Editorial
What can we Save for the First Line Treatment of NSCLC in 2016?
David Rossi*
Department of Oncology, Salvatore Hospital, Italy
*Corresponding author: David Rossi, Department of Oncology, Salvatore Hospital, Italy
Published: 31 Dec, 2016
Cite this article as: Rossi D. What can we Save for the First
Line Treatment of NSCLC in 2016?.
Clin Oncol. 2016; 1: 1176.
Editorial
Lung cancer is the leading cause of cancer-related deaths worlwide. In the United States accounts
for 221,200 new diagnoses and 158,400 deaths in 2015, according to the National Cancer Institute.
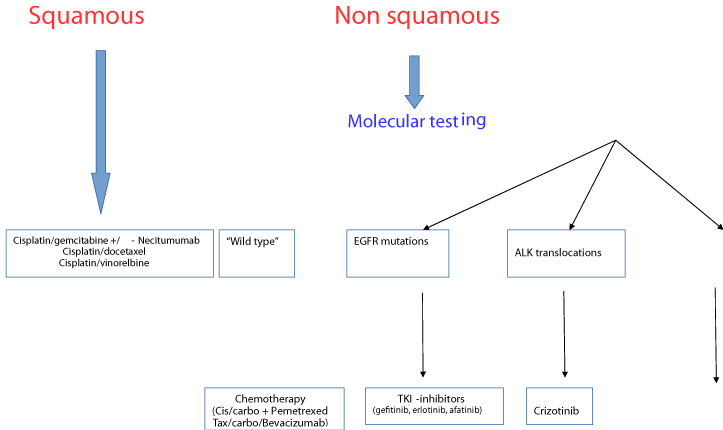
In these last years, hystologic definition has become crucial in the therapetic alghoritm of NSCLC.
To day we recognize three big groups of Lung Cancer: Non-squamous “wild type”, Non-squamous
with genetic mutations and squamous. For the non-squamous “wild type”, pemetrexed plus cisplatin
or carboplatin is the gold standard of care with chance of pemetrexed maintenance in patients with
responsive or stable disease; another option is the addition of bevacizumab to a doublet with
paclitaxel/carboplatin. For the non-squamous with genetic mutations, we can taking into account in
clinical practice of EGFR mutations, ALK translocations and ROS-1 rearrangements. Gefitinib,
erlotinib and afatinib for EGFR mutations, crizotinib for ALK translocations and ROS1-
rearrangements. For squamous cell carcinoma, a doublet with platinum derivate plus gemcitabine,
vinorelbine or docetaxel is the gold standard; last year, necitumumab (a new anti-EGFR monoclonal
antibody) has been approved by FDA in combination with cisplatin and gemcitabine (SQUIRE
trial). In these last two years, immuno check-point inhibitors documented high activity as second
line in squamous and non-squamous cell carcinoma. In 2015, FDA approved nivolumab (Opdivo®)
as second line, regardless of PD-L1 expression, in patients with NSCLC progressed during or after
platinum-based chemotherapy according to results of CheckMate 017 (squamous) and CheckMate
057 (non-squamous). In the same year, Pembrolizumab (Keytruda®) was approved by FDA as
second line in squamous and non-squamous patients with PD-L1 expression, according to results of
a large multicenter, open-label, multi-part study (Keynote 010). The therapeutic alghoritm of
NSCLC first line at the end of 2015 has been summarizied on Figure 1. But what's new in 2016? The
main results concerning immuno check-point inhibitors. On October 24, FDA approved
Pembrolizumab (Keytruda®) for the treatment of patients with metastatic NSCLC whose tumors
express PD-L1 ≥50% as determined by an FDA approved test. This is the first approval of a checkpoint
inhibitor for the first line of lung cancer. Approval was based on results of two randomized,
controlled trials that demonstrated statistically significant improvements in Progression-Free
Survival (PFS) and Overall Survival (OS) for patients randomized to pembrolizumab compared
with chemotherapy. In a trial of 305 patients (Keynote 024) who had no prior treatment for
metastatic NSCLC and PD-L1 expression greater than or equal to 50%, those who received
pembrolizumab (200 mg every 3 weeks) had a significant improvement in PFS (HR 0.50 [95% CI:
0.37, 0.68]; p <0.001) with a median PFS of 10.3 months versus 6.0 months for those receiving
platinum-based chemotherapy. A pre-specified interim analysis demonstrated a statistically
significant improvement in OS for patients randomized to pembrolizumab as compared with
chemotherapy (HR 0.60 [95% CI: 0.41, 0.89]; p <0.005). In a three-arm trial of 1033 patients who
were previously treated for metastatic NSCLC with PD-L1 expression greater than or equal to 1%,
those randomized to pembrolizumab 2 mg/kg every 3 weeks (HR 0.71 (95% CI: 0.58, 0.88]; p <0.001)
or pembrolizumab 10 mg/kg every 3 weeks (HR 0.61 [95% CI: 0.49, 0.75]; p <0.001) had an improved
OS compared with patients receiving docetaxel. The median survival was 10.4 months in the
pembrolizumab 2 mg/kg arm, 12.7 months in the pembrolizumab 10 mg/kg arm, and 8.5 months in
the docetaxel arm. The most common side effects of treatment with pembrolizumab included
decreased appetite, fatigue, nausea, dyspnea, cough, and constipation. Rare but serious adverse
events included immune-mediated pneumonitis, colitis, hepatitis, endocrinopathies, and nephritis.
Unlike Pembrolizumab, Nivolumab (Opdivo®) fails to demonstrate a benefit in PFS and OS in the
same setting of patients. In Checkmate 026 trial, 541 pts with PD-L1 expression ≥5% were
randomized to receive Nivolumab 3 mg/kg every two weeks versus best chemotherapy. Progressionfree
survival was the main endpoint but was not met (4,2 months with Nivolumab versus 5,9 months
with chemotherapy; HR, 1,15; P = .25); overall survival was similar in the two arms of treatment (14,4 and 13,2 months for Nivolumab and chemotherapy, respectively;
HR, 1.02). Several explanations were proposed to clarify so different
results in the same setting of patients. The main issue appears to be
patient selection: whereas pembrolizumab trial was conducted in
patients whose tumors showed at least 50% PD-L1 expression,
nivolumab trial enrolled patients with PD-L1 expression ≥5%.
However, the same results was documented in a small subgroup of
patients who had at least 50% PD-L1 expression (HR for PFS: 1.1; HR
for OS: 0.90); nevertheless, the trial was not statistically powered for
this analysis. Other explanations may be that OS was better than
historical data in the chemotherapy arm possibly due to a different
proportion of women and Asian patients; moreover, there was a high
crossover (60%) among patients who progressed while receiving
chemotherapy. This difference in efficacy between nivolumab and
pembrolizumab could be elucidated by the ongoing phase III study
CheckMate-227. This trial explores the potential of the combination
of Nivolumab plus Ipilimumab for PD-L1 patients and Nivolumab
plus Ipilimumab or Nivolumab plus chemotherapy in PD-L1-
negative patients. This year, at ASCO meeting, was presented the
results of CheckMate 012; a phase 1b study with the combination of
Nivolumab and Ipilimumab in PD-L1 positive patients (≥1%).
Overall Response Rate (ORR) was 57% but in those patients with
≥50% PD-L1 expression, the ORR was 92%; in patients who tested
PD-L1 negative, the ORR with the combination regimen was 15%.
These data seems to go in a single direction: PD-L1 expression is
crucial to maximize activity and efficacy of check-point inhibitors in
order to administer “right drug to right patient” with a real
improvement of cost-effectiveness. Concerning patients with nonsquamous
cell lung cancer and genetic mutations, we want to focus
on data presented at ESMO meeting for afatinib, at ELCC for
Osimertinib and at ASCO meeting for Alectinib. At ESMO meeting,
updated results of LUX-LUNG 7 was presented. In this trial,
Progression-free survival (median 11.0 months with afatinib vs. 10·9
months with gefitinib; HR) and time-to-treatment failure (median
13.7 months with afatinib vs. 11.5 months with gefitinib) were
significantly longer with afatinib than with gefitinib. Updated results
shows that median survival of patients treated with afatinib was 27.9
months compared to 24.5 months for those receiving gefitinib,
without reaching significance. However, updated results confirmed
the primary analysis concerning two of its three co-primary endpoints:
progression-Free Survival (PFS) by independent review and time to
treatment failure. We think that afatinib remains one of the three
options in first line treatment with gefitinib and erlotinib: a careful
evaluation of different toxicity between drugs (skin rash and diarrhea
for afatinb and erlotinib, AST/ALT increase for gefitinib) should be
made for the best tailored treatment (“right drug to right patient”). At
ELCC, AURA expansion study with Osimertinib was presented.
Osimertinib is a third generation Epidermal Growth Factor Receptor
(EGFR) Tyrosine Kinase Inhibitor (TKI), highly effective in patients
with T-790M mutation. The study included 60 patients from two
phase I expansion cohorts of the AURA trial that had locally advanced
or metastatic EGFR mutated NSCLC. Thirty patients received 80 mg
a day and 30 received 160 mg a day in the first-line setting. The
median follow-up was 16.6 months. The overall response rate was
77%. Median Progression Free Survival (PFS) was 19.3 months for
the 160 mg dose and has not yet been reached for the 80 mg dose.
Median duration of response has not been reached. The drug was well
tolerated with few adverse events, particularly at the approved 80 mg
dose, where just 10% of patients required dose reduction to manage
toxicities. At ASCO meeting, J-ALEX study was presented. This is a
japanese phase III trial that randomized patients to Alectinib or
Crizotinib as first line treatment in patients with ALK-translocation.
On December 11, 2015, FDA approved alectinib (Alecensa®) for
patients with ALK-rearrangement positive NSCLC that is refractory
to Crizotinib. The J-ALEX study, after a planned interim analysis, was
stopped for efficacy, as an improvement in PFS was already
demonstrated. In the alectinib patients, the median PFS was not
reached, compared with 10.2 months in the crizotinib patients,
yealding a HR of 0.34 (p <.0001). Toxicity was very low (constipation
was tha main side effects); grade 3 and 4 adverse events also occured
more frequently with crizotinib (51.9%) than with alectinib (26,2%).
Cizotinib still remains the best ooption in patients with ROS-1
rearrangements: on March 11, 2016 FDA approved crizotinib
(Xalkori®) to treat people with advanced NSCLC whose tumors have a
ROS-1 gene alteration. The safety and efficacy of Xalkori for the
trreatment of patients with ROS-1 positive tumors were evaluated in
a multi-center, single arm study of 50 patients with ROS-1 positive
NSCLC. Results showed 66% of participants experienced a complete
or partial shrinkage of their NSCLC tumors, an effect that lasted a
median of 18,3 months.
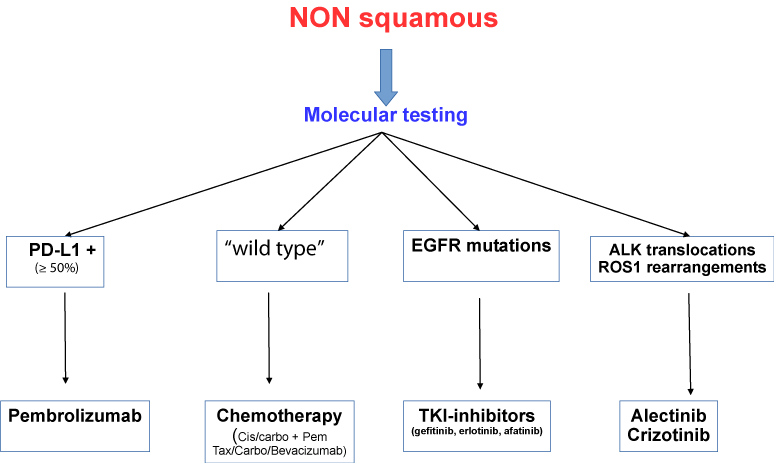
In conclusion, 2016 was an important year for the treatment of
Lung Cancer with good news for non-squamous, regardless of genetic
mutations or not. An attempt of new alghoritm for the first line
treatment of non-squamous NSCLC showed in the Figure 2.