turley H, Jones M, Jackson DG, Crocker PR. Characterizationof human sialoadhesin, a sialic acid binding receptor expressed by resident andinflammatory macrophage populations. Blood. 2001; 97: 288-296.
- Saunderson SC, Dunn AC, Crocker PR, McLellan AD. CD169 mediates the capture of exosomes in spleen and lymph node. Blood. 2014;123(2):208-16.
- Lores B, García-Estevez JM, Arias C. Lymph nodes and human tumors (review). Int J Mol Med. 1998;1(4):729-33.
- Asano K, Nabeyama A, Miyake Y, Qiu CH, Kurita A, Tomura M, et al. CD169-positive macrophages dominate antitumor immunity by crosspresenting dead cell-associated antigens. Immunity. 2011;34(1):85-95.
- Martens JH, Kzhyshkowska J, Falkowski-Hansen M, Schledzewski K, Gratchev A, Mansmann U, et al. Differential expression of a gene signature for scavenger/lectin receptors by endothelial cells and macrophages in human lymph node sinuses, the primary sites of regional metastasis. J Pathol. 2006; 208:574-589.
- Tanaka M, Asano K, Qiu CH. Immune regulation by apoptotic cell clearance. Ann N Y Acad Sci. 2010;1209:37-42.
- Bernhard CA, Ried C, Kochanek S, Brocker T. CD169+ macrophages are sufficient for priming of CTLs with specificities left out by cross-priming dendritic cells. Proc Natl Acad Sci U S A. 2015;112(17):5461-6.
- Chen WC, Kawasaki N, Nycholat CM, Han S, Pilotte J, Crocker PR, et al. Antigen delivery to macrophages using liposomal nanoparticles targeting sialoadhesin/CD169. PLoS One. 2012;7:e39039.
- Poderoso T, Martínez P, Álvarez B, Handler A, Moreno S, Alonso F, et al. Delivery of antigen to sialoadhesin or CD163 improves the specific immune response in pigs. Vaccine. 2011;29(29-30):4813-20.
- Detienne S, Welsby I, Collignon C, Wouters S, Coccia M, Delhaye S, et al. Central Role of CD169+ Lymph Node Resident Macrophages in the Adjuvanticity of the QS-21 Component of AS01. Sci Rep. 2016;6:39475.
- Pucci F, Garris C, Lai CP, Newton A, Pfirschke C, Engblom C, et al. SCS macrophages suppress melanoma by restricting tumor-derived vesicle-B cell interactions. Science. 2016;352(6282):242-6.
- Naito Y, Saito K, Shiiba K, Ohuchi A, Saigenji K, Nagura H, et al. CD8+ T cells infiltrated within cancer cell nests asa prognostic factor in human colorectal cancer. Cancer Res. 1998;58:3491-3494.
- Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, et al. Type, density, and location of immunecells within human colorectal tumors predict clinical outcome. Science. 2006; 313:1960-1964.
- Gooden MJ, de Bock GH, Leffers N, Daemen T, Nijman HW. The prognostic influence of tumour-infiltrating lymphocytes in cancer: a systematic review with meta-analysis. Br J Cancer. 2011;105(1):93-103.
- Sasada T, Suekane S. Variation of tumor-infiltrating lymphocytes in human cancers: controversy on clinical significance. Immunotherapy. 2011;3:1235-1251.
- Ohnishi K, Komohara Y, Saito Y, Miyamoto Y, Watanabe M, Baba H, et al. CD169-positive macrophages in regional lymph nodes are associated with afavorable prognosis in patients with colorectal carcinoma. Cancer Sci. 2013;104:1237-1244.
- Saito Y, Ohnishi K, Miyashita A, Nakahara S, Fujiwara Y, Horlad H, et al. Prognostic Significance of CD169+ LymphNode Sinus Macrophages in Patients with Malignant Melanoma. Cancer Immunol Res. 2015;3:1356-63.
- Ohnishi K, Yamaguchi M, Erdenebaatar C, Saito F, Tashiro H, Katabuchi H, et al. Prognostic significance of CD169-positive lymph node sinus macrophages in patients with endometrial carcinoma. Cancer Sci. 2016;107(6):846-52.
- Shiota T, Miyasato Y, Ohnishi K, Yamamoto-Ibusuki M, Yamamoto Y, Iwase H, et al. The Clinical Significance of CD169-Positive Lymph Node Macrophage in Patients with Breast Cancer. PLoS One. 2016;11(11):e0166680.
- Puryear WB, Akiyama H, Geer SD, Ramirez NP, Yu X, Reinhard BM, et al. Interferon-inducible mechanism of dendritic cell-mediated HIV-1 dissemination is dependent on Siglec-1/CD169. PLoS Pathog. 2013;9(4):e1003291.
- van den Berg TK, van Die I, de Lavalette CR, Döpp EA, Smit LD, van der Meide PH, et al. Regulation of sialoadhesin expression on rat macrophages. Induction by glucocorticoids and enhancement byIFN-beta, IFN-gamma, IL-4, and lipopolysaccharide. J Immunol. 1996;157:3130-8.
- Fujita M, Scheurer ME, Decker SA, McDonald HA, Kohanbash G, Kastenhuber ER, et al. Role of type 1 IFNs in antiglioma immunosurveillance--using mouse studies to guide examination of novel prognostic markers in humans. Clin Cancer Res. 2010;16(13):3409-19.
- Kitano S, Kageyama S, Nagata Y, Miyahara Y, Hiasa A, Naota H, et al. HER2-specific T-cell immune responses in patients vaccinated with truncated HER2 protein complexed with nanogels of cholesteryl pullulan. Clin Cancer Res. 2006;12(24):7397-405.
- Kawabata R, Wada H, Isobe M, Saika T, Sato S, Uenaka A, et al. Antibody response against NY-ESO-1 in CHP-NY-ESO-1 vaccinated patients. Int J Cancer. 2007;120(10):2178-84.
- Uenaka A, Wada H, Isobe M, Saika T, Tsuji K, Sato E, et al. T cell immunomonitoring and tumor responsesin patients immunized with a complex of cholesterol-bearing hydrophobizedpullulan (CHP) and NY-ESO-1 protein. Cancer Immun. 2007;7:9.
- Kageyama S, Kitano S, Hirayama M, Nagata Y, Imai H, Shiraishi T, et al. Humoral immune responses in patientsvaccinated with 1-146 HER2 protein complexed with cholesteryl pullulan nanogel. Cancer Sci. 2008;99:601-607.
- Aoki M, Ueda S, Nishikawa H, Kitano S, Hirayama M, Ikeda H, et al. Antibody responses against NY-ESO-1 andHER2 antigens in patients vaccinated with combinations of cholesteryl pullulan(CHP)-NY-ESO-1 and CHP-HER2 with OK-432. Vaccine. 2009; 27:6854-61.
- Kageyama S, Wada H, Muro K, Niwa Y, Ueda S, Miyata H, et al. Dose-dependenteffects of NY-ESO-1 protein vaccine complexed with cholesteryl pullulan(CHP-NY-ESO-1) on immune responses and survival benefits of esophageal cancerpatients. J Transl Med. 2013;11: 246.
- Saito T, Wada H, Yamasaki M, Miyata H, Nishikawa H, Sato E, et al. Highexpression of MAGE-A4 and MHC class I antigens in tumor cells and induction ofMAGE-A4 immune responses are prognostic markers of CHP-MAGE-A4 cancervaccine. Vaccine. 2014;32:5901-7.
- Barral P, Polzella P, Bruckbauer A, van Rooijen N, Besra GS, Cerundolo V, et al. CD169(+) macrophages present lipid antigens to mediate early activation of iNKT cells in lymph nodes. Nat Immunol. 2010;11(4):303-12.