Research Article
Clinical Prognostic Factors are better Outcome Predictors than Molecular Factors in Glioblastoma
Samprón N1,2,3,4*, Undabeitia J1,3, Torres-Bayona S1, Aldaz P4, Carrasco-Garcia E4, Arrazola
M1,3, Villanua J3,4,5, Egaña L3,4,6, Caballero MC3,7, Etxegoien I3,8, Bollar A1,2,3, Querejeta A3,9, Armendáriz M1,3, Torres P1,3, Pardo E3,8, Liceaga G3,10, Urtasun M3,11, Ruiz I3,4,7, Matheu A3,4 and Urculo E1,3
1Department of Neurosurgery, Hospital Universitario Donostia, Spain
2Instituto Oncológico, Spain
3Neurooncology Tumor Board, Hospital Universitario Donostia, Spain
4Cellular Oncology Research Group, Spain
5Institute of Radiology, Spain
6Department of Medical Oncology, Hospital Universitario Donostia, Spain
7Department of Pathology, Hospital Universitario Donostia, Spain
8Department of Radiology, Hospital Universitario Donostia, Spain
9Department of Radiation Oncology, Hospital Universitario Donostia, Spain
10Department of Pharmacology, Hospital Universitario Donostia, Spain
11Department of Neurology, Hospital Universitario Donostia, Spain
*Corresponding author: Nicolás Samprón, Department of Neurosurgery, Hospital Universitario Donostia, Paseo Dr. Beguiristain 117 20080, San Sebastian, Spain
Published: 05 Jan, 2017
Cite this article as: Samprón N, Undabeitia J, Torres-
Bayona S, Aldaz P, Carrasco-Garcia E,
Arrazola M, et al. Clinical Prognostic
Factors are better Outcome Predictors
than Molecular Factors in Glioblastoma.
Clin Oncol. 2017; 2: 1178.
Abstract
The World Health Organization (WHO) classification of central nervous system tumors 2016
classifies glioblastoma in the highest grade of aggressiveness. Nowadays, all the treatment for this
type of tumor remains palliative. Surgical resection is the primary treatment. Radiotherapy and
chemotherapy have been shown to prolong survival, although most patients die within two years.
Due to the poor prognosis and discreet effectiveness of the treatment, the patients’ quality of life
throughout the whole process should be the main concern. Over the last 15 years, several clinical
and molecular prognostic markers have been identified, but their impact varies within different
studies. In this work, we have done a follow-up of a clinical cohort of patients with glioblastoma and
have identified that age, functional status and number of surgeries is the main predictors of survival.
Keywords: Glioblastoma; Oncology; Survival; Temozolomide; Neurosurgery; Clinical information; Prognostic factors
Introduction
Glioblastoma is a form of brain cancer that belongs to the astrocytic lineage and may arise from
mature glial cells, precursor cells or neural stem cells. Among the over a hundred different types
of neoplasms that human being are prone to develop, glioblastoma is one of the most aggressive
and shows high resistance to multimodal treatment [1]. It remains an incurable disease with poor
prognosis; half of the patient will die in about a year, most of the remaining in the second year and
less than 5% will eventually make it to 5 years [2]. These are the existing figures in high resources
clinical setting despite rapid access to the current, costly, aggressive, non-inocuos state of the art
treatment which includes maximal safe surgical resection and concomitant and adjuvant radio and
chemotherapy [3,4]. On the other hand, it is estimated that patients left untreated have an average
mean survival of only 3 months [5].
Glioblastoma should be considered a special type of cancer that usually affects since the outset
the intellectual, cognitive, behavioral, speech and physical functions, tackling not only the extension
of survival but also the quality of life [6]. The disease progresses and the patients gradually and
restlessly lose their neurological functions and independence, the ability of self-care, and need
assistance thus transforming the patients and their relatives lives. Death occurs at the end of a
ruthless process of neurological deterioration [7].
The failure of the current approach to treatment, surgery plus radiation plus alkylating agents, occurs in every single case, hence reflecting the particularities of the
brain as an organ that is isolated from the external environment,
as well as the complex biological nature of the tumor. Whereas
in many types of neoplasms there were important advances in
research, treatment, and outcome, the outcome of patients who
bear a glioblastoma does not seem to have varied significantly in
the past decades [8]. Nevertheless, over the last ten years or so, an
inconspicuous extension of the overall survival has been reported in
some clinical series and in clinical trials [9,10].
Meanwhile, the incorporation into clinical practice of multimodal
and readily available imaging in diagnostic of brain tumors as well as
complementary tolls for diagnosis and therapeutic planning, steroid
administration, refinement of anesthetic technique, postoperative
care, the development of microneurosurgery and fluorescents
guided tumor resection, the introduction of intraoperative imaging,
neuronavigation and brain mapping techniques have managed to
shorten the time between the first symptoms and the diagnosis and
to decrease preoperative morbidity and mortality, thus, improving
quality of life [9,11,12].
Over the last 15 years, several studies have shown that
chemotherapy with temozolamide (an alkylating oral administrated
drug) is effective to increase both progression free and overall survival
[10]. It has also been observed that epigenetic data obtained from
tumor tissue, particularly the methylation of the promoter of MGMT,
a DNA repairing gen, could predict which patient will benefit the
most, although it is also recognized that this indicator is not strong
enough to preclude the use of this non-inocuos treatment in the
remaining patients [3,4,10,13,14]. However, the mean survival time
does not seem to have varied significantly and remains approximately
in 15 months in clinical series while in population based studies or in
uninsured patients, this survival drops significantly [15,16].
Clinical observation suggested significant inter-individual
differences in survival that can be only partially explained by clinical
criteria such as age, performance status, cognitive impairment, and
location [9,17].These variations could be attributed to the particular
biological behavior of a given tumor and/or to host response. The
term multiforme, now deleted from the WHO classification of tumors
of the central nervous system, was initially used by the pioneer
pathologist and neurosurgeons to describe the morphological
heterogeneity of the tumor, but it could be applied with the same
precision to its molecular aberrations and clinical impact [1].
Glioblastoma resists treatment attempts owing to its biological
characteristics that should be understood from the systemic, tissular,
cellular and molecular perspectives. Current research techniques,
especially large-scale multidimensional studies on the genome, epigenome,
transcriptome and proteome, are promising tools to uncover
the process of gliomagenesis as a necessary background to design
rational therapeutic strategies and truly effective solutions [18].
This oncogenic process, that involves a set of molecular alterations
associated with differentiation, proliferation control, growth, cell
death and the cell cycle, has been understood as the anomalous
implementation of physiological programs of development. The
identification of molecular markers and pathways associated with
these alterations can explain the otherwise unexplained differences
between patients´ outcomes and responses to treatment [18,19].
In this context, we have set out to analyze the survival of a
cohort of glioblastoma patients in actual clinical practice after the
incorporation of new treatment protocols in order to learn about its
impact on survival. We also analyzed survival in the light of clinical
prognostic factors.
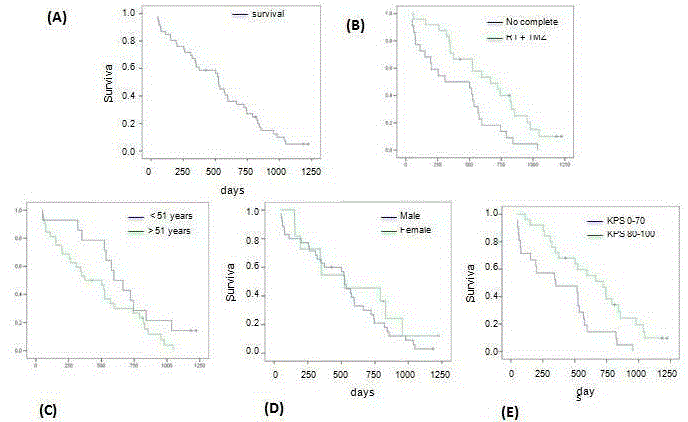
Figure 1
Figure 1
Evaluation of clinical prognostic factors in glioblastoma.
A) Analysis of the survival according to the Kaplan-Meier method corresponding to the 46 patients included in the final analysis. The patients who were alive at the
end of the analysis or “censored” were marked with a cross. B) Comparative analysis of the survival of the patients who received the indicated treatment protocol
and those who did not receive it. RT = radiotherapy. TMZ = temozolomide concomitant and adjuvant. C) Comparative analysis of the survival of patients older and
younger than 51 years old. D) Analysis of the survival according to sex. E) Analysis of the survival of the patients with a Karnofsky Performance Status Scale score of 80% or higher compared with 70% or lower.
Materials and Methods
Patient and data collection
The patients who were included in the study were of any age
and gender with an anatomopathological diagnosis of glioblastoma
- WHO grade IV astrocytoma [20], and have undergone surgical
treatment or biopsy in Donostia Hospital of San Sebastian.
A total of 46 patients were included in the final analysis of the
study. For data collection, we used the hospital database records
of the patients diagnosed with glioblastoma. Subsequently, we
proceeded to systematically review the medical records, including the
neuroimaging studies. We checked that the biopsy material available
was adequate for molecular studies and for diagnostic confirmation.
Finally, with the clinical and molecular information, we made a
new database that served as a source for statistical studies. The
clinical management of the patients included in this study was done
according to the multidisciplinary care model of the Neuro-Oncology
Committee of Donostia Hospital of San Sebastian.
Statistical analysis
The date of the first surgical intervention, whose results resulted
in the diagnosis of glioblastoma (diagnostic surgery), was considered
the origin, or time at which the individual was included in the analysis.
The end point of the analysis was the date of death. The end point
of the analysis was mortality from any cause. The survival associated
with each variable was compared by Kaplan-Meier. The analysis was
performed using the SPSS statistical package.
Histopathological study
The material for the histological study was obtained from
representative sections of the tumor after making 5-μm slices of
the study material, dewaxing and staining with hematoxylin and
eosin. The samples were reviewed by a neuropathologist to confirm
the diagnosis and select the most representative area of the sample,
avoiding areas of necrosis and artifacts.
The material for the immunohistochemical study of proliferation
was obtained from representative sections of the tumor areas after
making 5-μm slices and staining with monoclonal antibody for the
KI-67 marker [21].
Analysis of the MGMT gene promoter methylation
Firstly, 10-μm slides were obtained from the paraffin blocks. The
methylation analysis was carried out by treating the samples with
hydroquinone sodium bisulfite and then methylation-specific PCR
amplification. For quality control of the procedure, we did a PCR
with specific primers for the TERF gene (assuming that this gene is
always methylated), so that DNA amplification only occurred if the
methylation treatment had been done correctly [13].
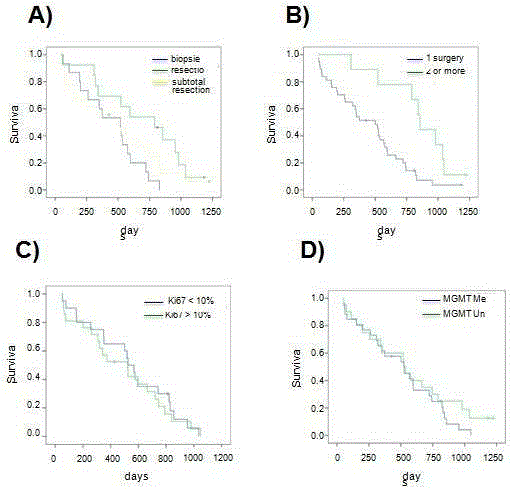
Figure 2
Figure 2
Evaluation of surgery and molecular prognostic factors in glioblastoma.
A) Comparative analysis of the survival of the patients who underwent complete macroscopic surgical resection, subtotal resection or biopsy according to the
Kaplan-Meier method. B) Analysis of the survival of patients who underwent one tumor resection surgery in comparison with those who had surgery two or more
times. C) Analysis of the survival of the patients in which positivity for KI-67 was found in 10% or more of the nuclei or less compared with the rest. D) Analysis of the survival according to the state of MGMT gene promoter methylation.
Results
Evaluation of outcome and clinical prognostic factors in
glioblastoma
The median overall survival of the sample was 525 days (17.5
months) (standard error 36.6; 95% confidence interval 453-597)
ranging between 48 and 1223 days. Fifteen (32.6%) of the 46 patients
survived over two years, and two of them (4.3%) survived 3 or more
years (Figure 1A). Of the 46 patients included in the final analysis, 24
(52%) received the complete postoperative treatment protocol with
radiotherapy in doses equal to or over 54 Gy and concomitant and
adjuvant temozolomide. The median survival of the patients who
received this protocol was 665 days (22.2 months) (standard error
153; 95% confidence interval 364-966), whereas it was 308 days (10.3
months) (standard error 186; 95% confidence interval 0-672) in those
that, for some reason, did not receive the complete treatment protocol
(Figure 1B). This difference was statistically significant (Mantel-Cox
test p = 0.008).
The mean age of the patients included in the study was 54.15
years old (median 56.5), ranging between 7 years old for the youngest
patient and 80 years old. The cut-off point arbitrarily selected to
analyze the impact of age on survival was 51 years old. Of the total
number of patients, 14 (30%) were 51 years old or less at the time of the first surgery and 32 (70%) were older than 51. The median
survival of the patients over 50 years old was 374 days (12.5 months)
(standard error 120; 95% confidence interval 138-610), whereas it
was 597 days (19.9 months) (standard error 83; 95% confidence
interval 432-758) in those under that age. This difference, although
it showed a clear tendency in favor of the younger patients, did not
reach statistical significance (Mantel-Cox test p = 0.087) (Figure 1C).
Of the 46 patients, 35 (76%) were men and 11 (24%) were women.
The male to female ratio was 3.2. The median survival of the men was
526 days (17.5 months) (standard error 25; 95% confidence interval
477-575), whereas it was 523 days (17.4 months) (standard error 247;
95% confidence interval 38-1007) in the women. This difference did
not reach statistical significance (Mantel-Cox test p = 0.42) (Figure
1D). Finally, twenty-five patients (54%) of the 46 analyzed had a score
on the Karnofsky Performance Status (KPS) scale of 80% or higher,
whereas the other 21 cases (46%) scored 70% or less. This division was
based on previous studies [22]. The median survival of the patients
with a KPS of 80% or higher was 722 days (24 months) (standard
error 117; 95% confidence interval 492-952), whereas it was 351 days
(11.7 months) (standard error 242; 95% confidence interval 0-825)
in the patients with a lower KPS. This difference was statistically
significant (Mantel-Cox test p = 0.004) (Figure 1E).
Evaluation of surgery-related factors in glioblastoma
Fifteen patients (33%) of the total number underwent a stereotactic
biopsy. In 13 cases (28%) there was a complete macroscopic resection
confirmed with a magnetic resonance study with contrast obtained
within the first 72 postoperative hours. In the other 18 patients
(39%), the resection was subtotal or total without being able to be
confirmed by neuroimaging studies. The median survival of those in
which a complete macroscopic resection was achieved was 791 days
(26.4 months) (standard error 187; 95% confidence interval 424-
1157), whereas it was 501 days (16.7 months) (standard error 171;
95% confidence interval 166-836) in those in which the resection
was subtotal, and 518 days (17.3 months) (standard error 112; 95%
confidence interval 298-738) in those who had a biopsy (Figure 2A).
These differences did not reach statistical significance (Mantel-Cox
test p = 0.08) (Figure Comparing the survival of the patients who
underwent complete macroscopic resection with the rest of the
patients, there was statistical significance (791 vs. 518 days) (Mantel-
Cox test = 0.05) (Figure 2A). Finally, 9 of the 46 patients (20%)
underwent more than one surgical intervention for the removal of the
tumor, one of which had surgery on three occasions. This last group
of patients, who had 2 or more surgical interventions for the tumor
removal, showed an advantage in survival in comparison with the rest
that was statistically significant (501 vs. 856 days) (Mantel-Cox test p
= 0.005) (Figure 2B).
Evaluation of molecular prognostic factors in glioblastoma
The results of the immunohistochemical analysis of cell
proliferation, based on the monoclonal antibody KI-67, were available
in 41 of the 46 cases included in the final analysis (89%). Positivity for
the marker in 10% of the nuclei was arbitrarily considered the cut-off
point for the analysis. Twenty patients (49%) were positive for KI-
67 in values less or equal to 10%, and the median survival of these
patients was 526 days (standard error 56; 95% confidence interval
416-635). In the other 21 patients, positivity for KI-67 was considered
positive in more than 10% of the nuclei, and the median survival
in these patients was 523 days (standard error 132; 95% confidence
interval 264-781). This difference did not reach statistical significance
(Mantel-Cox test p = 0.77) (Figure 2C).
The results of the MGMT gene promoter methylation analysis
were available in all 46 patients. Twenty-six samples (57%) were
positive for MGMT gene promoter methylation, whereas 20 (43%)
were negative. The median survival of the patients in which the
MGMT gene promoter was methylated was 525 days (17.5 months)
(standard error 117; 95% confidence interval 297-553), whereas it was
523 days (17.4 months) (standard error 20; 95% confidence interval
484-562) in the patients in which the MGMT gene promoter was
not methylated (Figure 2D). This difference did not reach statistical
significance (Mantel-Cox test p = 0.32).
Discussion
Currently, the diagnosis of glioblastoma continues to be
associated with a poor prognosis that, despite the development of all
the disciplines related to neuro-oncology, has not varied significantly
in the last decades, and has remained around the 12 months for
decades. The median survival of the patients who underwent
the treatment that is currently considered optimum - that is, the
maximum possible surgical resection without causing deficit, plus
radiotherapy and chemotherapy - was 14.6 months and only 26.5%
of the patients survived longer than two years, of which 10.7% were
free of progression at that time[10]. This study, and other similar ones
including ours, demonstrated the moderate effectiveness of the tested
therapeutic measures, although they revealed certain variability in the
response.
In our study, the observed median survival (17.5 months) was
higher than that expected considering that age, functional status and
comorbidities were not exclusion criteria (Figure 2). These results can
be explained, at least in part, by methodological reasons. Indeed, it is
possible that there were certain inaccuracies due to the sample size, to
the high percentage of exclusion for lack of material and exclusion of
those who passed away during the first 30 days of the postoperative
period; however, the vast majority of the exclusions were due to causes
typical of retrospective studies (lack or deterioration of samples and
of data). Besides the methodological reasons, other factors that can
explain this advantage in the survival are: real effectiveness of the
treatment (which includes, in our study, an active multidisciplinary
approach), the high proportion of resurgery (20%), the use of salvage
chemotherapy - especially antiangiogenic drugs and topoisomerase
inhibitors - and the characteristics of the palliative and end of life
care.
Undoubtedly, a correct comprehension of the factors that
determine the prognosis is important to provide personalized
information to patients and to make therapeutic decisions,
considering their impact on quality of life and cost effectiveness
[23]. Stratification by risk factors (clinical and molecular) is,
likewise, key to the design and interpretation of future clinical trials
and other studies on therapeutic effectiveness. Characterization
of dysfunctional molecular pathways in the tumor cells (in specific
patients and times) is also a fundamental principle in the age of
oncological treatment addressed toward specific molecular structures
[24]. Finally, the point of view of prognostic factors puts the analysis
of survival in the individual’s perspective by providing a tool to
adapt and to improve diverse aspects of the clinical management
of patients. Gorlia et al. [17] in an exploratory study on patients
from a clinical trial, determined that the most relevant factors that
independently conditioned the prognosis were age, functional status
(e.g. Karnofsky Index), cognitive impairment, volume of surgical
resection, radiotherapy, administration of temozolomide, MGMT gene promoter methylation in the tumor tissue and the need for
corticosteroids in the postoperative period [13,17]. In our study,
we confirmed the impact that some of these factors have on the
survival of the patients. In particular, we observed that age of the
patient, functional status, volume of surgical resection and number of
surgeries, are the most relevant prognostic factors.
Some authors, based on the contrasted molecular heterogeneity
of glioblastoma, have proposed molecular prognostic factors to
increase the precision of predictions based on pre- and posttreatment
clinical factors. The molecular markers researched in
glioblastoma include KI-67, MGMT, EGFR, PDGFR, p53 and PTEN
[13,21,25]. The KI-67 marker that we have analyzed in this study has
been proposed as a predictor of clinical evolution in patients with
glioblastoma, hypothesizing that tumors with a greater proportion
of cells in division (situation that reveals the expression of this
indicator) are more aggressive neoplasms and determine a poorer
prognosis [26,27]. However, this hypothesis has not been confirmed
in our study and the analysis of the positivity of KI-67 in 10% or
more of the cell nuclei could not discriminate groups of patients
with a different clinical evolution. Our results are in agreement with
additional studies [28]. The expression of the MGMT gene is the main
mechanism of resistance of tumors to alkylating agents by correcting
the DNA damage induced by this type of drug. It has been observed
that this marker indicates a favorable prognosis while predicting the
response to temozolomide in different studies [4,13,29]. Surprisingly,
in our study, however, the MGMT gene promoter methylation state
did not predict the clinical evolution when the complete sample
was analyzed, and it was not a predictor when the patients who had
received temozolomide or those who had received the complete
postoperative treatment protocol were analyzed individually. The
interpretation of these results does not seem simple and previous
stratification is needed based on the clinical factors mentioned to
extract certain working hypotheses more than conclusive information
[21,30]. Furthermore, the different studies that include an analysis
of molecular markers associated with clinical evolution are difficult
to compare for various reasons. Some of these studies are based on
samples that include other lower-grade gliomas, the techniques used
for the analysis of the markers are not the same (for example FISH vs.
PCR in the analysis of MGMT gene promoter methylation) and some
were carried out in clinical trials whereas others were not, so the type
of treatment the patients underwent can vary significantly [30].
Overall, our study seems to indicate that there is a more reliable
relationship between individual clinical markers than molecular
markers (at least in regard to KI-67 and MGMT promoter
methylation) and the clinical evolution of patients with glioblastoma
multiforme.
Acknowledgements
Paula Aldaz is recipient of a predoctoral fellowship from the AECC. AM is granted with an Ikerbasque Research Fellow position. This work was supported by grants from the Department of Industry of the Basque Government (12-PE12BNO13 to NS), Kutxabank to NS.
References
- Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, et al. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol. 2016; 131: 803-820.
- Ostrom QT, Bauchet L, Davis FG, Deltour I, Fisher JL, Langer CE, et al. The epidemiology of glioma in adults: a "state of the science" review. Neuro Oncol. 2014; 16: 896-913.
- Hottinger AF, Stupp R, Homicsko K. Standards of care and novel approaches in the management of glioblastoma multiforme. Chin J Cancer. 2014; 33: 32-39.
- Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009; 10: 459-466.
- Malmstrom A, Gronberg BH, Marosi C, Stupp R, Frappaz D, Schultz H, et al. Temozolomide versus standard 6-week radiotherapy versus hypofractionated radiotherapy in patients older than 60 years with glioblastoma: the Nordic randomised, phase 3 trial. Lancet Oncol. 2012; 13: 916-926.
- Bampoe J, Ritvo P, Bernstein M. Quality of life in patients with brain tumor: what's relevant in our quest for therapeutic efficacy? Neurosurg Focus. 1998; 4: e6.
- Thier K, Calabek B, Tinchon A, Grisold W, Oberndorfer S. The Last 10 Days of Patients With Glioblastoma: Assessment of Clinical Signs and Symptoms as well as Treatment. Am J Hosp Palliat Care. 2016; 33: 985-988.
- deSouza RM, Shaweis H, Han C, Sivasubramiam V, Brazil L, Beaney R, et al. Has the survival of patients with glioblastoma changed over the years? Br J Cancer. 2016; 114: 146-150.
- Thakkar JP, Dolecek TA, Horbinski C, Ostrom QT, Lightner DD, Barnholtz-Sloan JS, et al. Epidemiologic and molecular prognostic review of glioblastoma. Cancer Epidemiol Biomarkers Prev. 2014; 23: 1985-1996.
- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005; 352: 987-996.
- Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ, et al. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol. 2006; 7: 392-401.
- Yasargil MG, Kadri PA, Yasargil DC. Microsurgery for malignant gliomas. J Neurooncol. 2004; 69: 67-81.
- Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005; 352: 997-1003.
- Weller M, Tabatabai G, Kastner B, Felsberg J, Steinbach JP, Wick A, et al. MGMT Promoter Methylation Is a Strong Prognostic Biomarker for Benefit from Dose-Intensified Temozolomide Rechallenge in Progressive Glioblastoma: The DIRECTOR Trial. Clin Cancer Res. 2015; 21: 2057-2064.
- Oertel J, von Buttlar E, Schroeder HW, Gaab MR. Prognosis of gliomas in the 1970s and today. Neurosurg Focus. 2005; 18: e12.
- Kelly PJ. Technology in the resection of gliomas and the definition of madness. J Neurosurg. 2004; 101: 284-286; discussion 286.
- Gorlia T, van den Bent MJ, Hegi ME, Mirimanoff RO, Weller M, Cairncross JG, et al. Nomograms for predicting survival of patients with newly diagnosed glioblastoma: prognostic factor analysis of EORTC and NCIC trial 26981-22981/CE.3. Lancet Oncol. 2008; 9: 29-38.
- Brennan CW, Verhaak RG, McKenna A, Campos B, Noushmehr H, Salama SR, et al. The somatic genomic landscape of glioblastoma. Cell. 2013; 155: 462-477.
- Cancer Genome Atlas Research N. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature. 2008; 455: 1061-1068.
- Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007; 114: 97-109.
- Simmons ML, Lamborn KR, Takahashi M, Chen P, Israel MA, Berger MS, et al. Analysis of complex relationships between age, p53, epidermal growth factor receptor, and survival in glioblastoma patients. Cancer Res. 2001; 61: 1122-1128.
- Lamborn KR, Chang SM, Prados MD. Prognostic factors for survival of patients with glioblastoma: recursive partitioning analysis. Neuro Oncol. 2004; 6: 227-235.
- Rosenberg J. How well are we doing in caring for the patient with primary malignant brain tumor? Are we measuring the outcomes that truly matter? A commentary. Neurosurg Focus. 1998; 4: e5.
- Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell. 2010; 17: 98-110.
- Ohgaki H, Dessen P, Jourde B, Horstmann S, Nishikawa T, Di Patre PL, et al. Genetic pathways to glioblastoma: a population-based study. Cancer Res. 2004; 64: 6892-6899.
- Jaros E, Perry RH, Adam L, Kelly PJ, Crawford PJ, Kalbag RM, et al. Prognostic implications of p53 protein, epidermal growth factor receptor, and Ki-67 labelling in brain tumours. Br J Cancer. 1992; 66: 373-385.
- Barker FG, Prados MD, Chang SM, Davis RL, Gutin PH, Lamborn KR, et al. Bromodeoxyuridine labeling index in glioblastoma multiforme: relation to radiation response, age, and survival. Int J Radiat Oncol Biol Phys. 1996; 34: 803-808.
- Bouvier-Labit C, Chinot O, Ochi C, Gambarelli D, Dufour H, Figarella-Branger D. Prognostic significance of Ki67, p53 and epidermal growth factor receptor immunostaining in human glioblastomas. Neuropathol Appl Neurobiol. 1998; 24: 381-388.
- Esteller M, Garcia-Foncillas J, Andion E, Goodman SN, Hidalgo OF, Vanaclocha V, et al. Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N Engl J Med. 2000; 343: 1350-1354.
- Weller M, Pfister SM, Wick W, Hegi ME, Reifenberger G, Stupp R. Molecular neuro-oncology in clinical practice: a new horizon. Lancet Oncol. 2013; 14: e370-379.