Mini Review
Nuclear Scanning in Evaluation and Treatment of Thyroid Disorders: A Beginners Guide
Vandana K Dhingra*
Department of Nuclear Medicine, All India Institute of Medical Sciences, India
*Corresponding author: Vandana K Dhingra, Department of Nuclear Medicine, All India Institute of Medical Sciences, India
Published: 04 Apr, 2017
Cite this article as: Dhingra VK. Nuclear Scanning in
Evaluation and Treatment of Thyroid
Disorders: A Beginners Guide. Clin
Oncol. 2017; 2: 1260.
Abstract
Thyroid disorders are common worldwide and the biggest fear amongst most patients with neck swellings is thyroid cancer. We need simple techniques in our clinics to diagnose and treat patients with thyroid disorders in order to initiate the right treatment as well as to alleviate patient’s anxiety. Incomplete information and ignorance regarding these techniques prevents many clinicians to benefit from these simple and cost effective methods. Thus radioisotope modalities are still underutilized in many countries. We put together a short review by compiling our experience of common ways to effectively utilize radioisotope techniques in thyroid clinics along with literature review from current guidelines in a most simple and easy to understand way for the clinician.
Introduction
Common thyroid disorders we may come across in our clinic include hypothyroidism, hyperthyroidism as well as nodular thyroid disease (solitary and multinodular goitre). Most thyroid disorders are evaluated with the a combination of the following modalities: the biochemical (Thyroid functions or T4, TSH) values, radiological imaging (US/CT Scan) for anatomical status/ nodules, functional imaging (nuclear scanning and uptake) and pathological (FNAC, Biopsy ) evaluation. The Nuclear Medicine or thyroid scanning remains one of the lesser known modalities in general practices due to its limited availability. We would like to bring forth the practical aspects of utilization of nuclear scanning in practice for decision making in thyroid disorders and highlight the ease with which a nuclear scan can be performed. We will also introduce to the reader about the common Nuclear Medicine therapeutic modalities for thyroid disorders.
Introduction to Nuclear Imaging of the Thyroid (Thyroid Scintigraphy)
Thyroid gland has the unique ability to take up iodine an essential
component of its hormones. The phenomenon of accumulation of iodine in the thyroid gland allowed for the use of iodine
isotopes in the diagnosis of thyroid disease as early as about 70 years ago, although the mechanism
of iodine uptake at the molecular level has been carefully examined until the late twentieth
century. In 1939, a group of scientists from the University of Berkeley documented the uptake of radioactive
iodine in human thyroid for the first time. This gave rise to first therapeutic radioiodine applications
in patients with hyperthyroidism and thyroid cancer [1,2].
The uptake of iodine in the thyroid gland is attributed to the sodium-iodide symporter (NIS),
described in 1993 by Kaminsky et al. [3] though the uptake of iodine by the thyroid cells is still
widely used in the evaluation of thyroid function by means of radioiodine uptake test and thyroid
Scintigraphy; thyroid evaluation with Technetium Pertechnetate-TC04 (Thyroid Scan) remains a
mainstay in functional evaluation of thyroid disorders.
In addition to the morphological information obtained on the basis of ultrasonography, thyroid
scintigraphy visualizes the distribution of active thyroid tissue. Therefore, the general indications for
scintigraphy are quite wide and include both single thyroid nodule, and multinodular goitre. The test
is also used to evaluate the extent of retrosternal goitre (when ultrasound is not able to visualize the
lower pole of the thyroid gland) and in suspected ectopic thyroid. In the case of goitre without clear
dominance of one of the nodules, scintigraphy is used to select the biopsy site. Thyroid scintigraphy
should be performed in all patients with nodular goitre undergoing treatment with radioiodine,
since it allows to assess the anatomical distribution of active thyroid tissue, which may be important
in the selection of therapeutic 131I activity [4,5]. Keeping in mind the widespread availability and
expertise of Ultrasound and needle biopsy the indications have been made very specific to bring maximum benefits from the scanning. In 2009–2010 American Thyroid Association (ATA) and other guidelines have published their
recommendations suggesting that the thyroid scintigraphy is useful
only in patients with nodule diameter greater than 1 cm, provided
that TSH concentration is subnormal. The role of scintigraphy in this
case is to show whether a nodule, that is manifested in ultrasound, is
a focus of excessive hormonal activity (hot nodule) or accumulates
the radiotracer at a level similar to or lower than the rest of the gland
(warm or cold nodule). Since the hot nodules are rarely malignant,
according to ATA guidelines fine-needle biopsy is not necessary in
such a case. At the same time, performing Scintigraphy in all patients
with nodular goitre is not recommended by experts, since adequate
assessment obtained with ultrasound and cytology is sufficient in
most cases [6].
Patient preparation for thyroid scintigraphy
Usually no prior patient preparation is needed for Thyroid
Scintigraphy.
In case the patient is taking thyroid hormone replacement
therapy or iodine, the study should be done four to six weeks after
stopping these drugs.
The female patients who are pregnant or breast feeding the
babies should inform the nuclear medicine physician before taking
a diagnostic test.
Although the radiation exposure involved is very low, however,
in case of pregnancy the procedure would be performed only id really
needed at that point (benefit vs. risk evaluation).
Table 1
The Basic Gamut of Nuclear Medicine in Thyroid Disorder Evaluation
While referring and interpreting results of thyroid Scintigraphy,
the physician should be aware of the current situation of the patient:
• History of thyroid disease
• Ultrasound findings
• Current TSH
Used drugs, in particular thyroid hormones, antithyroid drugs
and iodine-containing agents (amiodarone, disinfectant and
expectorant drugs), these may have effect on overall functioning of
the thyroid gland as well as on scan findings
Thyroid Scintigraphy is functional imaging and is interpreted
with full knowledge of the patient status for optimal results [6].
• Post surgical detection of remnant /metastasis –diagnostic
• Ablation of remnant/metastasis –therapeutic
Principles of interpretation of thyroid scan
Thyroid scintigraphy produces an image of distribution of
radiotracer in thyroid parenchyma. It helps to diagnose thyroid
diseases on the basis of level of radiotracer uptake as compared to surrounding structures, radiotracer distribution in thyroid, any
extrathyroidal uptake etc. It gives an idea about thyroid location.
It also helps to know thyroid morphology including its size and
overall & regional level of thyroid function both qualitatively and
quantitatively.
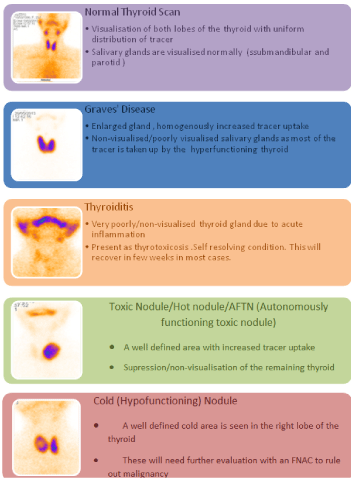
A normal thyroid scan
Normally the gland is symmetrical and the lateral borders of lobes
are straight to convex. Tracer is normally seen in salivary glands and
in capillary network of the neck tissue also, called as ‘blood pool’. This
is seen as a light background along the neck contour.
Graves’ disease
Typical clinical features of Graves’ with biochemical parameters
and a thyroid scan would complete workup of these patients.
Radioiodine therapy is the treatment of choice in non-complicated,
small to medium sized goitres . Thyroid Scintigraphy usually shows
uniform diffuse enlargement. Tracer is uniformly distributed in the
thyroid. Not normally seen pyramidal lobe (a remnant of thyroglossal
duct) can be seen in a hypertrophied gland. Tracer is barely trapped
in salivary glands due to less tracer availability for extraction to them
consequent to higher trapping by thyroid.
Thyroiditis
The clinical features on recent onset hyperthyroid symptoms
accompanied by upper respiratory tract infection features or neck
pain are classically accompanied by a scan image of very low or
no tracer uptake. This is due to the stunned thyrocytes during the
acute phase of imflammation. These patients can be conservatively
managed with symptomatic therapy in the form of NSAIDs, low dose
steroids, beta blockers. A repeat scan after 3 months would show a
resolving thyroid in various stages showing various types of uptake.
The scan ensures recovery has been attained and serves as a baseline
for future.
Solitary thyroid nodule
They are usually palpable nodules and are mainly evaluated to
rule out malignancy. On thyroid scanning the following types are
identified:
• Warm: Tracer uptake equivalent to normal tissue, usually
means normal level of function in that area. Have a low probability
of malignancy.
• Cold: Tracer uptake nil or less than normal tissue, means nil
or less than normal level of function. Such nodules could result from
cystic changes, fibrosis, haemorrhage, adenoma, malignancy etc.
Have a high probability of malignancy.
• Hot: Tracer uptake higher than normal tissue, means more than
normal level of function. They can be Autonomously Functioning
Thyroid Nodules (AFTN). Have a low probability of malignancy.
Multinodular Goitre
It usually develops in the population living in iodine deficient regions due to periods of nutritional iodine deficiency interspersed
with iodine sufficiency resulting in compensatory thyroid
hypertrophy and regression to normalcy respectively. This causes
nodularity, focal haemorrhages, calcifications, cyst formation and
scarring in the gland. Thyroid size is usually grossly increased with
non-uniform enlargement in general. The tracer distribution can also
be inhomogeneous in case of a nodular pathology because different
areas/nodules can have different levels of function. Therefore, on
scintigraphy the tracer trapping is variable, higher/lower than
normal in some areas. Some nodules may become autonomous i.e.
independent of TSH control and can show hyperfunction resulting in
toxic MNG. In such cases areas of increased radiotracer uptake i.e. hot
nodules can be seen. The toxic multinodular goitre needs treatment
to prevent long term complications of suppressed TSH. Usually
high doses (15-25 mCi) of Radioiodine are very successful for subtle
control of the subclinical toxicosis status. However if compression
symptoms are present then surgery is more beneficial.
Toxic autonomous nodule
It is also called as Autonomously Functioning Thyroid Nodule
(AFTN) i.e. independent of TSH control. Image shows a focal
increased tracer uptake confined to a nodule that occupies most or
all of the thyroid lobe. The remaining gland is non-visualized due to
total suppression of TSH stimulation . These patients can be treated
successfully with radioiodine therapy/surgery.
Thyroid dysgenesis/Agenesis/Ectopic thyroid
Thyroid scanning with I-131 is the gold standard for imaging
of functioning thyroid tissue in the body. A screening with Tc-99m
scanning can be done followed by I-131 imaging if indicated.
Figure 1
Thyroid Cancer Management
Radioiodine is used widely in management of differentiated
thyroid cancers. Initially role of thyroid Scintigraphy is in the
diagnosis by the way of detection of cold nodules.
In any differentiated thyroid cancer (papillary/follicular)
thyroidectomy is the recommended modality of therapy (Details of
types of surgery are beyond the scope of this text). Total thyroidectomy
followed by a radioiodine ablation is the treatment of choice. Ablation
means destruction of all the thyroid tissue (including normal) with
Radioiodine (I-131).
The principles involved are:
1. It would help to locate any functioning metastasis (whole
body scanning is done post therapy).
2. Follow up with Serum Thyroglobulin and /or I-131
scanning- it becomes easier to detect any recurrence after ablation as
the thyroglobulin levels would show a subtle rise which is not reliable
if the ablation has not been done. Common areas involved are local
recurrence (neck); cervical/mediastinal lymph node metastasis; lung
metastasis; bone metastasis.
3. Any recurrence/metastasis can be managed with
radioiodine therapy in addition to surgical intervention.
PET (Positron Emission Tomography)
PET is a nuclear medicine modality commonly used for evaluation of cancers. The details are beyond the scope of this text but we can brush upon the common areas in thyroid disorders where PET (with F-18 FDG) imaging plays a role : • Thyroid incidentalomas are seen in about 2% of patients undergoing PET scan for other problems. Evaluation of these can lead to detection of several occult/asymptomatic thyroid cancers. • Evaluation of equivocal thyroid nodules by scanning and cytology [7]. • Management of thyroid carcinomas.
Conclusion
Thyroid Scanning/Scintigraphy is a simple, non-invasive,
cost effective tool for evaluation of most thyroid disorders. If used
appropriately it can answer the queries which a clinician sitting in
his clinic with a ‘patient’ and a ‘thyroid profile/US/FNA report’ needs
to answer.
In addition to diagnostics, radioiodine (I-131) therapy plays
a very important role in treatment of hyperthyroidism. It can be a
surgeon’s partner in effective management of thyroid carcinomas.
Acknowledgment
The authors are grateful for Foundation Medicine performing the genetic profiling of this distinctive tumor.
References
- Becker DV, Sawin CT. Radioiodine and thyroid disease: the beginning. Semin Nucl Med. 1996;26:155-164.
- Keston AS, Ball RP, Frantz VK, Palmer WW. Storage of radioactive iodine in a metastasis from thyroid carcinoma. Science. 1942;2466:362–363.
- Kaminsky SM, Levy O, Salvador C, Dai G, Carrasco N. The Na+/I- symporter of the thyroid gland. Soc Gen Physiol Ser. 1993;48:251–262.
- Meller J, Becker W. The continuing importance of thyroid scintigraphy in the era of high-resolution ultrasound. Eur J Nucl Med. 2002;29:S425–S438.
- Becker D, Charles ND, Dworkin H. Procedure guideline for thyroid Scintigraphy: 1.0. Society of Nuclear Medicine. J Nucl Med. 1996;37:1264–12669.
- Cooper DS, Doherty GM, Haugen BR. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167-1214.
- Sebastianes FM, Cerci JJ, Zanoni PH, Soares J, Chibana LK, Tomimori EK, et al. Role of 18F-fluorodeoxyglucose positron emission tomography in preoperative assessment of cytologically indeterminate thyroid nodules. J Clin Endocrinol Metab. 2007;92:4485-4488.