Commentary
Recent Molecular Immunological Chemotherapy for Gastrointestinal Malignancies
Masaya Mukai1*, Takayuki Tajima2, Sayuri Hasegawa1, Daiki Yokoyama1, Takuya Koike1, Shinichirou Hiraiwa3, Tomoko Sugiyama3 and Takuma Tajiri3
1Department of Digestive Surgery, Tokai University Hachioji Hospital, Japan
2Departmentof Digestive Surgery, Tokai University Oiso Hospital, Japan
3Department of Pathology, Tokai University Hachioji Hospital, Japan
*Corresponding author: Masaya Mukai, Department of Digestive Surgery, Tokai University Hachioji Hospital, Japan
Published: 21 Mar, 2017
Cite this article as: Mukai M, Tajima T, Hasegawa S,
Yokoyama D, Koike T, Hiraiwa S, et
al. Recent Molecular Immunological
Chemotherapy for Gastrointestinal
Malignancies. Clin Oncol. 2017; 2:
1233.
Abstract
Efficacy of anticancer agents is generally dose-dependent and thus is determined by the Maximum
Tolerated Dose (MTD) and Dose-Limiting Toxicities (DLT). Based on new theories such as the role
of immune checkpoints in tumor cell survival, it seems important to investigate the optimum dosing
method for antibody-drug conjugates that can have multimodal pharmacological actions, rather
than inducing bone marrow suppression/immunosuppression at the MTD/DLT like traditional
chemotherapy, allowing the cytotoxic anticancer agent to be administered at the Minimally Effective
Cytotoxic Dose (MECD) that maintains host antitumor immunity together with cytotoxic activity.
At present, both molecular-targeting agents and immune checkpoint inhibitors are available. As a
future treatment strategy, we need to develop regimens that do not cause bone marrow suppression
and do not require suspension or discontinuation of treatment due to toxicities. With such new
regimens, cancer cells will be killed effectively while adverse reactions are reduced, so patients can
enjoy improved high quality of life and longer progression-free survival. It is strongly anticipated
that molecular-targeting therapy will be developed for gastrointestinal cancer, which will allow the
total dose to be given safely at optimal schedules.
Keywords: Molecular-targeting therapy; Minimally effective cytotoxic dose (MECD); Maximum tolerated dose (MTD); Dose-limiting toxicities (DLT); Immunosuppression
Commentary
In recent years, molecular-targeting therapy directed against tumor mutations has made dramatic
advances. As a result, treatment of malignant melanoma, lung cancer, breast cancer, leukemia
and lymphoma has undergone significant changes [1]. Many tumor-specific antibodies are now
available, such as trastuzumab (an anti-HER2 humanized antibody) for breast cancer, rituximab (an
anti-CD20 chimeric antibody) for B cell non-Hodgkin lymphoma, and mogamulizumab (an anti-
CCR4 antibody) for adult T cell leukemia/lymphoma [1]. Furthermore, antibody-drug conjugates
that combine antibodies with anticancer agents, such as gemutuzumabo zogamicin (anti-CD33
humanized antibody + calicheamicin) for acute myeloid leukemia (AML), trastuzumab emtansine
(anti-HER2 antibody + tubulin polymerization inhibitor) for breast cancer, and brentuximab
vedotin (anti-CD30 antibody + microtubule inhibitor) for CD30-positive Hodgkin lymphoma,
have led to significant progress in pharmacological treatment strategies targeting specific tumors
[1]. Moreover, immune checkpoint inhibitors have become available, such as nivolumab (an anti-
PD-1/PD-L1 antibody) and ipilimumab (an anti-CTLA4 antibody), which are based on the new
concept of suppressing immune checkpoints that prevent the immune system from attacking cancer
cells. Some clinical studies have shown better results with these new agents than with conventional
cytotoxic anticancer agents [2-5]. In addition to utilizing CD4+ regulatory T cells (Treg) and
myeloid-derived suppressor cells (MDSC), which are potent immune inhibitory cells, to avoid the
host immune response, tumor cells inhibit the production/secretion of various cytokines such as
interleukin-10 (IL-10), Transforming Growth Factor (TGF)-β, and Vascular Endothelial Growth
Factor (VEGF). As a result, they become invisible to the host immune system. It is thought that
unresponsive Cytotoxic T Lymphocytes (CTL) inhibit CTLA4 signaling and PD-1/PD-L1 signaling
in the tumor environment, thus inducing potent anti-tumor immunity [6-10]. Molecular-targeting
therapy has expanded the options for treatment and has extended the progression-free survival
and overall survival of patients. Antibody drugs (high molecular weight agents) and low molecular
weight chemical entities can be classified into 1) agents that target the molecular biological characteristics of cancer cells and 2) agents that act on the tumor
microenvironment. The mechanisms of action of these new drugs
are quite different from those of traditional cytotoxic chemotherapy
agents. Mutations of oncogenes in cancer cells help to create a
suitable environment for the survival and growth of these cells. Since
cancer cells proliferate excessively, they are dependent on activation
signals from these gene mutations, which has been calledoncogene
addiction [11,12]. Many molecular-targeting agents are designed
to preferentially kill cancer cells by blocking the activation of
specific signaling pathways [13,14]. For example, three EGFR-TKI
agents (gefinitib, erlotinib, and afanitib) are already available that
target EGFR mutation-positive non-small cell lung cancer. When
resistance develops to these agents due to T790M mutation, the
third-generation agent osimertinib is now available. Moreover, two
agents (crizotinib and alectinib) are available for ALK-translocated
non-small cell lung cancer, while ceritinib can be used when tumors
develop resistance to these treatments. However, there is concern
that history may be repeated similar to the development of resistance
to many antibiotics. In other words, the continuous development
of new molecular-targeting agents for new cancer gene mutations
may be needed. Unfortunately, development of a new drug is both
hugely expensive and time-consuming, leading to a rapid increase
of drug prices that has placed significant pressure on the health
system in Japan and become an important social issue. The efficacy
of cytotoxic anticancer agents, such as those inhibiting the growth
of tumor cells and inducing apoptosis, is usually dose-dependent
and thus is defined by the Maximum Tolerated Dose (MTD) and
the Dose-Limiting Toxicities (DLT) [15-17]. Chemotherapy for
hematological malignancies may be fundamentally different from
that for solid tumors due to differences of cancer cell biology. To
treat hematological malignancies such as leukemia, potent remission
induction therapy is conducted according to the MTD/DLT with the aim of reducing the leukemic cell count to around 109 cells,
followed by allogeneic hematopoietic stem cell transplantation.
However, a novel theory of tumor immunity was recently proposed,
which suggests that unlocking immune checkpoints protecting
cancer cells in the tumor microenvironment will allow these cells to
be recognized by the host immune system, leading to selective and
efficient killing of tumor cells [2-5]. At present, trastuzumab has
been approved for some patients with HER2-positive gastrointestinal
cancer, specifically gastric cancer. In the case of colorectal cancer,
tumor suppressor genes such as P53/APC/DCC and oncogenes such
as K-ras/c-myc interact in a complex manner during a multi-step
process of carcinogenesis, and treatment that can selectively block
oncogenes (as is available for lung cancer) has not yet been identified.
Accordingly, panitumumab and cetuximab cannot be used unless the
most upstream RAS gene is maintained in colorectal cancer, while
bevacizumab and ramucirumab do not require this precondition
and are used relatively often in daily practice. However, appropriate
dose selection for combination chemotherapy such as FOLFOX
and FOLFIRI remains a challenge. Doses are selected based on the
Phase 3 randomized trial, where the regimen was selected based
on the MTD/DLT in Phase 1 and 2 studies performed in western
countries. In Japan, a lower percentage of patients experience bone
marrow suppression or suspension / discontinuation of therapy and
there are more patients who can complete treatment. Accordingly,
FOLFOX6 (oxaliplatin at 85mg/m2) is often preferred. For the IRIS
regimen that was developed in Japan, the dose of irinotecan was
recently reduced from 125mg/m2 to a range from 100mg/m2 to 75mg/
m2 (25% reduction) [18,19]. Sensitivity to this drug is closely related
to the level of expression of enzymes involved in its metabolism
and degradation. Based on new theories such as the role of immune
checkpoints in tumor cell survival, it seems important to investigate
the optimum dosing method for antibody-drug conjugates that can have multimodal pharmacological actions, rather than inducing
bone marrow suppression/immunosuppression at the MTD/DLT
like traditional chemotherapy, allowing the cytotoxic anticancer
agent to be administered at the Minimally Effective Cytotoxic
Dose (MECD) that maintains host antitumor immunity together
with cytotoxic activity. Currently, both molecular-targeting agents
and immune checkpoint inhibitors are available. As a future
treatment strategy, we need to develop regimens that do not cause
immunosuppression / bone marrow suppression and do not require
suspension or discontinuation of treatment due to toxicities. With
such new regimens, cancer cells will be killed effectively while
adverse reactions are reduced, so patients can enjoy improved high
Quality Of Life (QOL), high Quality-Adjusted Life Year (QALY)
and longer progression-free survival. Also for colorectal cancer, new
chemotherapy is being sought that allows the specified dose to be
administered safety in a defined period.
Case presentation of chemotherapy using the Minimally
Effective Cytotoxic Dose (MECD)
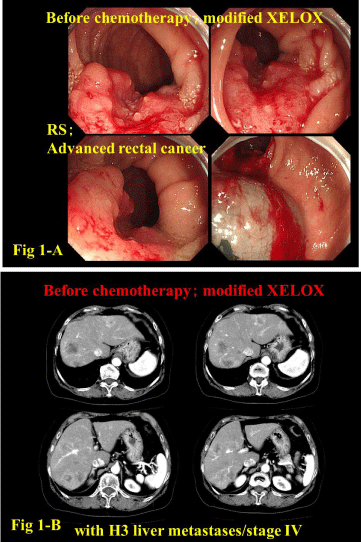
A 78-year-old woman with inoperable advanced rectal cancer
(stage IV) underwent palliative chemotherapy for multiple liver
metastases (H3). Her height, body weight, and body surface area
were 159.0cm, 59.0kg, and 0.781m2, respectively. Both the patient
and her family members were elderly and only mild chemotherapy
was desired. The primary tumor showed ulceration. She was treated
with XELOX without the use of Avastin (bevacizumab). The dose of
XELOX was reduced to approximately 50% of the recommended dose
(second dose reduction step: oxaliplatinat 60mg/m2). Five courses
were administered [20,21]. Colonoscopy findings and liver CT scans
before and after chemotherapy are shown in Figure 1-A and 1-B,
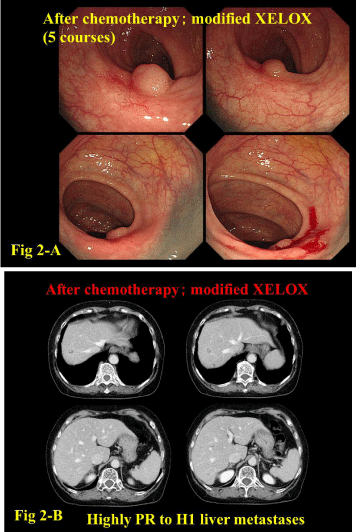
respectively. After treatment, the primary tumor almost disappeared
(follow-up biopsy; cancer +) and H3 metastases improved to H1
(Figure 2-A, 2-B). Although conversion surgery was recommended,
the patient refused it. At present, her performance status is 0. In order
to achieve complete remission of multiple lesions, an additional 5
courses of outpatient chemotherapy combined with Avastin are being
conducted.
Figure 1
Figure 1
Colonoscopy findings and liver CT scans before and after
chemotherapy are shown in Figure 1-A and 1-B, respectively.
Figure 2
Figure 2
After treatment, the primary tumor almost disappeared (follow-up
biopsy; cancer +) and H3 metastases improved to H1 (Figure 2-A, 2-B).
References
- Yano S, Mouri H. An outline of Molecular Targeting Therapy. The textbook for general clinical oncologist by Japanese Board of Cancer Therapy. 2016.
- Spranger S, Spaapen RM, Zha Y. Up-regulation of PD-L1, IDO, and T (regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci Transl Med. 2013; 5: 2013.
- Snyder A, Makarov V, Merghoub T. Genetic Basis for Clinical Response to CTLA-4 Blockade in Melanoma. N Engl J Med. 2014; 2189-2199.
- Robert C, Long GV, Brady B. Nivolumab in Previously Untreated Melanoma without BRAF Mutation. N Engl J Med. 2015; 372: 320-330.
- Gettinger S, RizviNA, Chow LQ. Nivolumab Monotherapy for First-Line Treatment of Advanced Non–Small-Cell Lung Cancer. J Clin Oncol. 2016; 34: 2980-2987.
- Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009; 162-174.
- Youn JI, Gabrilovich DI. The biology of myeloid-derived suppressor cells: The blessing and the curse of morphological and functional heterogeneity. Eur J Immunol. 2010; 40: 2969-2975.
- Gabrilovich DI, Ostrand-Rosenberg S, Bronte V. Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol. 2012; 12: 253-268.
- Simpson TR, Li F, Montalvo-Ortiz W, Sepulveda MA, Bergerhoff K, Arce F, et al. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-definites the efficacy of anti-CTLA-4 therapy against melanoma. J Exp Med. 2013; 201: 1695-1710.
- Maeda Y, Nishikawa H, Sugiyama D, Ha D, Hamaguchi M, Saito T, et al. Detection of self-reactive CD8⁺ T cells with an anergic phenotype in healthy individuals. Science. 2014; 346: 1536-1540.
- Weinstein IB. Cancer. Addiction to oncogenes-the Achilles-heel of cancer. Science. 2002; 297: 63-64.
- Weinstein IB, Joe AK. Mechanisms disease: Oncogene addiction—a ratinonale for molecular targeting in cancer therapy. Nat Clin Pract Oncol. 2006; 3: 448-457.
- Sharma SV, Settleman J. Oncogene addiction: Setting the stage for molecularly targeted cancer therapy. Genes Dev. 2007; 21: 3214-3231.
- Weinstein IB, Joe A. Oncogene addiction. Cancer Res. 2008; 68: 3077-3080.
- Fuse E, Kobayashi T, Inaba M. Application of pharmacokinetically guided dose escalation with respect to cell cycle phase specificity. J Natl Cancer Inst. 1994; 86: 989-996.
- Newell DR. Pharmacologically based phase I trials in cancer chemotherapy. Hematol Oncol Clin N Am. 1994; 257-275.
- Eisenhauer EA, O’Dwyer PJ, Christian M. Phase I clinical trial design in cancer drug development. J Clin Oncol. 2000; 18: 684-692.
- Muro K, Boku N, Shimada Y, Tsuji A, Sameshima S, Baba H, et al. Irinotecan plus S-1 (IRIS) versus fluorouracil and folinic acid plus irinotecan (FOLFIRI) as second-line chemotherapy for metastatic colorectal cancer: a randomized phase 2/3 non-inferiority study (FIRIS study). Lancet Oncol. 2010; 853-860.
- Takaoka T, Kimura T, Shimoyama R, Kawamoto S, Sakamoto K, Goda F, et al. Panitumumab in combination with irinotecan plus S-1 (IRIS) as second-line therapy for metastatic colorectal cancer. Cancer Chemother Pharmacol. 2016; 78: 397-403.
- Cassidy J, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, et al. Randomized Phase III Study of Capecitabine Plus Oxaliplatin Compared with Fluorouracil/Folinic Acid Plus Oxaliplatin As First-Line Therapy for Metastatic Colorectal Cancer. J Clin Oncol. 2008; 26: 2006-2012.
- Díaz-Rubio E, Gómez-España A, Massutí B, Sastre J, Abad A, Valladares M, et al. First-line XELOX plus bevacizumab followed by XELOX plus bevacizumab or single-agent bevacizumab as maintenance therapy in patients with metastatic colorectal cancer: the phase III MACRO TTD study. Oncologist. 2012; 17: 15-25.