Case Report
Intracranial Dural Marginal Zone Lymphoma: A Case Report
Ahmed Gilani1*, Khurram Shafique1, Jordan Iordanou2, Ali Sadr3, Charles Shao3 and Jinli Liu3
1Department of Pathology, Kings County Hospital Center, USA
2SUNY Downstate School of Medicine, USA
3Department of Neurosurgery, Kings County Hospital, USA
*Corresponding author: Ahmed Gilani, Department of Pathology, Kings County Hospital Center, Brooklyn, NY, USA
Published: 28 Nov, 2016
Cite this article as: Gilani A, Shafique K, Iordanou J, Sadr
A, Shao C, Liu J. Intracranial Dural
Marginal Zone Lymphoma: A Case
Report. Clin Oncol. 2016; 1: 1145.
Abstract
We present a case of low grade Marginal Zone Lymphoma (MZL) presenting as a dural mass in a 21 year old woman. The patient presented with symptoms of raised intracranial pressure. Imaging findings were suggestive of meningioma. Histological analysis showed proliferation of CD20 positive small lymphoid cells forming nodules that were reminiscent of lymphoid follicles with attenuated germinal centers. In between the follicles, cells with plasmacytic differentiation were seen, which were CD138 positive and showed kappa light chain restriction by immunohistochemistry. Ki 67 proliferation index was 20-30%. The patient’s symptoms were alleviated after the surgery and the patient remains disease free more than two yearsafter resection. This case illustrates that although rare, Extra Nodal MZL should be considered in the differential diagnosis of a dura-based mass that fails to show typical meningioma morphology.
Introduction
Intracranial dural Marginal Zone Lymphoma (DMZL) represents a rare group of low-grade B-cell neoplasms with less than 100 cases reported to date. The patient typically present with a few month history of gradually worseningnausea, vomiting, headache and other symptoms of raised intracranial pressure. Radiology reveals an extra-axial contrast enhancing mass without brain involvement. The clinical presentation and radiological appearance is identical to that of the much more common meningioma [1] or sub-dural hematoma [2,3].
Case Presentation
A 21-year-old female presented with several weeks history of dull intermittent headache that was
intractable and excruciating and accompanied by nausea and vomiting at presentation. Past medical
history was unremarkable. Neurological examination revealed hyper-reflexia and downward drifting
of the right upper extremity. Initial head non-contrast Computed Tomography (CT) showed a
lenticular mass along the left frontal convexity with mild edema of the adjacent frontal and parietal
cortices. These findings initially raised suspicion of epidural or subdural hematoma. In the absence
of any history of trauma, vascular malformation was considered; CT angiogram however failed to
show any vascular malformation. Magnetic Resonance Imaging (MRI) was then performed, which
showed an extra-axial dural based lesion with contrast enhancement and a prominent dural tail
(Figure 1a and b). These findings were considered highly suggestive of meningioma.
Complete resection of the mass was planned after discussion with the patient and her
family, Intraoperative biopsy showed a dense aggregate of plasmacytoid cells, raising the
suspicion for a lymphoid neoplasm. Complete resection of the lesion was performed and the
specimen was sent in 10% formalin for pathological examination. Post-operative MRI showed
no evidence of residual neoplasm. The patient recovered uneventfully and was symptom free
with no residual neurologic deficit soon after the procedure. After the histology (see pathology
section below) confirmed the diagnosis of Marginal Zone Lymphoma, extensive clinical and
imaging workup was conducted to search for extra-cranial disease. These studies however failed
to find any signs of lymphoma. The patient is being periodically followed up by hematologyoncology
and neurosurgery teams and remains free of disease two years after resection.
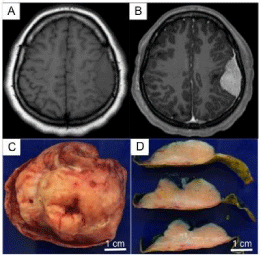
Gross inspection showed a tan white firm lenticular mass encapsulated by dura (Figure 1C and
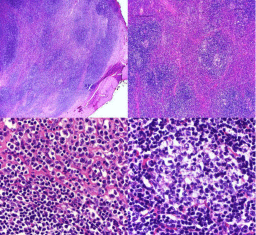
D). Microscopic examination revealed a dense lymphoplasmacyticaggregates separated by fibrous
sepatae, giving a vague nodular arrangement (Figure 2A). Cytologically, three types of cells were present, 1) cells with small centrally placed nuclei and a clear or place cytoplasm resembling centrocytes (Figure 2C), 2) Cells
with plasmacytic differentiation (Figure 2C), 3) cells of germinal
center (Figure 2D). Based on the location and histopathologic
findings the differential diagnosis included: a low grade lymphoma
such as MALT or Mantle cell lymphoma, plasmacytoma, IgG4
related sclerosing pachymeningitis, and lymphoplasmacyte-rich
meningioma. Immunohistochemical (IHC) analysis was performed
to rule out these possibilities. The results are summarized in Table
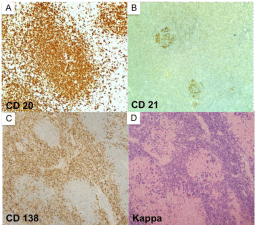
1. In summary, CD20 stain outlined the follicles with attenuated
germinal centers, which were positive for CD21 (Figure 3). Between
the follicles, the plasmatic cells were positive for CD138. Kappa
light chain restriction was demonstrated by both IHC and in situ
hybridization (ISH) (Figure 3). Heavy chain studies showed that the
plasma cells were weakly positive for IgG, however negative for IgG4.
Ki 67 proliferation index was estimated to be 20-30%, consistent with the indolent feature of this entity. The specimen was received prefixed
in formalin; hence flow cytometry was not attempted. These findings
are diagnostic for a low gradeB cell non-Hodgkin’s lymphoma with
plasmacytoid differentiation, most consistent with marginal zone
lymphoma.
Figure 1
Figure 1
Imaging and Gross Examination Findings: (A, B) Axial MRI images,
the lesion was isointense on T1 (A) and showed contrast enhancement (B).
(C&D) Gross examination showed that the mass was completely enclosed
in dura.
Figure 2
Figure 2
(A,B): Diffuse nodular pattern was seen, with attenuated germinal
centers (B). (C) Higher magnification view of the periphery of a nodule,
showing centrocyte like cells (lower left) and plasmacytoid cells (upper right).
(D) Higher magnification view of the germinal center like region.
Table 1
Figure 3
Figure 3
(a-d, clockwise from top left) Immunohistochemical staining
showing CD20 staining of lymphoid follicles (A), CD21 staining of germinal
centers (B), Cd138 (C) and kappa light chain (D) staining of plasmacytoid
cells in the intermodal region.
Discussion
Primary CNS lymphoma accounts for approximately 3-4% of
primary brain tumors, the majority of these (90%) being large B-cell
diffuse lymphomas. These neoplasms usually present as aggressive
intra-parenchymal tumors, which may secondarily involve the
leptomeninges [4]. Lymphomas primary to the dura mater are much
less frequent. Marginal zone lymphoma, designated as extranodal
marginal zone lymphoma’ in the World Health Organization
Lymphoma classification, is exceedingly rare [2-8]. This neoplasm
was initially discovered in mucosa associated lymphoid tissue (MALT
lymphoma) of the stomach, but it can also occur in sites without
mucosa such as the orbit, skin, thyroid, thymus, breast, and liver [9].
DMZL most commonly presents in middle aged women with
female to male ratio of 4:1 and an average age of presentation of
55 years (range 29 to 70 years) [5,6,10]. This case illustrates that the diagnosis should also be considered in younger patients. The
diagnosis of primary dural MZL requires exclusion of systemic
involvement with lymphoma [9]. Diagnostic workup may include:
a variety of imaging modalities such as: chest and abdomino-pelvic
MRI or CT, Positron Emission Tomography, bone marrow biopsy,
ophthalmologic examination (including slit lamp), HIV serology, and
lumbar puncture [11].
DMZL typically mimics meningioma in clinical and radiological
findings [5]. Typical radiology findings include a single or multiple
masses present along the cerebral convexities, falx, tentorium, and
sellar/parasellar regions. Rarely, intraventricular and spine masses can
occur. The masses are typically lenticular with thickened meninges
and frequently show a dural tail. Contrast enhancement with a dural
tail has been reported in most cases [11,12]. These features make
a radiological distinction from meningioma extremely difficult.
However, recently it has been suggested that DMZL shows restricted
diffusion behavior on diffusion-weighted images which may help in
differentiating these lesions from the typical meningiomas [4].
Although there is no consensus on the treatment of DMZL,
surgical resection, chemotherapy and radiation have been tried in
combination or stand-alone therapies. Some authors have suggested
that radiation or chemotherapy should be reserved for cases in which
complete resection is not possible [5,6,11]. Unlike DLBCL, Marginal
zone lymphoma has a good prognosis. Most patients attain complete
response and have good local disease control. In a series of 8 cases,
Iwamoto et al. [11] reported complete response in all cases, however
3 patient developed systemic relapse.
Other rare entities that should be considered as differentials
for a dural based mass includes: solitary fibrous tumor/
hemangiopericytoma, metastatic tumor, lympho-plasmacyterich
meningioma, IgG4 related sclerosingpachymeningitis, and
Rosai-Dorfman disease. These entities differ from DMZL in
histologic and immunohistochemical features. Lymphoplasmacyterich
meningioma features dense infiltrates of lymphocytes and
plasma cells mixed with regions of EMA positive meningothelial
cells. Idiopathic hypertrophic pachymeningitis typically shows a
polymorphic infiltrate of lymphocytes, plasma cells and ultinucleated
giant cells. IgG4 related sclerosing disease can be picked up by IgG4
immunohistochemistry. Langerhans cell histiocytosis shows CD1a
and Langerin positivity and Rosai-Dorfman disease is usually S100
and CD68 positive and showsemperiopoies is [13]. These features
were not seen in the current case, favoring the diagnosis of extranodal
marginal zone lymphoma, arising from dura (DMZL).
References
- KM Kulkarni, Sternau L, Dubovy SR, Lam BL. Primary dural lymphoma masquerading as a meningioma. J Neuroophthalm. 2012; 32: 240-242.
- Gocmen S, Gamsizkan M, Onguru O, Sefali M, Erdogan E. Primary dural lymphoma mimicking a subdural hematoma. J Clinic Neurosci. 2010; 17: 380-382.
- Goetz P, Lafuente J, Revesz T, Galloway M, Dogan A, Kitchen N. Primary low-grade B-cell lymphoma of mucosa-associated lymphoid tissue of the dura mimicking the presentation of an acute subdural hematoma. Case report and review of the literature. J Neurosurg. 2002; 96: 611-614.
- Sebastian C, Vela AC, Figueroa R, Marin MA, Alfaro J. Primary intracranial mucosa-associated lymphoid tissue lymphoma. A report of two cases and literature review. Neuroradiol J. 2014; 27: 425-430.
- George AC, Ozsahin M, Janzer R, Agassis S, Meuli R, Baur AS, et al. Primary intracranial dural lymphoma of mucosa-associated lymphoid tissue (MALT) type: report of one case and review of the literature. Bulletin du cancer. 2005; 92: E51-E56.
- Kumar S, Kumar D, Kaldjian EP, Bauserman S, Raffeld M, Jaffe ES, et al. Primary low-grade B-cell lymphoma of the dura: a mucosa associated lymphoid tissue-type lymphoma. Am J Surg Pathol. 1997; 21: 81-87.
- Razaq W, Goel A, Amin A, Grossbard ML. Primary central nervous system mucosa-associated lymphoid tissue lymphoma: case report and literature review. Clin Lymphoma Myeloma. 2009; 9: E5-9.
- Rottnek M, Strauchen J, Moore F, Morgello S. Primary dural mucosaassociated lymphoid tissue-type lymphoma: case report and review of the literature. J Neurooncol. 2004; 68: 19-23.
- Pavlou G, Pal D, Bucur S, Chakrabarty A, van Hille PT. Intracranial non- Hodgkin's MALT lymphoma mimicking a large convexity meningioma, Acta Neurochir (Wien). 2006; 148: 791-793.
- Venkataraman G, Rizzo KA, Chavez JJ, Streubel B, Raffeld M, Jaffe ES, et al. Marginal zone lymphomas involving meningeal dura: possible link to IgG4-related diseases. Mod Pathol. 2011; 24: 355-366.
- Iwamoto FM, Abrey LE. Primary dural lymphomas: a review. Neurosurg Focus. 2006; 21: E5.
- Reis F, Schwingel R, Queiroz Lde S, Zanardi Vde A. Primary dural lymphoma: a rare subtype of primary central nervous system lymphoma (PCNSL). Arq Neuropsiquiatr. 2011; 69: 264-265.
- P. Mahzoni, MH Zavareh, M Bagheri, N Hani, B Moqtader. Intracranial ROSAI-DORFMAN Disease. J Res Med Sci. 2012; 17: 304-307.