Review Article
Hepatic Artery Infusion for Recurrent or Chemo Resistant Hepatic Malignancy
Wanebo HJ1*, Sanikommu SR2, Taneja C3, Begossi G4, Belliveau J5 and Rathore R6
1Roger Williams Medical Center, USA
2Department of Surgery, Case Western Medical Center, USA
3Rhode Island Hospital, USA
4Alta Bates Summit Medical Center, USA
5Division of Surgical Oncology, Landmark Medical Center, USA
6Department of Medical Oncology, Roger Williams Medical Center, USA
*Corresponding author: Harold J. Wanebo, Director of Surgical Oncology, Landmark Medical Center, Woonsocket, RI 02895, USA
Published: 17 Feb, 2017
Cite this article as: Wanebo HJ, Sanikommu SR, Taneja
C, Begossi G, Belliveau J, Rathore R.
Hepatic Artery Infusion for Recurrent or
Chemo Resistant Hepatic Malignancy.
Clin Oncol. 2017; 2: 1208.
Abstract
Background: Previously treated Hepatic Colorectal Metastases (CRC) and advanced Hepato
Cellular Cancer (HCC) are tumor challenges frequently unresponsive to systemic Chemo Therapy
(CT). We reviewed survival outcome in chemo resistant/high risk patients following Hepatic Artery
Infusion (HAI) in 21 CRC pts, 10 HCC pts, and 6 miscellaneous metastatic cancers.
Methods: Patient groups: 21 CRC pts, (16M, 5F), mean age 63, 16 had metachronous (DFI-17 mos),
and 5 Synchronous CA; liver extent: 76% multiple (>5) mets or extensive bilateral, CEA (ng/m),
>100, 8 pts >50 (3pts) and, NA - 7 pts. Previous CT: FU/LV (11 pts), Oxaliplatin (OX) or Irinotecan
(IR) 10 pts. Liver surgery: Partial Resection/RFA - 9 pts. HCC 9 pts, cholangio CA 1 pt, M/F 5/5,
average age 63. Previous RX Hepatic lobectomy + HAI were done in metastatic lung (1), Breast (1),
advanced gallbladder (GBCA) (T3-4) (2 pts); HAI alone was done in Br CA (1) carcinoid (1).
Treatment Protocols: CRC Protocol: HAI-FUDR 12-15mg/kg/d, dexamethasone 2mg/kg/d,
Leukovorin 20mg/m2/d (14 d) plus bolus infusion (d1), Oxaliplatin (OX) 130mg/m2 (or Cisplatin
(CIS) 100mg m2 d1); Systemic RX: d20-30. OX I>V. 130mg/m2, capecitabine 750-1000mg/m2/d x
10 days (also used in Miscel.Grp.). HCC Protocol: HAI-14 d as in CRC Protocol. Bolus infusion d1-
doxorubicin 75mg/m2 or OX or CIS as in CRC schema.
Results: CRC: OS-CRC post start HAI was 17 mos, (2yr/5yr = 27%/6%). HCC OS was 7 mos.
Median (3-12 mos in 9 evaluable pts; 1 HCC pt, with recurrence 2 yr. post hepatectomy was treated
over 3.5 yrs. with HAI + RFA/TACE - (OS- 67mos). Miscellaneous group included lung (11 mos),
Br CA (23, 9 mo) adv. carcinoid (3 mos), GBCA -2 pts >60 mos). Complications included infected
pocket (2 pts) and duodenal fistula (1 pt).
Conclusion: Hepatic artery infusion alternating with systemic chemotherapy has apparent survival
benefit in selected patients with persistent or progressive chemo resistant cancer from metastatic
CRC, HCC or selected cancers (breast, lung, liver, gallbladder cancer) and warrants further study.
Introduction
Colorectal hepatic metastases occurs as synchronous (20-25%) or metachronous lesions (60%)
within 3 years following primary resection of colorectal cancer with most (75%) being unresectable
[1-3]. Curative hepatic resection is possible in 15-25% of patients with survival ranging from 30-40%
or higher in selected series from specialized centers. Modern Chemotherapy (CT) with 5FU, LV +
Oxaliplatin (FOLFOX) OR 5FU, Irinotecan (FOLFIRI) has increased Overall Survival (OS) from
the historic 6 months to 21 months in non-resectable patients [4-10]. Recent studies combining
biologics with multi drug therapy has recorded responses up to 60% with Overall Survival of 25
months as in Crystal Study: utilizing Cetuximab and FOLFIRI in patients with KRAS Wildtype
metastatic CRC Van Cutsem et al. [11]. Unfortunately, 30-35% of CRC pts have KRAS mutated
cancers not responsive to biologics and have a recurrence rate of 36-40% [11-16]. Hepatic Artery
Infusion (HAI) has provided an alternative to systemic CT failures as well as demonstrating
significant survival advantage following hepatic resection and may be a useful adjunct in high risk
patients or in good performance patients progressing with multi-drug therapy [17-33].
An earlier experience with hepatic infusion therapy in patients
with advanced but not previously treated metastatic colorectal cancer
has provided base line information for the current study. HAI-FUDR
has a steep dose response curve resulting in a high first pass extraction
previously ratio (100-400), fold concentration in the liver (with 15x
increase in the tumor compared to <10 fold increase in liver after
systemic therapy with 5FU, MMC(3) or Oxaliplatin. HAI has a half
life of 15-19 hours. HAI/Irinotecan has an infusion advantage limited
to 5 days [21,22, 24,30,32].
Methods
Patients with advanced hepatic malignancy unresponsive to
systemic therapy were restaged by clinical and lab exam (LFT,
Tumor markers), CT, MRI (conventional and with angiography)
or CT angiography. In most cases an operative HAI catheter with
infusion pump was placed or occasionally attached to a porta cath
via percutaneous placed catheter. Selected resections were done if
considered reasonable to augment oncologic outcome for patients
with poorly responding lesions after multi drug therapy.
Preoperative imaging included CT scan, (Hepatic focus +/-
contrast MRI +/- Angiogram (or CT angiogram), liver function
tests including CEA levels, and ICG clearance studies were done in
selected patients and were reassessed with CT scans and CEA levels
after 2 treatment cycles. Patients were generally re-explored with
planned resection if considered resectable or had resections of high
risk lesions if easily and safely resected to reduce tumor burden.
Treatment Protocols are listed in Table 3. Colorectal cancer therapy
included baseline continuous infusion of FUDR dexamethasone with
systemic leukovorin (14 days) and bolus infusion of Oxaliplatin given
on day 1 of HAI therapy. Systemic therapy was given after 5-6 day rest
period on days 20-30 with bolus infusion of Oxaliplatin/Irinotecan
and daily Capecitabine.
Initial Study: Pilot Study
The initial protocol utilized baseline data from earlier studies by
Kemeny et al. [17,20,27-29]. Chemo therapy Regimen: (Table 1A)
HAI.
A pilot study was carried out in 36 patients with untreated
metastatic colorectal cancer, of whom 13 (36%) generated a
measurable radiologic response in liver as demonstrated by pre
therapy and post therapy volumetric measurements at 2 and 4 months
(Table 2). Cross sectional measurements with volume calculation
provided data on target lesions before treatment and at designated
time points during and after treatment (2 and 4 months minimum).
Greater than 50% volumetric regression occurred in 7 of 13 patients
(54%) with <50% regression in 6 patients (46%). Progression of liver
disease occurred in 7/36 (20%) and stable disease was measured in 16
(44%) of patients (Table 1B). The survival outcome in the 36 patients
is shown in Figure 1.
Follow-up study
Patient groups under study include colorectal cancer 21
pts, primary liver cancer 10 pts (9 HCC, 1 cholangio CA), and
miscellaneous group (6 pts). (Table 2,3) The colorectal cancer group
(Table 2) consisted of 21 pts, 5 with synchronous cancer and 16 with
metachronous lesions. Patients were considered failures of previous
therapy, or had progressed after previous therapy with 5FU/LV or
combination therapy with 5FU and Oxaliplatin or Irinotecan. Ten pts
had progressed following previous resections or RFA (9 pts). Primary
liver cancer included 10 pts (HCC) in 9 and intrahepatic cholangio
CA in 1 (Table 3). A third group consisted of 6 pts having resection of
miscellaneous cancers originating in lung (1), breast (2), gallbladder
(2) or carcinoid (1) (Table 2).
Hepatic treatment for HCC mimicked the CRC protocol with
bolus therapy of Doxorubicin being given on day 1 of the 14 day
infusion with FUDR, Leukovorin/ Dexamethasone. Systemic therapy
was given as in the CRC protocol with Oxaliplatin/Irinotecan on
day 20 and daily Capecitabine on days 20-30. Major complications
of HAI: Pump malfunction (4 pts required pump replacement),
misperfusion 2 pts (required radiologic intervention to occlude
gastroduodenal artery flow to duodenum), infected pocket 2 pts,
duodenal fistula 1 pt from embedded catheter in duodenal wall.
Current study outcome: (Summarized in Table 4)
Colorectal Cancer Group: Of 21 previously treated pts subjected
to HAI post liver recurrence (16 metachronous, 5 synchronous), the
median survival was 18 months with 1 pt surviving over 60 month
(6%). Among the 10 HCC pts (1 cholangio carcinoma), the median
survival was 9 mos with 1 surviving 41 months. The single long
term survivor had developed recurrence of HCC following a central
hepatic resection at 26 months. Patient received HAI and selective
RFA/TACE and survived 3.5 years post recurrence of the HCC (total
survival was 67 months).
Discussion
Hepatic arterial chemo infusion was utilized to treat patients
having failed or progressed on standardized therapy for hepatic
metastases of colorectal cancer (21 pts), hepatocellular carcinoma
(9 pts), and 6 miscellaneous hepatic metastases. The focus of this
study was on overall survival rather than response rate in the liver
which was considered too variable for precise measurements of target
lesions in view of the patterns of recurrence in the liver and variability
(regression of selected lesions with simultaneous progression of
adjacent lesions). An initial study of HAI alone with FUDR, LV
in patients with primary CRC recurrence of CRC in liver utilizing
volumetric measurements demonstrated objective regression in
13 of 36 pts (36%) with calculated volumetric regression showing
>50% volumetric regression in 7 pts (54%) and <50% in 6 pts (46%).
Of the remaining 44%, 16 pts had stable disease and 20% (7 pts)
showed progression. The median OS was 17 mos and 3 yr survival
was 18%. The current treatment group pts included 21 patients with
colorectal hepatic metastases CRC, of whom one half had progressed
on FOLFOX/FOLFIRI and one half had progressed after resection
+/- RFA. The median survival in the group was 17 months. Of the
remaining 10 pts with primary liver cancer, there were 9 HCC (1
cholangio carcinoma). Recurrence or progression of disease occurred
as failure of resection (4 pts) TACE/RFA (3 pts) and the group had
median survival post HAI of 9 months. One patient had developed
hepatic recurrence 24 mos after central hepatic resection for HCC and
survived 3.5 yrs after initiation of HAI with supplemental RFA/TACE
with an overall survival over 5.5 years. The remaining 6 pts represented
various problems including advanced/recurrent malignancy (lung 1
pt, Br CA 2 pts, gallbladder CA 2 pts) and advanced hepatic carcinoid
1 pt. Overall survival in the miscellaneous group included 4 mos
and 11 mos in the advanced carcinoid pt and lung cancer pt, 10
and 24 mos in 2 Br CA pts (post recurrence), and >60 mos in the
2 gallbladder patients post resection. Treatment was well tolerated although there were technical problems with pump malfunctions (4 pts), misperfusion (2 pts), duodenal fistula and infected pocket (2 pts
in the group of pump treated pts). Major drug toxicity was averted by
close hematologic and liver function monitoring and dose reduction
as needed. Any suggestion of intolerance to Oxaliplatin in previously
exposed pts prompted switching to Cisplatin as the bolus therapy
prior to HAI and as part of systemic therapy. Although selected pts
showed significant objective responses in the liver, there was marked
variability of responses within the same liver segments.
Of interest was a miscellaneous group of previously treated pts
including 2 with advanced gallbladder cancer, both of whom survived
>60 months after regional resection for Stage III gallbladder cancer,
and subsequent HAI. Other patients had more variable outcome
(metatastic Br CA pts had OS 23 & 9 mos), lung CA (11 mos), and 1
advanced carcinoid pt (4 mos). These patients represented a highly
selected group with good PS and liver only disease.
The largest series of Regional Therapy for metastatic CRC is
reported by Dr. Kemeny and colleagues at MSKCC [17,20,23,25,27].
She has also demonstrated significant improved DFS with adjuvant
HAI in treated pts vs. systemic therapy [23,25,27]. The data is
supported by previous reports by Kemeny [17,20] and also confirmed
by Lygidakis, [34,35] but not by Lorenz where FU intra hepatic 5FU
was used instead of 5FUDR [26].
A CALGB study reported by Kemeny utilized HAI/FUDR/Dex
vs. Systemic FU/L in a multi center trial and recorded a response rate
of 47% in HAI pts vs. 24% in systemic therapy pts with 2 yr estimated
survival of 51% HAI vs. 35% with systemic therapy. The update of
overall progression free survival was 31 mos with HAI [23,27].
Kemeny et al. [20] have also explored HAI with FUDR/Dex vs.
Systemic Irinotecan/Oxaliplatin in 49 pts with non-resectable and
previously treated CRC Hepatic Mets (86% had 6) or more segments
involved and achieved resectability of 31% (-OS is pending) [25,28,29].
They also used HAI plus FUDR/Dex and systemic Irinotecan in
another group of 39 previously treated CRC pts resulting in 18%
resection/ablation rate [28,29].
A randomized study by Fiorentini reported on chemonaive pts
with unresectable liver mets who were randomized to HAI FUDR +/-
Systemic FU/LV [18]. Med survival was 20 months with combined
therapy (HAI & Systemic 5 FU/LV) vs. 14 mos with HAI alone. HAI
with systemic therapy was utilized as second line. In a Phase 2 study
by Ducreux (36 pts), 89% had first line therapy, with HAI-FUDR/
DEX and systemic Ox/IR vs. 15 pts treated with FUDR/DEX+Ox+FU
[21]. The med survival was 36 mos in the Ox/IR gp, the RR was 90%,
with the median survival of 22 mos in pts receiving Ox/FU. In followup
study, the median OS was 41 mos in the chemo therapeutic treated
group vs. 50.8 mos in the patients receiving HAI plus Systemic IR as
second line therapy.
Gallagher reported on 39 pts previously treated with Oxaliplatin
using HAI FUDR + Systemic Irinotecan therapy which resulted in
partial response of PR of 44% and median survival of 20 mos [19].
Boige reported 44 pts with unresectable metastatic CRC of
whom 95% had prior Oxaliplatin or Irinotecan or both [24] Therapy
included 9 cycles HAI Ox + Systemic FU and leukovorin in 7 pts
(10%) who went on to have an RO resection [24]. Ducreux utilized
HAI- Ox + systemic FU + LV in 26 pts of whom 5 (19%) had surgical
resection [21]. Phase I/II studies by Fiorentini evaluated HAI with Irinotecan given at 20mg/m2/d x 5 days as continuous infusion or as
a 200mg/m² (30 minutes) infusion in 100cc saline q 3 wks in a Phase
II therapy study of 12 pts (6 post FOLFOX progression) [30]. This
resulted in 4 PR and 6 progressions. Toxicity included diarrhea and
myelosuppression in 6 pts (50%). They considered the 200mg dose to
be well tolerated.
Advanced HCC is a greater challenge and is poorly responsive
to systemic CT but frequently treatable with TACE/RFA. The BCLC
(Barcelona Clinic Liver Ca Group) has utilized TACE based therapy
for Stage B (Child B) good PS or (Child A) based on 7 randomized
trials and demonstrated median OS ranging from 11-20 mos and RR
27% [33]. For more advanced stage C (portal invasion, N1, M1, PS
1-2), Sorafenib or clinical trial equivalent was recommended [34-36].
In our current study, HAI was examined in a selected group of
HCC pts to explore regional CT; in this setting, TACE may provide
added benefit and merits study.
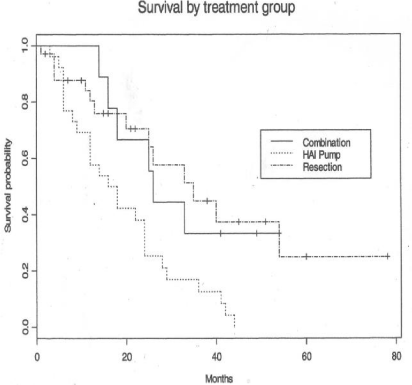
Figure 1
Figure 1
Hepatic Artery Infusion Metastatic Colorectal Cancer.
HAI Pump=36 pts - Initial study of Hepatic Metastasis group treated by HAI
(36 pts), Hepatic resection (72 pts) +/- chemotherapy - Total Group.
Hepatic Artery Infusion in Metastatic Colorectal Cancer
HAI Pump 36 pts (combination HAI and Systemic therapy 9 patients),
resection in 86 patients. Median OS=17 mos (HAI pump), 26 mos combination
HAI and systemic CT and 34 mos with resection.
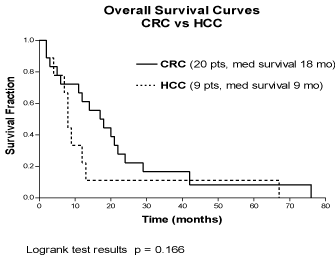
Figure 2
Figure 2
Overall Survival Curves – in 20 colorectal cancer (CRC) vs. 9
Primary Liver Cancer pts (9 with HCC).
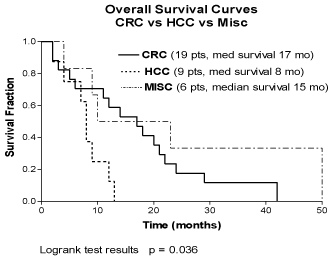
Figure 3
Figure 3
Overall Survival – Colorectal Cancer (CRC) vs. Primary Liver
Cancer HCC 9, Cholangio CA 1 vs. Miscellaneous Hepatic Metastases (6
pts: Br CA (2 pts), Lung CA (1 pt), Carcinoid (1 pt), Gallbladder CA (2 pts).
Conclusion
Hepatic artery infusion alternating with systemic chemo therapy has apparent survival benefit in selected patients with persistent or progressive chemo resistant malignancy from metastatic CRC, HCC, or selected miscellaneous cancers (breast, lung, carcinoid, and gallbladder cancer) and warrants further study.
References
- Riles LAG, Melbert D, Krapcho M, Stinchcomb DG, Howlader N, Horner MJ, et al. SEER Cancer Statistics Review 1975-2005. U.S. National Cancer Institutes of Health, Bethesda, MD. Ann Surg Oncol. 2010; 17: 492-501.
- Nordlinger B, Rougier P. Liver metastases from colorectal cancer: the turning point. J Clin Oncol. 2002; 20: 1442-1445.
- Ensminger WD, Gyves JW. Clinical pharmacology of hepatic arterial chemotherapy. Semin Oncol. 1983; 10: 176-183.
- Tournigand C, André T, Achille E, Lledo G, Flesh M, Mery-Mignard D, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004; 22: 229-237.
- André T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, et al. Oxaliplatin, fluorouracil and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004; 350: 2343-2351.
- Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, et al. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000; 355: 1041-1047.
- Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, et al. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol. 2004; 22: 23-30.
- Tanaka K, Adam R, Shimada H, Azoulay D, Lévi F, Bismuth H. Role of neoadjuvant chemotherapy in the treatment of multiple colorectal metastases to the liver. Br J Surg. 2003; 90: 963-969.
- Adam R, Avisar E, Ariche A, Giachetti S, Azoulay D, Castaing D, et al. Five-year survival following hepatic resection after neoadjuvant therapy for non-resectable colorectal. Ann Surg Oncol. 2001; 8: 347-353.
- Bondarenko I, Makhson A, Hartmann JT, Aparicio J, de Braud F, et al. Flourouracil, leucovorin, irinotecan, and oxaliplatin with and without cetuximab in the first-line treatment of metastatic colorectal cancer. J Clin Oncol. 2009; 27: 663-671.
- Amado RG, Wolf M, Peeters M, Van Cutsem E, Siena S, Freeman DJ, et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol. 2008; 26: 1626-1634.
- Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004; 350: 2335-2342.
- Saltz LB, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008; 26: 2013-2019.
- Bardelli A, Siena S. Molecular mechanisms of resistance against cetuximab and panitumumab in colorectal cancer. J Clin Oncol. 2010; 28: 1254-1261.
- Sartore-Bianchi A, Martini M, Molinari F, Veronese S, Nichelatti M, Artale S, et al. PIK3CA mutations in colorectal cancer are associated with clinical resistance to EGFR-targeted monoclonal antibodies. Cancer Res. 2009; 69: 1851-1857.
- Laurent-Puig P, Cayre A, Manceau G, Buc E, Bachet JB, Lecomte T, et al. Analysis of PTEN, BRAF and EGFR status in determining benefit from cetuximab therapy in wild-type KRAS metastatic colon cancer. J Clin Oncol. 2009; 27: 5924-5930.
- Kemeny N, Daly J, Reichman B, Geller N, Botet J, Oderman P. Intrahepatic or systemic infusion of fluorodeoxyuridine in patients with liver metastases from colorectal carcinoma: a randomized trial. Ann Int Med. 1987; 107: 459-465.
- Fiorentini G, Cantore M, Rossi S, Vaira M, Tumolo S, Dentico P, et al. Hepatic arterial chemotherapy in combination with systemic chemotherapy compared with hepatic arterial chemotherapy alone for liver metastases from colorectal cancer: results of a multi-centric randomized study. In Vivo. 2006; 20: 707-709.
- Gallagher DJ, Capanu M, Raggio G, Kemeny N. Hepatic arterial infusion plus systemic irinotecan in patients with unresectable hepatic metastases from colorectal cancer previously treated with systemic oxaliplatin: a retrospective analysis. Ann Oncol. 2007; 18: 1995-1999.
- Kemeny N, Huang Y, Cohen AM, Shi W, Conti JA, Brennan MF, et al. Hepatic arterial infusion of chemotherapy after resection of hepatic metastases from colorectal cancer. N Engl J Med. 1999; 341: 2039-2048.
- Ducreux M, Ychou M, Laplanche A, Gamelin E, Lasser P, Husseini F, et al. Hepatic arterial oxaliplatin infusion plus intravenous chemotherapy in colorectal cancer with inoperable hepatic metastases: a trial of the gastrointestinal group of the Federation Nationale des Centres de Lutte Contre le Cancer. J Clin Oncol. 2005; 23: 4881-4887.
- van Riel JM, van Groeningen CJ, de Greve J, Gruia G, Pinedo HM, Giaccone G. Continous infusion of hepatic arterial irinotecan in pretreated patients with colorectal cancer metastatic to the liver. Ann Oncol. 2004; 15: 59-63.
- Kemeny N, Melendez FD, Capanu M, Paty PB, Fong Y, Schwartz LH, et al. Conversion to resectability using hepatic artery infusion plus systemic chemotherapy for the treatment of unresectable liver metastases from colorectal carcinoma. J Clin Oncol. 2009; 27: 3465-3471.
- Boige V, Malka D, Elias D, Castaing M, De Baere T, Goere D, et al. Hepatic arterial infusion of oxaliplatin and intravenous LV5FU2 in unresectable liver metastases from colorectal cancer after systemic chemotherapy failure. Ann Surg Oncol. 2008; 15: 219-226.
- Kemeny N, Melendez FD, Capanu M, Paty PB, Fong Y, Schwartz LH, et al. Conversion to resectability using hepatic artery infusion plus systemic chemotherapy for the treatment of unresectable liver metastases from colorectal carcinoma. J Clin Oncol. 2009; 27: 3465-3471.
- Lorenz M, Müller HH, Schramm H, Gassel HJ, Rau HG, Ridwelski K, et al. Randomized trial of surgery versus surgery followed by adjuvant hepatic arterial infusion with 5-fluorouracil and folinic acid for liver metastases of colorectal cancer. German Cooperative on Liver Metastases (Arbeitsgruppe Lebermetastasen). Ann Surg. 1998; 228: 756-762.
- Kemeny NE, Niedzwiecki D, Hollis DR, Lenz HJ, Warren RS, Naughton MJ, et al. Hepatic arterial infusion versus systemic therapy for hepatic metastases from colorectal cancer: a randomized trial of efficacy, quality of life, and molecular markers (CALBG 9481). J Clin Oncol. 2006; 24: 1395-1403.
- Kemeny N, Jarnagin W, Paty P, Gönen M, Schwartz L, Morse M, et al. Phase I trial of systemic oxaliplatin combination chemotherapy with hepatic arterial infusion in patients with unresectable liver metastases from colorectal cancer. J Clin Oncol. 2005; 23: 4888-4896.
- Kemeny N, Gonen M, Sullivan D, Schwartz L, Benedetti F, Saltz L, et al. Phase I study of hepatic arterial infusion of floxuridine and dexamethasone with systemic irinotecan for unresectable hepatic metastases from colorectal cancer. J Clin Oncol. 2001; 19: 2687-2695.
- Fiorentini G, Rossi S, Dentico P, Bernardeschi P, Calcinai A, Bonechi F, et al. Irinotecan hepatic arterial infusion chemotherapy for hepatic metastases from colorectal cancer: a phase II clinical study. Tumori. 2003; 89: 382-384.
- Meyer L, Hildebrandt B, Riess H. 5-fluorouracil, folinic acid and oxaliplatin administered via hepatic arterial infusion as regional second-line therapy for advanced colorectal cancer. Oncology. 2003; 64: 473-483.
- Kern W, Beckert B, Lang N, Waggershauser T, Braess J, Schalhorn A, et al. Hepatic arterial infusion with oxaliplatin, folinic acid and 5-fluorouracil in patients with hepatic metastases from colorectal cancer: a role of carcino-embryonic antigen in assessment of response. Anticancer Research. 2000; 20: 4973-4975.
- Lencioni R, Chen XP, Dagher L, Venook AP. Treatment of intermediate/advanced hepatocellular carcinoma in the clinic: how can outcomes be improved? Oncologist. 2010; 15: 42-52.
- Lygidakis NJ, Stringaris K, Kokinis K, Lyberopoulos K, Raptis S. Locoregional chemotherapy versus locoregional combined immuno-chemotherapy for patients with advanced metastatic liver disease of colorectal origin: a prospective randomized study. Hepatogastroenterology. 1996; 43: 212-220.
- Lygidakis NJ, Sgourakis G, Vlachos L, Raptis S, Safioleas M, Boura P, et al. Metastatic liver disease of colorectal origin: the value of locoregional immunochemotherapy combined with systemic chemotherapy following liver resection. Results of a prospective randomized study. Hepatogastroenterology. 2001; 48: 1685-1691.
- Llovet JM ,Di Bisceglie AD, Bruix Jordi, Kramer BS, Lencioni R, Zhu AX, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008; 100: 698-711.