Case Report
Targeting RAGE Expression in Human Ovarian Cancer
Tekabe Y1*, Li Q1, Rodriguez K1, Johnson J1, Khaw BA2, Kokoshka J3, Ray R4, Rai V4 and Johnson L1
1Department of Medicine, Columbia University Medical Center, New York, USA
2Department of Pharmaceutical Sciences, Northeastern University, USA
3Department of Technology Ventures, Columbia University, New York, USA
4Institute of Life Sciences, Department of Biotechnology, Bhubaneswar, India
*Corresponding author: Yared Tekabe, Department of Medicine, Columbia University Medical Center, New York, USA
Published: 30 Jul, 2016
Cite this article as: Tekabe Y, Li Q, Rodriguez K, Johnson
J, Khaw BA, Kokoshka J, et al.
Targeting RAGE Expression in Human
Ovarian Cancer. Clin Oncol. 2016; 1:
1055.
Abstract
Receptor for Advanced Glycation Endproducts (RAGE) is expressed in ovarian tissue and associated
with ovarian carcinoma. With a radiolabeled anti-RAGE antibody, we proposed to show extent of
RAGE expression in animal model and effect of blocking RAGE on ovarian cancer cell growth.
Methods: We measured inhibition of p-Akt and p-stat3 in SKOV-3 human ovarian carcinoma cell
line and measured cell growth suppression in culture. For imaging, female nude mice (n = 15) at 6
wks of age were injected with luciferase expressing human ovarian cancer cells into the right flank.
Four-five weeks later, animals were injected with luciferin and imaged on optical imager followed
by injection with 111In-anti-RAGE F(ab’)2 (n = 7) or 111In-control IgG F(ab’)2 and 48 h later, were
imaged on micro-SPECT/CT. Focal tracer uptake on scans was quantified, the tumors removed,
radioactivity counted, and sectioned for histological and immunohistochemical examination.
Results: RAGE antibody pretreatment inhibited p-stat3 and p-AKT expression in SKOV-3 cells
and there was a dose related reduction in cell growth in culture. There was good co-localization of
the luciferase producing tumor on the optical scan and tumor location at necropsy with the focal
uptake of the 111In-anti-RAGE F(ab’)2. Quantitative tracer uptake in the tumor from scans showed
that uptake of 111In-anti-RAGE F(ab’)2 as %ID was 2.4 fold higher than 111In-control F(ab’)2 (P =
0.01) confirmed by gamma well counting. Dual immunofluorescent staining for RAGE and PAX8
in tumors showed high expression of RAGE and co-localization with PAX8 positive stained cells.
Conclusion: RAGE expression in ovarian tumors in live animals can be imaged and quantified. An
anti-RAGE F(ab’)2 used for imaging shows blocking properties and suppresses ovarian cancer cell
growth.
Keywords: Ovarian cancer; RAGE; SPECT/CT Imaging
Introduction
Ovarian cancer is the fourth leading cause of cancer deaths in women [1]. Serum biomarker CA-125 is used to monitor the effects of ovarian cancer treatment, but is not accurate for detecting early disease [2]. Single-photon emission computed tomography/computed tomography (SPECT/ CT) imaging with 111In-satumomab pendetide (OncoScint) has limited diagnostic accuracy [3]. In biopsy and tissue samples paired-box gene 8 (PAX8) that encodes transcription factors associated with organogenesis, morphogenesis, thyroid, renal and Millerian cell differentiation and is highly expressed in ovarian cancer cells is used as a tissue marker [4]. Expression of Receptor for Advanced Glycated Endproducts (RAGE) in the tissue of reproductive human ovaries has been identified in multiple areas of ovarian dysfunction that make it a key component in identifying abnormalities similar to ovarian carcinoma [5] and is associated with the development of ovarian carcinoma [6]. RAGE is a multifunctional receptor binding a number of ligands known to play important roles in diabetes, atherosclerosis, cancer, and Alzheimer’s disease [7-9]. The V domain on the extracellular RAGE receptor binds endogenous molecules referred to as damage associated molecular pattern molecules (DAMPs) that serve as ligands for RAGE, playing prominent roles in inflammation and cancer [10-13]. These ligands include proinflammatory cytokines S100 proteins, high-mobility group box 1 (HMGB1) proteins, and lysophosphatidic acid (LPA) [10-13]. The RAGE/ligand binding activates signaling pathways that enhance cell proliferation, activate pro-survival proteins, enhance autophagy, limit apoptosis, promote angiogenesis, and promote invasion and metastasis [14-21]. Cytoplasmic S100A4 expression is significantly stronger in ovarian cancers compared to benign ovarian tumors, as well as being a strongly positive nuclear staining associated with poor prognosis [22]. The administration of s-RAGE, a soluble extracellular ligand-binding domain that acts as a decoy for RAGE ligands, or administration of an anti-RAGE antibody, blocked growth of C6 glioma cells engrafted into nude mice [23]. These findings indicate that blocking RAGE expression in ovarian cancer may suppress tumor growth. We developed a murine monoclonal anti-RAGE F(ab’)2 antibody fragment that binds a unique peptide sequence on the C-terminal of the V extracellular domain of RAGE. Using a 99mTc-labeled anti-RAGE (Fab’)2, we have shown uptake in atherosclerosis and peripheral artery disease in diabetes [24,25]. Furthermore, we have shown blocking properties of the antibody to suppress RAGE expression and thereby improve hind limb angiogenesis after femoral artery ligation in diabetic mice [26]. The present study extends our investigation into imaging and blocking antibody techniques on human SKOV-3 ovarian cancer cell line. We tested the effect of the antibody to change cell survival in culture and performed in-vivo imaging experiments using 111Inlabeled anti-RAGE F(ab’)2 and a small animal hybrid SPECT/CT imaging.
Materials and Methods
All animal studies were performed with the approval of the
Institutional Animal Care and Use Committee of Columbia
University.
Antibody
The antibody was developed against a unique peptide sequence
on the V-domain of RAGE designed to display immunoreactivity in
mice, pigs, and humans. Based on Genbank sequences, a peptide was
prepared and used to immunize mice and subsequently hybridomas
produced. Monoclonal antibodies (mouse IgG2a kappa) were
produced in-vitro and purified by protein A and low endotoxin
units (less than 3 endotoxin units/mg of purified antibody) (Strategic
Diagnostics or SDIX, Newark DE). Monoclonal anti-RAGE F(ab’)2
fragments were developed as previously described [24].
Western blot analysis
SKOV-3 (Cell Biolabs, San Diego, CA) cells were maintained in
DMEM medium supplemented with 10% fetal bovine serum (FBS)
and 1% penicillin/streptomycin (Gibco-Life Technologies, Grand
Island, NY) in a 5% CO2 incubator at 37°C. For experiments, cells
were plated at a density of 1x 105 cells/ml in 60 mm dish. Cells were
serum starved for overnight after they reached 70-80% confluency
and next day cells were pre-treated with 50 μg/ml IgG or anti-RAGE
Fab for one hour followed by treatment with 10 μg/ml of AGEs
for 20 minutes. Tumor cells were lysed and total cell lysates were
immunoblotted and probed with RAGE, p-AKT, AKT, p-stat3 and
stat3 antibodies (Cell Signaling (Danvers, MA). Briefly, protein (35
μg) from cell lysates were denatured, resolved by 10% acrylamide
gels and transferred to a nitrocellulose membrane. Membranes were
blocked in 5% non-fat dry milk and then incubated with primary
antibodies: p-AKT, p-stat3, RAGE. HRP-conjugated anti-rabbit IgG
and anti-mouse (Amersham, Piscataway, NJ) was used to identify
sites of primary antibody binding. Membranes were stripped of
bound primary antibodies and re-probed with total AKT, total stat-
3 and β-actin antibodies. The ratios (phospho/total) were quantified
and normalized using ImageJ software (NIH, Bethesda, MD, USA).
Cell viability assay
SKOV-3s (paclitaxel-sensitive) and SKOV-3TR (paclitaxelresistant)
cell lines were used in cell culture experiments. Briefly, cells
were plated (4000 cells/well) in to 96 well plates and maintained at
37ºC for 24 hours (h) in a humidified incubator with 5% CO2. Media
was removed and serial dilutions of anti-RAGE antibody or control
antibody was added to the plates: 50, 25, 12.5, 6.25, 3.125, 1.562,
0.781 μg/ml. Plates were incubated for 24 h at 37°C and 5% CO2 then
washed with respective media. CellTiter Blue (Promega, Madison,
WI) (50μl of 1:5 dilutions) was added to plates and incubated at 37°C
and 5% CO2 for around 2 h. Fluorescence was recorded (excitation,
560 nm: emission, 590 nm) using BioTek Fluorescence Plate Reader.
Animals
Female nude (J:NU) mice (6-7 wks of age) were purchased from
The Jackson Laboratories (Indianapolis, IN). Mice were injected
subcutaneously with luciferase producing SKOV-3 cells (3 x 106
cells in 0.1 ml PBS) (n = 13) into the right flank, Tumor sizes on the
flanks were measured weekly with calipers and calculated as: (length
x height x width x π)/6 [27,28]. Imaging experiments were performed
when tumor reached 1070±279 mm3, occurring 4-5 w ks after tumor
injection.
Radiotracer preparation
For 111In labeling, approximately 1 mg of the F(ab’)2 in 0.1
M NaHCO3 buffer (pH 8.2) was conjugated with a 5 molar excess
of [(R)-2-Amino-3-(4-isothiocyanatophenyl)propyl]-trans-(S,S)-
cyclohexane-1,2-diamine-pentaacetic acid (p-SCN-Bn-CHX-A’’-
DTPA; Macrocyclics). The reaction mixture was incubated at room
temperature for 1 h followed by overnight dialysis at 4ºC in 0.25 M
NH4Ac (pH 5.5). To 10 μl (74 - 111 MBq) (2-3 mCi) of 111In in 0.05
N HCl, 5 volumes of 0.1 M NH4Ac (pH 5.5) was mixed and after 10
min, 50 μl (50 μg) of DTPA conjugated anti-RAGE F(ab’)2 was added.
The reaction mixture was incubated at room temperature for 45 min.
111In-labeled antibody fragments were separated from free 111In using
PD 10 column pre-equilibrated with 0.1 M NH4Ac (pH 6). The mean
specific activity was 7.65 ± 0.75 MBq/μg of protein, and the mean
radiopurity was 97.7 ± 0.95% by instant thin-layer chromatography.
Biodistribution and blood pool clearance
Five tumor implanted nude mice were used to measure
biodistribution of 111In-anti-RAGE (Fab’)2. Each animal was
euthanized by an intraperitoneal injection of pentobarbital (100
mg/kg). Organs were dissected, weighed, and counted in a gamma
counter (Wallac Wizard 1470, PerkinElmer, Waltham, MA, USA) for
determination of the percent injected dose of radiotracer per gram
(%ID/g) tissue. Radiotracer activity in the samples was corrected for
background, decay time, and tissue weight. To determine optimal
imaging time, 3 mice were euthanized at 24 and 48 h after tracer
injection and tissue samples of the tumor and blood counted. At 24
h, the ratio of tumor/blood = 0.18 and at 48 h the ratio was 2.7 and
therefore 48 h time point was selected for imaging.
Imaging
Mice underwent micro-SPECT/CT (Bioscan/Mediso) imaging 48
h after tracer injection. Prior to SPECT imaging the mice underwent
bioluminescence imaging to detect the signal from the luciferase
producing tumor cells. Mice were anesthetized and injected IP with
luciferin (150 μg in 100 μl/mouse). Twenty min later, mice were
imaged using a PhotonImager optical imaging device (Biospace lab,
Paris, France). Light emission was detected by an ICCD camera and
quantified using image processing M3 Vision software (Biospace
lab). Following optical imaging, 4.81 ± 1.11 MBq (0.13 ± 0.03 mCi)
111In-anti-RAGE F(ab’)2 was injected via the femoral vein catheter.
Forty-eight h later, mice were imaged on a nano-SPECT/CT scanner
(Bioscan, Washington DC). A topogram (sequence of 2D side view
x-ray projections) was used to determine the axial scan range for
SPECT and CT imaging. CT images were acquired with an integrated
CT scanner using an x-ray tube at 45 kVp and an exposure time of
1000 ms per view. Following CT acquisition, helical SPECT scans
were acquired using dual-headed detectors each outfitted with
collimators with nine pinholes. Each pinhole had a diameter of 1.4
mm providing a transaxial field-of-view (FOV) of 30 mm and an
axial FOV of 16 mm, extendable through helical scanning to 270 mm.
SPECT data were acquired with the following parameters: step and
shoot rotation, 30° step in 360° rotation using 24 projections, 60 s
per projection, 256 x 256 frame size with 1.0 mm pixels, and 140 keV
with 10% energy window. The projection data were reconstructed by
OSEM algorithm with subset and iteration number set to 16 and 8,
respectively, and a voxel size of 300 μm and SPECT and CT datasets
fused. At the completion of in-vivo imaging, the animals were
sacrificed and tumors were harvested, radioactivity counted, and
fixed in 10% formalin.
Image analyses
Regions of interest (ROIs) with equal voxel thickness were drawn
on transaxial slices that comprised the tumor volume identified from
the CT scan. Uptake of 111In-anti-RAGE F(ab’)2 in the tumor was
quantified as MBq (mCi) using a calibration factor determined from
mouse-shaped phantoms filled with known levels of 111In imaged
with the same protocol and entered into the display software (In vivo
Scope software). The tumor uptake from the scans was divided by the
total injected dose to obtain percent injected dose (%ID).
Gamma well counting
Tumors were weighed and the radiotracer uptake was determined
in a gamma well counter (Wallac Wizard 1470, PerkinElmer) and
expressed as the percentage injected dose per gram (%ID/g) of tissue.
The radiotracer activity in the samples was corrected for background,
decay time, and tissue weight.
Histopathology
The explanted tumors were fixed in 10% formalin for 24 h followed
by 70% alcohol awaiting decay followed by paraffin embedding.
Serial sections (5-μm-thick) from paraffin-embedded tumors were
processed for hematoxylin and eosin (H&E) for morphological
evaluation and immunohistochemical characterization. Serial
sections were deparaffinized and rehydrated followed by quenching
of endogenous peroxidase activity with 0.3% hydrogen peroxide.
Slides were then incubated overnight with primary antibody for
RAGE (50 μg/ml; mouse anti-RAGE F(ab’)2 and anti-Paired-box gene
8 (PAX;1:100). Slides were incubated for 30 min with biotinylated
respective secondary antibodies. Sections were treated for 30 min
with VECTASTAIN ABC reagent (Vector Laboratories). Color
reaction was visualized with 3’,3’-diaminobenzidine (DAB substrate
kit, Dako) and counterstained with Gill’s hematoxylin solution.
Images were captured using a digital camera mounted on a Nikon
microscope and analyzed using Image-Pro Plus software (Media
Cybernetics Inc., Bethesda, MD).
Immunofluorescence
Dual fluorescent confocal microscopy studies were performed to
co-localize RAGE with PAX8 positive cells and macrophages. Briefly,
tumor sections were incubated with anti-RAGE F(ab’)2 (Texas red;
1:100) and co-stained with anti-PAX antibody (FITC; 1:50) or antimacrophage
(1:50, Santa Cruz). The images were examined using a
fluorescence microscope (Nikon).
Statistical analyses
Comparisons between groups were made using the Student
t test. All statistical tests were two-tailed, with P < 0.05 denoting
significance. The cell viability assay data were analyzed with two-way
ANOVA and the Bonferroni multiple comparison tests.
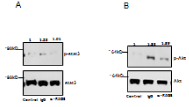
Figure 1
Figure 1
Levels of (A) phosphorylated/total stat3 and (B) phosphorylated
Akt (Ser473)/total Akt in SKOV-3 cells shown by western blotting upon
stimulation with 10 μg/ml of AGEs for 20 minutes after treatment with 30 μg/
ml of control F(ab’)2 or α-RAGE F(ab’)2.
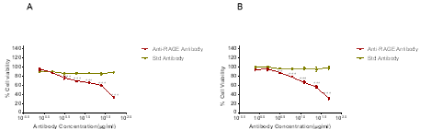
Figure 2
Figure 2
Cell viability assay. Cell viability of SKOV-3 paclitaxel-sensitive
(A) and paclitaxel-resistant SKOV-3 (B) cells after treatment with anti-RAGE
F(ab’)2 or with control F(ab’)2. P < 0.01** and P < 0.001***.
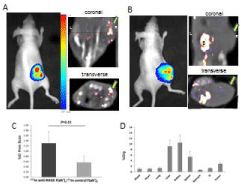
Figure 3
Figure 3
(A) Bioluminescent image on left of nude mouse engrafted in
left flank with luciferase producing SKOV-3 cells and coronal, and on right
transverse SPECT/CT images obtained from same mouse injected with 111Inanti-
RAGE F(ab’)2 showing focal tracer uptake in left flank. (B) Same format
as A for mouse injected with and 111In-control F(ab’)2 showing minimal tracer
uptake in the left flank. (C) Graph showing quantitative tracer uptake as %ID
for 111In labeled anti-RAGE ab and labeled inactive probe. (D) Biodistribution
of 111In-anti-RAGE F(ab’)2 in tumor and non-tumor organs in female nude
mice engrafted with SKOV-3 cells. B=Bladder.
Results
Western blot analysis
Western blot analysis demonstrated a reduction of p-AKT
(Ser473) in SKOV-3 cells pretreated with 10 μg/ml of anti-RAGE
F(ab′)2 compared to cells pretreated with control IgG F(ab′)2. RAGE
antibody fragment pretreatment inhibited p-stat3 and p-AKT
expression by 99.9% and 53%, respectively (Figure 1).
Cell viability assay
The effects of increasing doses of anti-RAGE antibody on
human ovarian cancer cell lines: SKOV-3s (paclitaxel-sensitive) and
SKOV-3TR (paclitaxel-resistant) are shown in (Figure 2A and B),
respectively. At 50 μg/ml dose, the anti-RAGE antibody significantly
lowered the % cell viability of both ovarian cancer cell lines in a doseresponsive
manner as compared to the negative control (P < 0.001).
Tumor grafts in nude mice
Macroscopically, tumor-bearing mice developed palpable masses
at the region of implantation with a mean tumor volume of 1070±279
mm3.
In-vivo imaging
Bioluminescent imaging on an optical imager (BioSpace Lab,
Paris, France) showed signal from luciferase producing SKOV-3 cells
in the flank where the tumors were grafted. The SPECT/CT imaging of
the uptake of 111In-anti-RAGE F(ab’)2 showed uptake of radiolabeled
antibody fragment that co-localized to the optical signal from the
luciferase producing tumor cells (Figure 3A). Non immune control
antibody fragment showed minimal uptake at the target (Figure 3B).
Quantitative tracer uptake in tumors
The quantitative accumulation of 111In-anti-RAGE F(ab’)2 in the
tumor from the scan (1.3 ± 0.45% ID) was significantly higher than the
uptake of 111In-control F(ab’)2 (0.56 ± 0.25, P=0.01) (Figure 3C). The
higher uptake of 111In-anti-RAGE F(ab’)2 in the tumor was confirmed
by ex-vivo gamma counting. There was a significantly greater tumor
uptake of 111In-anti-RAGE F(ab’)2 (2.86 ± 0.57% ID/g) compared with
111In-control F(ab’)2 (0.52± 0.21%; P < 0.001).
Tracer biodistribution
Accumulation of 111In-anti-RAGE F(ab’)2 in non-target organs
is shown in (Figure 3D). The liver and kidneys had the highest
accumulation uptake of tracer.
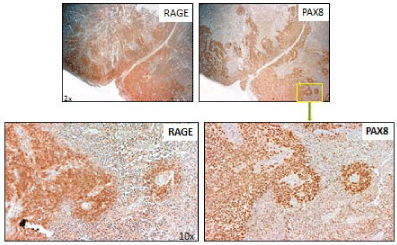
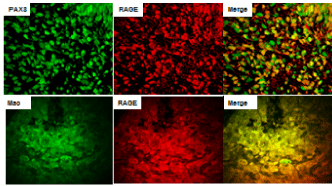
Histopathology
Immunohistochemical staining of the explanted tumor sections
revealed RAGE visually co-localized with sites of PAX8-positive
cells (Figure 4). Dual immunofluorescent staining confirmed colocalization
of RAGE with PAX8 and also with macrophages (Figure
5).
Figure 4
Figure 4
Immunohistological sections at 20x (top) and 100X (lower) from
explanted tumors with secondary HRP (brown chromogen) stain showing
expression of RAGE that corresponds to staining for SKOV-3 antigen PAX8.
Figure 5
Figure 5
Dual fluorescent confocal microscopic images from SKOV-3 tumor
sections. The top row shows dual immunofluorescent staining for RAGE (red)
and PAX8-positive cells (green) show colocalization in the merged image
(yellow). The bottom row shows colocalization of Macrophage staining with
RAGE. Magnification x400.
Discussion
We showed that a radiolabeled anti-RAGE antibody fragment targeting luciferase producing ovarian tumors co-localizes with the luciferase signal on optical imaging and can be visualized on SPECT/ CT scans and correlates with immunohistology. By Western blot analysis, we showed that our antibody has blocking properties based on a marked reduction of p-AKT (Ser473) in SKOV-3 cells pretreated with anti-RAGE antibody F(ab′)2 and that exposing these cancer cells in culture to the antibody, growth is reduced. Immunohistochemistry and dual fluorescence staining reported in this paper show RAGE colocalized with PAX stain for the paired box growth gene expressed in tumor cells and also with Mac stain identifying macrophages. Together, these two co-localizing stains document the role of RAGE in pathways of inflammation and tumor genesis. DAMPs include ligands that are released in the setting of cellular stress, damage, inflammation, or necrotic cell death bind to receptors that activate immune responses or tissue repair. The same process in addition to being beneficial can lead to chronic inflammation and neoplastic transformation or contribute to the inflammation induced by primary neoplasia leading to tumor growth and metastasis [11,12]. Currently, there are few imaging strategies that identify ovarian cancer in its early stage. Satumomab pendetide (OncoScint) is an IgG murine monoclonal antibody that targets the cell surface mucinlike glycoprotein antigen TAG-72 found on colorectal and ovarian cancers. The antibody labeled with 111In has been in clinical use for more than 10 years. While its sensitivity for detecting cancer spread in the abdomen is better than CT, it is not sensitive for detecting early disease [3]. In addition, since it is a murine antibody, about 40% of patients develop human anti-mouse antibody levels after injection. For further imaging and treatment, we have humanized our anti-RAGE antibody which would eliminate reactions to mouse antigens. High liver uptake as shown in our biodistribution data is common in radiolabeled antibodies and is a limitation for nuclear imaging of intra-abdominal malignancies. Potentially more important clinically than development of an ovarian cancer imaging tracer is development of a RAGE suppressing treatment agent in ovarian cancer. RAGE expression is increased and downstream pathways activated by RAGE ligands. Two classes of RAGE ligands or DAMPs - S100 and HMGB1 proteins - have been found in a number of tumors [14-17]. While S100 proteins are localized in the cytoplasm, HMGB1 is localized to the nucleus gaining access to bind nuclear membranes on tumor cells undergoing necrosis [16]. The intracellular signaling pathways, initiated by RAGE/HMGB1 binding in tumors, promote proliferation and invasion of tumor cells via several pathways. In-vitro binding studies have identified a cytoplasmic region of RAGE as an ERK docking site [21]. A recently reported additional ligand binding RAGE and stimulating tumor growth is LPA (lysophosphatidic acid) which regulates fundamental cellular processes such as proliferation, survival, motility and invasion [18]. Reported studies using tissue microarrays have documented RAGE expression in several human tumors. In particular, RAGE expression has been shown in human ovarian cancer cells and the ligands S100A4, S100AI4, HMGB2 overexpressed and secreted by ovarian carcinoma cells [22]. S100 ligands, low molecular weight proteins able to form homodimers and heterodimers and oligomers have a variety of functions in cell growth and members of the family have been shown to change the microenvironment favoring tumor metastasis [15]. Cytoplasmic S100A4 expression is significantly stronger in ovarian cancers compared to benign ovarian tumors and strongly positive nuclear staining is associated with poor prognosis [22]. The RAGE gene is found on chromosome 6p21.3 in the major histocompatibility locus (MHC). Polymorphisms at the RAGE gene locus was identified in 1998 [31]. Population based studies have investigated single nucleotide polymorphisms (SNPs) have recently focused on investigating functional polymorphisms in the RAGE gene that may affect the expression or function of RAGE and are associated with certain malignancies. One polymorphism was found to be a significant factor in several malignancies including ovarian cancer [32,33]. This SNP designated 82G/S codes for a change from glycine to serine within the putative ligand-binding domain of the receptor protein [31,32]. Specific mechanisms to link this change in the ligand-binding domain and carcinogenesis are not yet been worked out but in light of interest in personalized medicine identifying a target such as RAGE with a genetic variant in the RAGE gene linked to carcinogenesis and important role of RAGE in some specific tumors makes RAGE a potential target for identifying at risk patients for higher surveillance possibly involving imaging and for treatment strategies to reduce RAGE expression.
Summary and Future Directions
This current study is the first to report that a radiolabeled antibody against RAGE localizes to ovarian cancer cells injected into nude mice and can be imaged in live animals on SPECT/CT. The important role of RAGE expression and binding of ligands that activate many pathways necessary for tumor growth and metastasis indicate that RAGE expression is a key biological tumor marker. In addition, the antibody we developed has blocking properties that can inhibit tumor cell growth in culture. Further work is needed to demonstrate effect of treatment with antibody to inhibit tumor growth in mice.
References
- Berchuck A, Schildkraut JM, Marks JR, Futreal PA. Managing hereditary ovarian cancer risk. Cancer. 1999; 86: 2517-2524.
- Bast RC, Klug TL, St. John E, Jenison E, Niloff JM, Lazarus H, et al. A radioimmunoassay using a monoclonal antibody to monitor the course of epithelial ovarian cancer. New England Journal of Medicine. 1983; 309: 883-887.
- Pinkas L, Robins PD, Forstrom LA, Mahoney DW, Mullan BP. Clinical experience with monoclonal antbodies in detection of colorectal and ovarian cancer recurrence and review of the literature. Nucl Med Commun. 1999; 20: 689-696.
- Liliac L, Carcangiu ML, Canevari S, Amălinei C. The value of PAX8 and WT1 molecules in ovarian cancer diagnosis. Rom J Morphol Embryol. 2013; 54: 17-27.
- Merhi Z. Advanced glycation end products and their relevance in female reproduction. Hum Reprod. 2014; 29: 135-145.
- Diamanti-Kandarakis E, Piperi C, Patsouris E, Korkolopoulou P, Panidis D, Pawelczyk L, et al. Immunohistochemical localization of advanced glycation end-products (AGEs) and their receptor (RAGE) in polycystic and normal ovaries. Histochem Cell Biol. 2007; 127: 581-589.
- Bucciarelli LG, Wendt T, Rong L, Lalla E, Hofmann MA, Goova MT, et al. RAGE is a multiligand receptor of the immunoglobulin superfamily: implications for homeostasis and chronic disease. Cell Mol Life Sci. 2002; 59: 117-1128.
- Yan SF, D'Agati V, Schmidt AM, Ramasamy R. Receptor for Advanced Glycation Endproducts (RAGE): a formidable force in the pathogenesis of cardiovascular complications of diabetes and aging. Curr Mol Med. 2007; 7: 699-710.
- Yan SF, Ramasamy R, Schmidt AM. Mechanisms of disease: advanced glycation endproducts and their receptor in inflammation and diabetes complications. Nat Clin Pract Endocrinol Metab. 2008; 4: 285-293.
- Srikrishna G, Freeze HH. Endogenous damage-associated molecular pattern molecules at the crossroads of Inflammation and cancer. Neoplasia. 2009; 11: 615-628.
- Sparvero LJ, Asafu-Adjei D, Kang R, Tang D, Amin N, Im J, et al. RAGE (Receptor for Advanced Glycation Endproducts), RAGE Ligands, and their roles in Cancer and Inflammation. J Transl Med. 2009; 7: 17.
- Logsdon, CD, Fuentes MK, Huang EH, Arumugam T. RAGE and RAGE ligands in cancer. Curr Mol Med. 2007; 7: 777-789.
- Rojas A, Figueroa H, Morales E. Fueling inflammation at tumor microenvironment: the role of multiligand/RAGE axis. Carcinogenesis. 2010; 31: 334-341.
- Hoffman MA, Drury S, Fu C, Qu W, Taguchi A, Lu Y, et al. RAGE mediates a novel proinflammatory axis: a central cell surface receptor for S100/calgranulin polypeptides. Cell. 1999; 97: 889-901.
- Hsieh HL, Schafer BW, Sasaki N, Heizmann CW. Expression analysis of S100 proteins and RAGE in human tumors using tissue microarrays. Biochem Biophys Res Commun. 2003; 307: 375-381.
- Kostova N, Zlateva S, Ugrinova I, Pasheva E. The expression of HMGB1 protein and its receptor RAGE in human malignant tumors. Mol Cell Biochem. 2010; 337: 251-258.
- Ellerman JE, Brown CK, deVera M, Zeh HJ, Billiar T, Rubartelli A, et al. Masquerader: high mobility group box-1 and cancer. Clin Cancer Res. 2007; 13: 2836-2848.
- Rai V, Touré F, Chitayat S, Pei R, Song F, Li Q, et al. Lysophosphatidic acid targets vascular and oncogenic pathways via RAGE signaling. J Exp Med. 2012; 209: 2339-2350.
- Kang R, Tang D, Schapiro NE, Livesey KM, Farkas A, Loughran P, et al. The receptor for advanced glycation end products (RAGE) sustains autophagy and limits apoptosis, promoting pancreatic tumor cell survival. Cell Death Differ. 2010; 17: 666–676.
- Kuniyasu H, Oue N, Wakikawa A, Shigeishi H, Matsutani N, Kuraoka K, et al. Expression of receptors for advanced glycation end-products (RAGE) is closely associated with the invasive and metastatic activity of gastric cancer. J Pathol. 2002; 196: 163-170.
- Malik P, Chaudhry N, Mittal R, Mukherjee TK. Role of receptor for advanced glycation end products in the complication and progression of various types of cancers. Biochim Biophs Acta. 2015; 1850; 1898-1904.
- Kikuchi N, Horiuchi A, Osada R, Imai T, Want C, Chen X, et al. Nuclear expression of S100A4 is associated with aggressive behavior of epithelial ovarian carcinoma: an important authocrine/paracrine factor in tumor progression. Cancer Sci. 2006; 97: 1061-1069.
- Taguchi A, Blood DC, del Toro G, Canet A, Lee DC, Qu W, et al. Blockade of RAGE-amphoterin signaling suppresses tumour growth and metastases. Nature. 2000; 18: 354-360.
- Tekabe Y, Li Q, Rosario R, Sedlar M, Majewski S, Hudson BI, et al. Development of receptor for advanced glycation end products-directed imaging of atherosclerotic plaque in a murine model of spontaneous atherosclerosis. Circ Cardiovasc Imaging. 2008; 1: 212-219.
- Tekabe Y, Shen X, Luma J, Weisenberger D, Yan SF, Haubner R, et al. Imaging the effect of receptor for advanced glycation endproducts on angiogenic response to hindlimb ischemia in diabetes. EJNMMI Res. 2011; 1: 3.
- Tekabe Y, Anthony T, Li Q, Ray R, Rai V, Zhang G, et al. Treatment effect with anti-RAGE F(ab')2 antibody improves hind limb angiogenesis and blood flow in Type 1 diabetic mice with left femoral artery ligation. Vasc Med. 2015; 20: 212-218.
- Connolly DC, Hensley HH. Xenograft and transgenic mouse models of epithelial ovarian cancer and non invasive imaging modalities to monitor ovarian tumor growth in situ- applications in evaluating novel therapeutic agents. Curr Protoc Pharmacol. 2009; 45: 14.
- Tomayko MM, Reynolds CP. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother Pharmacol. 1989; 24: 148-154.
- DiNorcia J, Moroziewicz DN, Ippagunta N, Lee MK, Foster M, Rotterdam HZ, et al. RAGE signaling significantly impacts tumorigenesis and hepatic tumor growth in murine models of colorectal carcinoma. J Gastrointest Surg. 2010; 14: 1680-1690.
- Tateno T, Ueno S, Hiwatashi K, Matsumoto M, Okumura H, Setoyama, et al. Expression of receptor for advanced glycation end products (RAGE) is related to prognosis in patients with esophageal squamous cell carcinoma. Ann Surg Oncol. 2009; 16: 440-446.
- Hudson BI, Strickland MH, Grant PJ. Identification of polymorphisms in the receptor for advanced glycation end products (RAGE) gene: prevalence in type 2 diabetes and ethnic groups. Diabetes. 1998; 47: 1155-1157.
- Xia W, Xu Y, Mao Q, Dong G, Shi R, Wang J, et al. Association of RAGE polymorphisms and cancer risk: a meta-analysis of 27 studies. Med Oncol. 2015; 32: 442.
- Zhang S, Hou X, Zi S, Wang Y, Chen L, Kong B. Polymorphisms of receptor for advanced glycation end products and risk of epithelial ovarian cancer in Chinese patients. Cell Physiol Biochem. 2013; 31: 525-531.