Review Article
The Impact of Climate Change on Paediatric Oncology
Dezső Schuler, Miklós Garami and Zsuzsanna Jakab*
Department of Paediatrics, Faculty of Medicine, Semmelweis University, Budapest, Hungary
*Corresponding author: Zsuzsanna Jakab, Department of Paediatrics, Semmelweis University, Hungary
Published: 17 Mar, 2017
Cite this article as: Schuler D, Garami M, Jakab Z. The
Impact of Climate Change on Paediatric
Oncology. Clin Oncol. 2017; 2: 1225.
Abstract
Background: According to some experts, climate change will be the greatest health-related hazard
in the 21st century. It is understandable that harmful effects due to climate change will be the greatest
among poor populations living in the hot climate zones; however, they will have impacts worldwide.
Aims: To summarize the evidence of climate change in the field of paediatric oncology.
Methods: Articles relating to climate change and paediatric oncology were reviewed. The review
was restricted to studies that included information on climate change and effects on subjects 18
years of age or younger.
Results: Currently detected harmful effects of climate change are expected to cause further
unfavourable consequences within the health system and therefore within the paediatric oncology:
1. Current relationship of certain tumours to geographic regions, as well as the relative occurrence
of these tumours, is changing.
2. Numbers of cases of childhood malignant diseases are expected to increase.
3. As a result of migration from economically underdeveloped countries, more foreign patients are
expected to arrive in the developed world and they will require health care.
Conclusion: Increasing health-related effects of climate change are expected to lead to new
challenges for the health care system – including the important field of paediatric oncology – that
must be dealt with as soon as possible.
Keywords: Climate change; Paediatric oncology; Migration; Incidence; Mortality; Socioeconomic status
Introduction
A number of publications have drawn attention to the serious consequences of global warming
caused by accumulation of greenhouse gases (mostly carbon dioxide) and potential unfavourable
effects global warming may have on children’s health. According to some experts, climate change
will be the greatest health-related hazard in the 21st century [1]. These harmful effects will be greatest
among poor populations living in the hot climate zone; however, they will have impacts worldwide
[2]. In the wealthy (developed) countries, childhood population contributes 20% to the total
population, while in developing countries it can go up till 40% [3]. However besides their number
and proportion it is more important to recognize the much higher sensitivity to all climate effects
that children experience compared to adults. Especially in the light of the importance children have
as trustees of the future [4].
Children suffer disproportionately more any harmful effects of the rapidly changing climate
[5,6]. Paediatricians should be adequately informed about these trends in order to be able to help
in ameliorating these adverse effects in the hope of health and well being of future generations.
Hence it is of high importance, the International Society of Social Paediatrics gives an overview
about the problem of climate change for children and propose action against these adverse effects
[7]. However among the few paediatric publications available we have not found any data about the
impact of climate change on paediatric malignant diseases. The aim of this publication is to describe
some aspects of this problem. We address two questions: what changes can be expected due to
climate change and what can be done about the anticipated harmful effects, as well as for prevention.
Methods
A systematic search for publications on the effect of climate change on paediatric oncology in PubMed and Medline found almost no citations. We therefore searched for publications on association of childhood cancer and consequences of climate change. The main search terms included: paediatric cancer and epidemiology (geographic patterns); childhood cancer and climate change; climate change and migration; population mixing and malignant diseases; transcultural-, communication problems and migration; (childhood) cancer and socioeconomic environment. Inclusion criteria were limited to representative publications related to different consequences of climate change. References cited in selected papers were tracked to find further studies related to the topic. We also used data described in handbooks by Pizzo and Poplack and by Kliegman et al. [8,9].
Table I
Results
Regional differences of the relative frequency of paediatric malignancies
There are significant differences in the relative distribution of
paediatric tumours between various geographical regions [10-27].
Selected differences are summarized in Table 1. Due to increasing
population mixing all over the world, the relative frequency of
paediatric tumours experienced in the developed world is expected
to change, too. In the developed world, leukaemia represents the
most frequent form of childhood cancer followed by tumours of the
central nervous system. Burkitt’s lymphoma makes up only 1.3% of
childhood tumours while in Africa it accounts for almost 50% of all
childhood tumours. This huge difference is explained by different
pathogenesis. While exposure to Epstein-Barr virus is proven in 95%
of the African Burkitt’s tumours, the same virus primarily causes
infectious mononucleosis or asymptomatic infection in temperate
zones. Similarly, T-cell Acute Lymphoid Leukaemia (ALL) caused
by the Human T-cell Lymphotropic Virus type 1 (HTLV-1) that
is observed in countries of the Caribbean region and Japan does
not occur in other regions. In Brazil, the childhood incidence of
adrenocortical carcinoma is 2.8 per million, ten times higher than the
number diagnosed in other regions.
Among the tumours with great differences in geographical
distribution, Kaposi’s sarcoma is of high importance. It is a very rare
disease within the temperate zone and occurs almost exclusively in
elderly people [28]. However, in Africa it makes up 9 to 13% of all
tumours, and it is often diagnosed in children, too. It is frequently
associated with Acquired Immune Deficiency Syndrome (AIDS);
in the United States 95% of all Kaposi’s sarcoma cases occur in
AIDS patients. Kaposi’s sarcoma accompanying AIDS has become scarcer in countries with well-developed health care system since the
introduction of antiretroviral therapy; on the other hand, in Africa it
continues to spread [28]. In Zambia researchers analyzed all Kaposi’s
sarcoma cases diagnosed between 1980 and 1992 [12]. Of 915 cases,
9.25% involved people younger than 14 years of age. The age range
was 7 months to 14 years, with an average of 5.62 years. In tropical
countries, incidence of Hodgkin’s lymphoma of childhood and
adolescence has increased due to the pandemic occurrence of HIV
infections [13].
In developing countries, liver cancer is also more frequent in
children because of endemic hepatitis B. 70% to 90% of children born
to HBV-positive mothers (early antigen: HBeAg) are reported to be
infected within 2 years after birth, and 90% of these patients – similar
to the conversion rate of elderly people – will convert to chronic
active hepatitis and to cirrhosis that may lead to liver carcinoma.
Vertical transmission of HBV infection of newborns and infants in
developed countries has decreased drastically since the introduction
of mandatory HbsAg screening of mothers and preventive vaccination
[29]. Unfortunately, this does not characteristically happen in
economically underdeveloped countries.
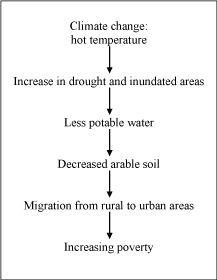
Climate change and Migration
Because of climate change, the South Temperate Zone will be
marked by increasing drought; at the same time, inundated areas are
spreading, vector-borne diseases increasing, and natural disasters are
becoming more and more frequent [30]. Due to drought arable soil
and water resources continue to decrease. Currently, food shortages
and starvation are on the increase; and today these problems
affect approximately 800 million people. Furthermore, drinking
water supplies are on the decrease. The current situation is hardly
comprehensible: 1.5 billion people lack clean potable water [31-35].
In addition, 2.6 billion people do not receive elementary health care
- and these numbers are going to increase [31,34]. All these problems
may lead to pandemics with enteropathogens [35]. Social tensions and
the number of international conflicts are increasing. Taken together,
these trends are expected to lead to increasing population migration
as people attempt to escape to safer areas [4]. Children among the 200
million people who are likely to have to flee their homes as deserts
spread and sea levels rise will suffer the most because of their high
vulnerability to diseases [36].
Poverty and the lack of arable land result in a flow of rural
populations to cities, leading to increased urbanization. On a larger scale, populations from tropical and low income countries are
migrating to other countries hoping for a better living. In 2007 16.7%
of the population of Sweden consisted of immigrants. In the United
States, just the Central and South American immigrants accounted
for 15% of the total population, and by 2050 this will probably
increase to 30% [37]. The proportion of people coming from Africa in
France, Italy and the Benelux countries is also increasing. In Germany
an ever-increasing proportion of urban population is of Turkish or
Chinese origin, from the migration crisis of 2015 of Syrian, Afghan
and Pakistan origin. Even in East-European countries like Hungary
the proportion of Chinese immigrants keeps increasing.
Climate change and the incidence of malignancies
Foreseeable challenges of climate change may include not only the
occurrence of tumours that are now unusual in developed countries
but also increased incidence of childhood cancer.
The role of climate change in increased incidence of skin cancer
is documented [38-42]. Furthermore, tumour-causing effect of
urbanisation has been shown, and as noted, urbanisation is increasing
as the climate changes. In 2005, Adelman et al. [42] studied the
incidence of acute lymphoid leukaemia in patients less than 5 years
old in large cities of various size and within the regions close to or far
from large cities [43]. They found an increased relative occurrence of
leukaemia in parallel with the size of community in which children
lived. In studies on the incidence of cancer in the vicinity of nuclear
power plants, higher rate of immigration within a short time period
was found to be associated with increased incidence of malignant
diseases. In Great Britain, Kinlen et al. [44] studied variations in
occurrence of childhood leukaemia and non-Hodgkin’s lymphoma
in regions where more than 1000 people were working on rural nonnuclear
construction sites at least 20 km from large towns. They stated
that during the construction and the following year there were 37%
more cases of leukaemia and non-Hodgkin’s lymphoma, which was
similar to or even exceeded the increase of frequency seen within the
construction area of the Sellafield nuclear plant.
A study of several case clusters of childhood leukaemia and
lymphoma has demonstrated that besides population-mixing
where sudden increase in population, there may be mixing when
there is higher proportion turnover without rapid growth; both of
these processes may contribute to an increased incidence of these
childhood malignancies, possibly as the result of infectious agents
that induce malignancies [45]. In Japan the occurrence of the
leukaemia caused by HTLV-1 infection was increased significantly by
migration from the endemic area – redrawing the epidemic map of
Tokyo [14]. In Hungary, Nyári et al. [46] demonstrated an increased
incidence of malignant diseases among children due to migration.
In France the effect of change of domicile between 1990 and 1999
on incidence of childhood hemopoietic malignancies was studied.
They found that migration increased the number of malignant cases,
and the farther away a person came from the greater the change
was. The higher percentage of population was not native born
(newly arrived population), the greater the relative risk of childhood
leukaemia was found [47-48]. From publications summarized here, it can be predicted that climate change will potentially increase the
incidence of childhood cancer due to a higher rate of migration from
the developing world, by population mixing, and furthermore by
increasing exposure of children to negative environmental effects of
climate change like ultraviolet radiation [40-41]. Table 2 However,
the exact magnitude of all these effects cannot be yet calculated.
Climate change and socio-economic changes
Besides urbanization and migration there is another link between
climate change and malignant diseases: increased poverty caused
by the climate change [34]. Figure 1 poverty means deprivation: an
unhealthy lifestyle, destitution and low access to health-care system.
It is not surprising that more than 30 foreign and several Hungarian
studies have shown a connection between the socio-cultural status
and mortality [49-52]. A similar association with respect to malignant
diseases has also been shown [53]. According to a Swedish survey
published in 2009 unfavourable effect of deprived social conditions
on tumour-related mortality continues to increase [54]. In 1997 the
International Agency for Research on Cancer (IARC, Lyon, France)
collected data on cancer-related mortality from 20 countries and
analyzed them according to socioeconomic status. The resulting
report revealed that the relative risk of tumour-related death in the
lowest income class was significantly higher than that of the highest
social class for both men and women [54].
In less favourable social conditions the diagnosis is often delayed
by lower education and limited access to health-care so malignant
diseases often get recognized in an advanced stage. Infections occur
with a higher rate, patients show decreased resistance, release after
intensive therapy is hampered by poor conditions seen in patients’
home, compliance is not optimal [55,56]. In consequence longer
hospitalization is needed and complication rate is higher. Increasing
poverty due to climate change is expected to lead to a less favourable
outcome of childhood malignant diseases.
Table II
Figure 1
Discussion
We can state that the currently increasing harmful effects of
climate change will cause unfavourable changes within the healthcare
system and therefore within paediatric oncology.
1. Current relationship of certain tumours to geographic
regions, as well as the relative occurrence of these tumours, is changing.
2. The numbers of cases of childhood malignant diseases are
expected to increase.
3. As a result of migration from economically undeveloped
countries, more foreign patients are expected in the developed world
and will challenge health care system.
What can be done to prevent or decrease the effects of
climate change?
The basic questions are:
- How can we get prepared for these changes?
- What can we do to prevent and/or diminish these negative
effects?
The most important step would consist of stopping the main
cause of the process – decrease gas emissions – that lead to climate
change and are in direct association with impoverishment of the
developing world, increase in number of starving people, and number
of early deaths. This is an issue of social attitude, of socio-political
decision-making. However, there is need for physicians’ help in
drawing attention to the serious health-related consequences of
climate change and to the fact that this may concern not only those
living in tropical areas and the developing world but will inevitably
have a global effect, concerning the developed world, too.
An imposing task will be to expand the medical university
curriculum to include the presentation of health effects of climate
change and related actions of health-care system that will be required
[57]. University undergraduate and post-graduate courses should
include special types of childhood malignant diseases that so far
occurred mostly in the tropical areas. This is prerequisite not only
in preparation for the expected effects, but also for informing the
society about the health effects of climate change. Further studies,
publications, and conferences dealing with climate change and its
challenges for health-care system, as well as implications for oncology,
may also help in drawing attention to this problem and may shape the
social context [34].
Supporting the developing world’s health-care system, partly
by voluntary personnel participating in education of students
who come from the developing countries to the universities of the
developed world may help a great deal in eliminating climate changerelated
hazards, as well as in preparation for them [58]. A significant
decrease in malignant diseases caused by hepatitis B and AIDS
could be achieved in the developing countries by the introduction
of systematic screening and vaccination of pregnant women and
newborns. A successful model is provided by Taiwan, where
incidence of hepatocellular carcinoma of between 6 and 9 years of
age was reported in 1997 to decrease from 0.52 per 100,000 to 0.13
per 100,000 after the introduction of hepatitis B vaccination [15]. The
treatment of HIV-positive mothers during pregnancy could decrease
congenital AIDS disease of newborns from the current 20 to 30% to
a level of 1 to 2%.
Due to significantly increased migration to the economically
developed countries, we will have to be able to treat more children
suffering oncologic diseases arriving from tropical or Far Eastern
countries. This will be complicated by the fact that often neither the
children nor the parents will be able to speak the local language. This
may create yet another problem: during the healing process it is very
important to establish a good physician-child and physician-parent
relationships which requires mutual understanding. Without this,
neither history-taking, nor explanation of treatment and obtaining
consent can be accomplished efficiently, and any further discussion
of the problems arising on both sides becomes impossible. Access to
help of interpreters and presentation of written material may solve
this problem. In order to gain trust – without which there is no
healing – we have to understand each other, and we have to respect
patients’ cultural and religious attitudes [37]. In different cultures
non-verbal communication may be different. We have to struggle
with this problem, which is globally affecting the developed world, by
providing special training on the importance and methods of a transcultural
approach to the staff of health-care system. We have to be
aware that it takes time to adapt this attitude [37,59]. Multiple studies
should be conducted to deal with this problem, and ways of solution
of arising difficulties should be presented during special training for
nurses and physicians. We must not forget that discrimination on
racial or ethnic basis has not disappeared completely, and our society
has not been able to eliminate it. We must see to it that it does not
have any effect in the health-care system.
Conclusion
In summary, the increasing health-related effects of climate change may pose problems and challenges for the health-care system – including the important area of paediatric oncology – that must be dealt with as soon as possible.
References
- Lim V, Stubbs JW, Nahar N, Amarasena N, Chaudry ZU, Weng SC, et al. Politicians must heed health effects of climate change. Lancet. 2009; 374: 973.
- Piotrowicz K. The occurrence of unfavourable thermal conditions on human health in central europe and potential climate change impacts: an example from Cracow, Poland. Environ Manage. 2009; 44: 766-775.
- Boyle P and Levin B. Cancer In Children - Cancer Site by Site. World Cancer Report Lyon. WHO-IARC. 2008; 482-486.
- Kim KH, Kabir E, Ara Jahan S. A review of the consequences of global climate change on human health. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2014; 32: 299-318.
- Gibbons ED. Climate change, children’s rights, and the pursuit of intergenerational climate justice. 2014.
- Rylander C, Odland JO, Sandanger TM. Climate change and the potential effects on maternal and pregnancy outcomes: an assessment of the most vulnerable – the mother, fetus, and newborn child. Glob Health Action. 2013; 6: 19538.
- ISSOP e-Bulletin No10, September Climate justice and the right to health. 2014.
- Pizzo PA, Poplack DG, eds. Principles and practice of pediatric oncology. Philadelphia, PA; Lippincott Williams & Wilkins. 2011.
- Kliegman RM, Behrman RE, Jenson HB, Stanton BF, eds. Nelson Book of Pediatrics. 18th ed. Philadelphia, PA; Saunders Elsevier. 2007.
- Stiller CA. International variations in the incidence of childhood carcinomas. Cancer Epidemiol Biomarkers Prev. 1994; 3: 305-310.
- Chang CM, Yu KJ, Mbulaiteye SM, Hildesheim A, Bhatia K. The extent of genetic diversity of Epstein-Barr virus and its geographic and disease patterns: a need for reappraisal. Virus Res. 2009; 143: 209-221.
- Athale UH, Patil PS, Chintu C, Elem B. Influence of HIV epidemic on the incidence of Kaposi's sarcoma in Zambian children. J Acquir Immune Defic Syndr Hum Retrovirol. 1995; 8: 96-100.
- Serraino D, Franceschi S. Kaposi's sarcoma and non-Hodgkin's lymphomas in children and adolescents with AIDS. AIDS. 1996; 10: 643-647.
- Uchimaru K, Nakamura Y, Tojo A, Watanabe T, Yamaguchi K. Factors predisposing to HTLV-1 infection in residents of the greater Tokyo area. Int J Hematol. 2008; 88: 565-570.
- Chang MH, Chen CJ, Lai MS, Hsu HM, Wu TC, Kong MS, et al. Universal hepatitis B vaccination in Taiwan and the incidence of hepatocellular carcinoma in children. Taiwan Childhood Hepatoma Study Group. N Engl J Med. 1997; 336: 1855-1859.
- Magrath I, Jain V, Bhatia K. Epstein-Barr virus and Burkitt’s lymphoma. Semin Cancer Biol. 1992; 3: 285-295.
- Leuschner I, Harms D, Schmindt D. The association of hepatocellular carcinoma in childhood with hepatitis B virus infection. Cancer. 1988; 62: 2363-2369.
- Chen WJ, Lee JC, Hung WT. Primary malignant tumour of liver in infants and children in Taiwan. J Pediatr Surg. 1988; 23: 457-461.
- de Vries E, Steliarova-Foucher E, Spatz A, Ardanaz E, Eggermont AM, Coebergh JW. Skin cancer incidence and survival in European children and adolescents (1978-1997). Report from the Automated Childhood Cancer Information System project. Eur J Cancer. 2008; 42: 2170-2182.
- Iwanaga M, Chiyoda S, Kusaba E, Kamihira S. Trends in the seroprevelence of HTLV-1 in Japanese blood donors in Nagasaki Prefecture. Int J Hematol. 2009; 90: 186-190.
- Parkin DM, Stiller CA, Draper GJ, Bieber CA. The international incidence of childhood cancer. Int J Cancer. 1988; 42: 511-520.
- Stiller CA, Parkin DM. International variations in the incidence of childhood renal tumours. Br J Cancer. 1990; 62: 1026-1030.
- Mutalima N, Molyneux EM, Johnson WT, Jaffe HW, Kamiza S, Borgstein E, et al. Impact of infection with human immunodeficiency virus-1 (HIV) on the risk of cancer among children in Malawi – preliminary findings. Infect Agent Cancer. 2010; 5: 5.
- Winter H, Cheng KK, Cummins C, Maric R, Silcocks P, Varghese C. Cancer incidence in the South Asian population of England. Br J Cancer. 1999; 79: 645-654.
- Stiller CA, McKinney PA, Bunch KJ, Bailey CC, Lewis IJ. Childhood cancer and etnic group in Britain: a United Kingdom Children’s Cancer Study Group (UKCCSG) Study. Br J Cancer. 1991; 64: 543-548.
- Eltom MA, Jemal A, Mbulaiteye SM, Devesa SS, Biggar RJ. Trends in Kaposi’s sarcoma and non-Hodgkin’s lymphoma incidence in the United States from through. J Nat Cancer Inst. 2002; 94: 1204-1210.
- Franchini G. Molecular mechanisms of Human T-cell leukaemia/lymphotropic virus type I infection. Blood. 1995; 86: 3619-3639.
- Davidson A. Kaposi sarcoma: The African HIV epidemic's partner in crime. Pediatr Blood Cancer. 2010; 54: 670-674.
- Qu C, Chen T, Fan C, Zhan Q, Wang Y, Lu J, et al. Efficacy of neonatal HBV vaccination on liver cancer and other liver diseases over 30-year follow-up of the Qidong hepatitis B intervention study: a cluster randomized controlled trial. PLoS Med. 2014; 11.
- Krisberg K. Climate change predicted to have direct effects on health: experts urge action now to prevent deaths. Nations Health. 2007; 37.
- McMichael AJ, Friel S, Nyong A, Corvalan C. Global environmental change and health: impacts, inequalities, and the health sector. BMJ. 2008; 336: 191-194.
- Kistin EJ, Fogarty J, Pokrasso RS, McCally M, McCornik PG. Climate change, water resources and child health. Arch Dis Child. 2010; 95: 545-549.
- Thompson B, Cohen MJ editors: The impact of climate change and bioenergy on nutrition. FAO Springer. 2012.
- Costello A, Abbas M, Allen A, Ball S, Bell S, Bellamy R, et al. Managing the health effects of climate change: Lancet and University College London Institute for Global Health Commission. Lancet. 2009; 373: 1693-1733.
- Ford TE, Colwell RR, Rose JB, Morse SS, Rogers DJ, Yates TL. Using satellite images of environmental changes to predict infectious disease outbreaks. Emerg Infect Dis. 2009; 15: 1341-1346.
- McKie R. Children will suffer most as climate change increases in coming decades, say scientists. BMJ. 2013; 347: 5799.
- Pergert P, Ekblad S, Enskar K, Bjork O. Bridging obstacles to transcultural caring relationships--tools discovered through interviews with staff in paediatric oncology care. Eur J Oncol Nurs. 2008; 12: 35-43.
- Bharath AK, Turner RJ. Impact of climate change on skin cancer. J R Soc Med. 2009; 102: 215-218.
- Schulte PA, Chun H. Climate change and occupational safety and health: establishing a preliminary framework. J Occup Environ Hyg. 2009; 6: 542-554.
- Autier P, Doré JF, Gefeller O, Cesarini JP, Lejeune F, Koelmel KF, et al. for the EORTC Melanoma Co-operative Group. Melanoma risk and residence in sunny areas. Bri J Cancer. 1997; 76: 1521-1524.
- Oliveria SA, Saraiya M, Geller AC, Heneghan MK, Jorgensen C. Sun exposure and risk of melanoma. Arch Dis Child. 2006; 91: 131-138.
- Bunyavanich S, Landrigan CP, McMichael AJ, Epstein, PR. The impact of climate change on child health. Ambulatory Pediatrics. 2003; 3: 44-52.
- Adelman AS, McLaughlin CC, Wu XC, Chen VW, Groves FD. Urbanisation and incidence of acute lymphocytic leukaemia among United States children aged 0-4. Br J Cancer. 2005; 92: 2084-2088.
- Kinlen LJ, Dickson M, Stiller CA. Childhood leukaemia and non-Hodgkin's lymphoma near large rural construction sites, with a comparison with Sellafield nuclear site. BMJ. 1995; 310: 763-768.
- Heath CW Jr. Community clusters of childhood leukemia and lymphoma: evidence of infection? Am J Epidemiol. 2005; 162: 817-822.
- Nyári TA, Kajtár P, Bartyik K, Thurzó L, Parker L. Childhood acute lymphoblastic leukaemia in relation to population mixing around the time of birth in South Hungary. Pediatr Blood Cancer. 2006; 47: 944-948.
- Bellec S, Baccaini B, Goubin A, Rudant J, Ripert M, Hemon D, et al. Childhood leukaemia and population movements in France, 1990-2003. Br J Cancer. 2008; 98: 225-231.
- Rudant J, Baccaini B, Ripert M, Goubin A, Bellec S, Hemon D, et al. Population-mixing at the place of residence at the time of birth and incidence of childhood leukaemia in France. Eur J Cancer. 2006; 42: 927-933.
- Bakacs M, Vitrai J. How do social-economic differences in residential characteristics affect mortality? A literature review. Orv Hetil. 2008; 149: 1317-1321.
- Skrabski A, Kopp M, Kawachi I. Social capital and collective efficacy in Hungary: cross sectional associations with middle aged female and male mortality rates. J Epidemiol Community Health. 2004; 58: 340-345.
- Kopp MS, Skrabski A, Kawachi I, Adler NE. Low socioeconomic status of the opposite sex is a risk factor for middle aged mortality. J Epidemiol Community Health. 2005; 59: 675-678.
- Pickett KE, Pearl M. Multilevel analyses of neighbourhood socioeconomic context and health outcomes: a critical review. J Epidemiol Community Health. 2001; 5: 111-122.
- Kogevinas M, Pearce N, Susser M, Bolfetta P, editors. Social Inequalities and Cancer. IARC Scientific Publications No. 1997; 138.
- Kristinsson SY, Derolf AR, Edgren G, Dickman PW, Bjorkholm M. Socioeconomic differences in patient survival are increasing for acute myeloid leukemia and multiple myeloma in Sweden. J Clin Oncol. 2009; 27: 2073-2080.
- Magnani C, Pastore G, Coebergh JW, Viscomi S, Spix C, Steliarova-Foucher E. Trends in survival after childhood cancer in Europe: report from the Automated Childhood Cancer Information System project (ACCIS). Eur J Cancer. 2006; 42: 1981-2005.
- Sankila R, Martos Jimenez MC, Miljus D, Pritchard-Jones K, Steliarova-Foucher E, Stiller C. Geographical comparison of cancer survival in European children (1988-1997): report from the Automated Childhood Cancer Information System project. Eur J Cancer. 2006; 42: 1972-1980.
- Hess JJ, Heilpern KL, Davis TE, Frumkin H. Climate change and emergency medicine: impacts and opportunities. Acad Emerg Med. 2009; 16: 782-794.
- Apjok E, Borsi JD, Koos R, Schuler D, Bolinger RE, Boysen CD, et al. Pediatric oncology outreach to Hungary: a registry of childhood malignancies. Ann N Y Acad Sci. 1997; 824: 232-233.
- Smith AK, Sudore RL, Perez-Stable EJ. Palliative care for Latino patients and their families: whenever we prayed, she wept. JAMA. 2009; 301: 1047-1057.