Case Report
Incidental Bone Lesions in a 78 Year Old Male with History of Grade I Meningioma
Siddarth Joshi, Cristian Dobre, Murat Anamur and Richard Zuniga*
Tuality Healthcare, USA
Hematology-Oncology, Lowell Gener4al Cancer Center, USA
*Corresponding author: Richard Zuniga, Department of Hematology-Oncology, Lowell General Cancer Center, 295 Varnum Avenue, Lowell, MA 01854, USA
Published: 13 Dec, 2016
Cite this article as: Joshi S, Dobre C, Anamur A, Zuniga
R. Incidental Bone Lesions in a 78
Year Old Male with History of GradeI
Meningioma. Clin Oncol. 2016; 1: 1162.
Abstract
The clinical phenomenon of metastatic meningioma is not well described in the literature owing to
the relative rarity of its incidence.
We report the case of a 78 year old male with a past medical history notable for successfully resected.
WHO grade I meningioma in June of 2006 who, upon presentation for left thigh pain from a
traumatic fall eight years later, was found to have incidental imaging findings of multiple bony
lesions in the left iliac crest and spine. Subsequent investigations revealed the diagnosis of WHO
grade I meningioma metastatic to the bone. Although its unequivocal rarity would reasonably
relegate metastatic meningioma to a diagnosis of exclusion, it is hoped that this case report will
prevail upon the reader the importance of keeping this diagnostic possibility on the differential in
the appropriate clinical context.
Background
Largely incidental findings from imaging for unrelated reasons, meningiomas account for
upwards of 35% of total primary brain tumors. Most are benign, localized, intracranial tumors.
When malignancy occurs in meningioma, this typically connotes a recurrence of the tumor and
not metastasis as metastasis outside the cranium is an overall rare occurrence. The latency period for
such an event has been reported to be from months to over twenty years. The majority of metastatic
meningioma travel to the lungs, intra-abdominal viscera and bone, as was the case in this patient.
Case Report
We report here the case of a 78 year old male who initially presented to the emergency department
in March of 2016 with the complaint of left thigh pain and discomfort from an injury sustained
after losing balance and falling from a ladder at work. His medical history is notable for a 2.9 cm
x 2 cm left parietal occipital falcine meningioma diagnosed in 2006. This was surgically resected
successfully that year (Simpson Grade I); subsequent surveillance MRIs of the brain in 2007 and
2008 showed no evidence of recurrence. Between the time of the aforementioned surgical resection
and the current presentation, the patient had been overall well, notwithstanding chronic neck and
back pain. In the ED on his current presentation, CT scans were done of the left lower extremity
and abdomen/pelvis, respectively; the latter was performed due to complaints of pain radiating to
the left groin area. The former study was notable for a large hematoma corresponding to findings on
physical examination. The latter study, however, had the incidental finding of a large lytic lesion in
the right iliac crest and multiple small rounded lucent lesions in the thoracolumbar spine. Shortly
after, an MRI of the cervical spine without contrast was performed; this revealed multiple focal
lesions in the marrow suggesting metastatic disease to the cervical spine. A CT of the chest done
the following month re-demonstrated several lesions at multiple levels of the thoracolumbar spine
and also showed a suggested lesion in the sternum. An MRI of the brain done a few days later found
an approximately 13 mm circular lesion in the parafalcine region abutting the right parietal lobe,
thought to be a recurrence of the original meningioma from 2006.
The patient underwent CT-guided biopsy of the right iliac crest lesion; pathology showed
infiltration of the medullary cavity with atypical spindle cells suspicious for metastatic meningioma.
Immunohistochemistry of the cells demonstrated: SMA+ (rare), EMA+ (multifocal), Pan-
Keratin-, Desmin-, S100-, CD31-, CAMP5.2-, and Pax-8-. Pathology review of specimens from the
intracranial meningioma from 2006 and the right iliac biopsied lesion from 2016 confirmed the diagnosis of grade I meningioma. Given the fact that the patient was
minimally symptomatic from the meningioma the decision was made
to monitor only and no medical intervention was initiated.
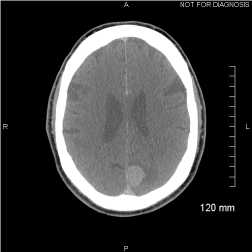
Figure 1
Figure 1
Axial CT image of the head demonstrate a mildly hyperdense left
posterior parafalcine mass compatible with a meningioma.
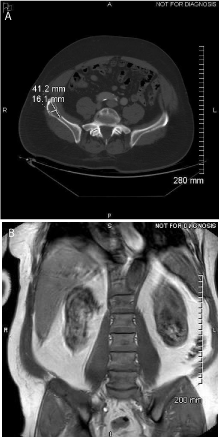
Figure 2
Figure 2
A) Axial CT image (soft tissue windows) of the abdomen/pelvis
demonstrates a mildly expansile and lytic lesion in the right iliac crest
with calipers super imposed on the lesion. B) Coronallocalizer MRI image
demonstrates low marrow signal in the right iliac crest.
Discussion
Among all of the primary CNS tumors, meningiomas are the
most common, numbering roughly one in every three primary
tumors found in the brain and spinal cord [1]. Meningiomas have a
gender predilection for females, the female to male ratio being about
2-3:1 [2]. The incidence of meningioma also has a positive correlation
with the advancement of age, with the median age at diagnosis being
65 years.
Multiple investigations have been undertaken to establish possible
etiologies for the development of meningioma. The most prominent
etiologic association is history of CNS exposure to ionizing radiation,
including radiation therapy used to treat other primary malignancies
[3,4]. Our patient has no such history.
Less commonly, there are certain genetic mutations implicated
in the development of meningioma. The most well established
is mutation of the NF2 tumor suppressor gene that results in
neurofibromatosis type 2 (NF2) [5]. As a consequence of this
mutation, patients with this syndrome are typically found to develop
distinct simultaneous malignant entities, predominantly vestibular
schwannomas and gliomas [6]. As many as 50% of these patients have
at least one meningioma but are oftentimes afflicted with multiple
tumors [5]. There are other less prominent risk factors which include
hormonal factors, the concomitant presence of breast cancer, obesity
and head trauma [7,8,9].
Meningiomas are classified by the WHO based on morphology
and histopathological characteristics; the latest iteration was rendered
in 2007 [10]. Meningiomas are delineated into 3 broad groups: Grade
I (benign), Grade II (atypical), and Grade III (malignant). The vast
majority (90%) of meningiomas are Grade I tumors [11].
There are distinct clinical characteristics and behaviors that
distinguish Grade II and III meningiomas from Grade I meningiomas;
these are: increased proclivity for invasion into local brain
parenchyma, an increased likelihood of recurrence following initial
treatment, and a shorter overall survival [12]. All three characteristics
are seen to the greatest degree in grade III meningiomas and to a
lesser extent in grade II meningiomas [13,14]. Grade I meningiomas,
on the other hand, infrequently recur, do not exhibit invasion into
surrounding brain structures, and have a considerably longer overall
survival and better prognosis [15]. The risk of recurrence after initial
treatment rises successively with increasing tumor grade and atypical
histopathological features which include high cellularity, tumor
necrosis, high mitotic rate and vascular invasion [16].
The present case defies these general conventions insofar as the
patient had an indolent recurrence of his original grade I tumor after
complete surgical resection.
Another telling difference that distinguishes Grade I meningioma
from Grade II/III meningioma is the likelihood of extracranial metastasis. Overall, extracranial metastasisis rare and is estimated
to occur in less than 0.1% - 0.2% of all patients diagnosed with
meningioma [16]. However, for the patients with grade II and grade
III tumors, extracranial metastasis can occur in as many as 5% and
30% of patients, respectively [10].
It is estimated that roughly 60% of extracranial metastases go to
the lungs, 34% go to the abdomen and liver, and 18% go to the cervical
lymph nodes and surrounding glandular tissue [16]. It is thought that
in these situations, tumor cells travel through venous channels from
which they enter the right-sided circulation and end in one of the
three sites specified. Less commonly, metastatic meningioma tumor
cells may access the vertebral venous system by which they may
travel to other distinct sites in the body [17]. In order of decreasing
incidence, these sites of metastasis are: the long bones, pelvis and
skull (11%); pleura (9%); vertebrae (7%); and mediastinum (5%) [16].
That metastasis is so exceedingly rare for grade I meningiomas in
and of itself makes this case being discussed remarkable. Moreover,
as outlined above, it is rare to find metastases of meningiomas to the
bone as in this case.
As discussed, the goal for the initial treatment of meningioma
is complete surgical resection with or without adjuvant radiation
therapy [18]. Surgical resection is frequently curative for grade I
meningioma [6]. Adjuvant radiation therapy is more commonly
used to treat grade II and always grade III meningioma; this is
because of invasion into surrounding tissue that makes complete
surgical resection implausible and the higher likelihood of recurrence
[19,20,21]. For grade II tumors that are incompletely resected,
radiation therapy is also standard [22]. It is less clear when it comes
to considering adjuvant radiation therapy for grade II meningiomas
for which complete surgical resection is achieved. Radiation therapy
is more likely to be incorporated in the treatment plan if the tumor
possesses features that portend for recurrence or progression and if
the predicted morbidity from therapy is low [23,24].
At this time, there is no demonstrated benefit to the use of systemic
chemotherapy after surgery and radiation therapy for treatment
refractory tumor burden, recurrent meningiomas or malignant
meningiomas. There have been a number of chemotherapeutic agents
tried for treatment of recurrent or malignant tumors; amongst these
are hydroxyurea, temozolomide, cyclophosphamide, doxorubicin
and vincristine [25-28]. Results have been disappointing as there
is no conclusive evidence that any of these agents have significant
potential in altering the biology of the tumor(s) and improving
clinical outcomes.
Other medical strategies that have attempted to exploit specific
aspects of the biology of recurrent or metastatic grade II and grade
III tumors do not as yet appear to have demonstrated benefit. These
include use of estrogen/progesterone receptor inhibitors, EGFR
tyrosine kinase receptor inhibitors, the angiogenesis inhibitor
bevacuzimab, interferon alfa-2b, and somatostatin analogs [25,29,30-
32].
Immunotherapy is a novel approach that appears to have promise
for systemic treatment of recurrent and metastatic meningiomas. It
attempts to change the mechanisms by which high grade meningiomas
inhibit the immune response in the immediate area of tumor growth.
Studies have demonstrated that PD-L1expression is increased with in
atypical meningioma cells and may correlate with its more aggressive
behavior and poorer prognosis.
Further advances in the understanding and unraveling of the
complex and sophisticated biologic profile of meningioma raise
hope that soon this will translate into more promising therapeutic
applications.
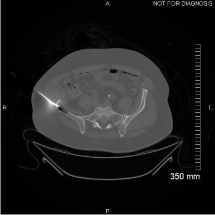
Figure 3
Figure 3
Axial CT image of the pelvis (bone windows) during biopsy
demonstrates a biopsy needle within the lytic lesion located in the right iliac crest.
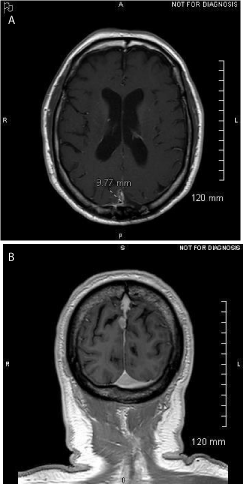
Figure 4
Figure 4
A) Axial T1 W post- contrast MRI image demonstrates post surgical
changes of the posterior parietal calvarium. Calipersareplacedona 9.77
mm avidly enhancing nodule along the posterior falx cerebri suspicious for
recurrent meningioma in the post-surgical bed. B) Coronal T1W post-contrast
MRI image demonstrates the same lesion asin (A.) and better demonstrates
its relationship to the superior sagittal sinus, which is situated just above and
slightly more posterior to the mass.
References
- Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C, et al. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2008-2012. Neuro Oncol. 2015; 17: iv1-iv62.
- Claus EB, Bondy, Schild kraut JM, Wiemels JL, Wrensch M, Black PM. Epidemiology of intracranial meningioma. Neurosurgery. 2005; 57: 1088-1095.
- Umansky F, Shoshan Y, Rosenthal G, Fraifeld S, Spektor S. Radiation-induced meningioma. Neurosurg Focus. 2008; 24: E7.
- Braganza MZ, Kitahara CM, Berringtonde Gonzalez A, Inskip PD, Johnson KJ, Rajaraman P. Ionizing radiation and the risk of brain and central nervous system tumors: asystematic review. Neuro Oncol. 2012; 14: 1316-1324.
- Goutagny S, Kalamarides M. Meningiomas and neuro fibromatosis. J Neurooncol. 2010; 99: 341-347.
- Longo DL, Fauci AS, Kasper DL, Hauser SL, Jameson J, Loscalzo J, eds. Harrison's Principles of Internal Medicine, 18e. NewYork, NY: McGraw-Hill; 2012: 602.
- Wiemels J, Wresnch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010; 99: 307-314.
- Niedermaier T, Behrens G, Schmid D, Schlecht I, Fischer B, Leitzmann MF. Body mass index, physical activity and risk of adult meningioma and glioma: Ameta-analysis. Neurology. 2015; 85: 1342-1350.
- Preston-Martin S, Pogoda JM, Schlehofer B, Blettner M, Howe GR, Ryan P, et al. An international case-control study of adult glioma and meningioma: the role of head trauma. Int J Epidemiol. 1998; 27: 579-586.
- Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, eds. WHO Classification of Tumors of the Central Nervous System. IARC Press, Lyon. 2007. 164.
- Halani SH, Appin C, Brat J, Hadjipanayis CG. Distant Multifocal Bony Metastasis of a Grade II Meningioma: Case Report. J Spine Neurosurg. 2016; 5: 3.
- Yang SY, Park CK, Park SH, Kim DG, Chung YS, Jung HW. Atypical and anaplastic meningiomas: prognostic implications of clinicopathological features. J Neurol Neurosurg Psychiatry. 2008; 79: 574-580.
- Slavin ML. Metastatic malignant meningioma. J Clin Neuro opthalmol. 1989; 9: 55-59.
- Ng TH, Wong MP, Chan KW. Benign metastasizing meningioma. Clin Neurol Neurosurg. 1990; 92: 152-154.
- Marciscano AE, Stemmer-Rachamimov AO, Niemienko A, Larvie M, Curry WT, Barker FG 2nd, et al. Benign meningiomas (WHO Grade I) with atypical histological features: correlation of histopathological features with clinical outcomes. J Neurosurg. 2016; 124: 106-114.
- Kanthan R, Senger JL. Distant Metastases from Meningiomas--A Mythor Reality? Ann Clin Pathol. 2013; 11: 1001.
- Abboud M, Haddad G, Kattar M, Aburiziq I, Geara FB. Extraneural metastases from cranial meingioma: a case report. Radiat Oncol. 2009; 4: 20.
- Aizer AA, Bi WL, Kandola MS, Lee EQ, Nayak L, Rinne ML, et al. Extent of resection and overall survival for patients with a typical and malignant meningioma. Cancer. 2015; 121: 43764381.
- Rogers L, Gilbert M, Vogelbaum MA. Intracranial meningiomas of atypical (WHO grade II) histology. J Neurooncol. 2010; 99: 393-405.
- Hanft S, Canoll P, Bruce JN. A review of malignant meningiomas: diagnosis, characteristics, and treatment. J Neurooncol. 2010; 99: 433-443.
- Dziuk TW, Woo S, Butler EB, Thornby J, Grossman R, Dennis WS, et al. Malignant meningioma: an indication for initial aggressive surgery and adjuvant radio therapy. JNeurooncol. 1998; 37: 177-188.
- Modha A, Gutin PH. Diagnosis and treatment of atypical and anaplastic meningiomas: a review. Neurosurgery 2005; 57: 538-550.
- Park HJ, Kang HC, Kim IH, Park SH, Kim DG, Park CK, et al. The role of adjuvant radio therapy in atypical meningioma. J Neurooncol. 2013; 115: 241-247.
- Mair R, Morris K, Scott I, Carroll TA. Radiotherapy for atypical meningiomas. J Neurosurg. 2011; 115: 811.
- Wen PY, Quant E, Drappatz J, Beroukhim R, Norden AD. Medical therapies for meningiomas. J Neurooncol. 2010; 99: 365-378.
- Swinnen LJ, Rankin C, Rushing EJ, Laura HF, Damek DM, Barge GR. Southwest Oncology Group S9811: a phase II study of hydroxyurea for unresectable meningioma. J Clin Oncol. 2009; 27: 15s.
- Chamberlain MC, Tsao-Wei DD, Groshen S. Temozolomide for treatment-resistant recurrent meningioma. Neurology. 2004; 62: 1210-1212.
- Chamberlain MC. Adjuvant combined modality treatment for malignant meningiomas. J Neurosurg. 1996; 84:733-736.
- Grunberg SM, Weiss MH, Russell CA, Spitz IM, Ahmadi J, Sadun A, et al. Long-term administration of mifepristone (RU486): clinical tolerance during extended treatment of meningioma. Cancer Invest. 2006; 24: 727-733.
- Goodwin JW, Crowley J, Eyre HJ, Stafford B, Jaeckle KA, Townsend JJ. A phase II evaluation of tamoxifen in unresectable or refractory meningiomas: a Southwest Oncology Group study. J Neurooncol. 1993; 15: 75-77.
- Lou E, Sumrall AL, Turner S, Peters KB, Desjardins A, Vredenburgh JJ, et al. Bevacizumab therapy for adults with recurrent/progressive meningioma: a retrospective series. J Neurooncol. 2012; 109: 63-70.
- Kaba SE, DeMonte F, Bruner JM, Kyritsis AP, Jaeckle KA, Levin V, et al. The treatment of recurrent unresectable and malignant meningiomas with interferonalpha-2B. Neurosurgery. 1997; 40: 271-275.