Case Report
Breast Cancer Occurring after Childhood Total Body Irradiation as Conditioning for Allogeneic Stem Cell Transplantation: Case Report and Review of the Literature
Michael A. Savin*
Division of Hematology and Medical Oncology, Knight Cancer Institute, Oregon Health & Science University, USA
*Corresponding author: Michael A. Savin, Division of Hematology and Medical Oncology, Knight Cancer Institute, Oregon Health & Science University, Mail Code: L586, 3181 S.W. Sam Jackson Park Road, Portland, Oregon 97239-3098, USA
Published: 08 Nov, 2016
Cite this article as: Savin MA. Breast Cancer Occurring
after Childhood Total Body Irradiation
as Conditioning for Allogeneic Stem
Cell Transplantation: Case Report and
Review of the Literature. Clin Oncol.
2016; 1: 1134.
Abstract
Exposure to ionizing radiation during childhood and adolescence is well known to increase the risk of later malignancy, including leukemia and a variety of solid cancers. Radiation to the chest or mediastinum during treatment of Hodgkin Lymphoma or metastatic solid cancers in young females is well known to cause a substantial increased risk of later breast cancer. Less well known is the risk of breast cancer arising after Total Body Irradiation (TBI) in this age group as part of conditioning for Hematopoietic Stem Cell Transplantation (HSCT). This report presents a young woman who developed high-grade invasive breast cancer at age 27, sixteen years after childhood treatment for relapsed acute leukemia with TBI conditioned allogeneic stem cell transplantation. Female patients treated in childhood with TBI conditioning for HSCT should be educated about their breast cancer risk, and should have regular surveillance for breast cancer beginning at a much younger age than the general population. The recent finding of two possible genomic variants associated with increased breast cancer risk after childhood radiation may offer a future tool to identify patients who need particularly close surveillance.
Introduction
The association between ionizing radiation exposure and malignancy has been the subject of
investigation for many years, beginning with the long-term surveillance of the survivors of the
Hiroshima and Nagasaki atomic bombs in 1945. Although thousands of people were killed by the
bombs, there were thousands of survivors who were exposed to varying levels of radiation. Many of
them were followed over the decades since the war, revealing much about the short-term and longterm
effects of radiation exposure. Leukemia was the first malignancy observed in survivors, but
over the subsequent years an increased risk of many other types of cancer has been described [1-3].
Many studies of solid tumors in atomic bomb survivors and in others exposed to non-therapeutic
radiation have shown an excess of breast cancer in these individuals [4-7]. Because of the recent
Fukushima Daiichi nuclear power plant accident in Japan and the 1986 Chernobyl accident in
Ukraine, we are learning more about the risk of solid cancers after accidental radiation exposure
[7,8].
Medical radiation for treatment of cancer and non-malignant conditions is also a wellestablished
cause of secondary malignancies, e.g., after radiation treatment of a primary cancer,
or radiation treatment of acne [9-11]. Many factors contribute to breast cancer carcinogenesis
after therapeutic radiation for solid cancers, including radiation dose, age when treated, hormonal
factors, and timing of radiation in relation to sexual development [11]. We have learned much from
studying women treated for Hodgkin Lymphoma with mantle field radiation as children or young
adults [12-15]. We have also learned that treating solid cancers in young girls with whole lung
radiation is another risk factor for breast cancer [12,14].
Although much of our knowledge comes from childhood radiation for solid malignancies, acute
leukemia is the most common childhood cancer. Children with acute leukemia are treated primarily
with chemotherapy, but some undergo allogeneic HSCT for high risk or relapsed disease. A few of
these children receive low dose TBI as part of the HSCT conditioning regimen prior to transplant.
Because it has been extensively studied, the risk of breast cancer after childhood or adolescent
radiation for Hodgkin Lymphoma or solid tumors is widely recognized by physicians. However,
many are unaware of the risk after treatment for childhood acute leukemia, particularly in patients
treated with TBI conditioning for HSCT. Clinicians who are not cognizant of this risk may fail to
consider appropriate surveillance for patients at risk.
The following case report describes a 27 year old woman who
developed breast cancer 16 years after treatment for relapsed acute
myelogenous leukemia with TBI-conditioned allogeneic HSCT. This
report includes a brief literature review of secondary breast cancer in
this clinical setting (Table 1).
Table 1
Table 1
Selected references examining the relationship of childhood radiation exposure from a variety of sources and subsequent development of secondary
malignancies in general and breast cancer in particular. Radiation exposure data analyzed includes atomic bomb survivors, other environmental exposure (e.g., nuclear
accidents), medical/therapeutic exposure, and total body irradiation for stem cell transplantation. Data bases searched include PubMed, online journal archives, other
online bibliographies, and web-based search engines. Reference numbers are in brackets in each element of the table.
Methods
A directed review of relevant literature was performed using several literature search tools, identifying reviews and compilations of patients with data pertinent to the questions raised in this case report. The literature data bases searched include PubMed, online journal archives, other online bibliographies, and web-based search engines.
Case Report
Acute leukemia history
In March, 2015, a 27-year-old gravida 0 para 0 woman was referred
for treatment of newly diagnosed breast cancer. In 1998, at age 10, she
was diagnosed with acute myeloid leukemia (AML - FAB M5a). Her
bone marrow was 100% cellular with 80% blasts, 10% promyelocytes,
10% erythroid cells and scattered megakaryocytes. Flow cytometry
confirmed AML with monocytic differentiation, and cytogenetics
showed a 46, XX, t(9;11)(p22;q23) karyotype in 100% of cells. She
received 6 months of chemotherapy on a Pediatric Oncology Group
(POG) regimen with mitoxantrone, high-dose cytarabine, etoposide,
and intrathecal cytarabine, achieving a complete remission.
In June, 1999, her AML relapsed. At that time, she was going
through menarche with early breast development. She was treated
with a POG salvage regimen of idarubicin and cladribine, achieving
a second remission. She then underwent allogeneic HSCT with
umbilical cord blood from a single male donor. Her conditioning
regimen consisted of TBI (1,350 cGy in nine fractions over 4.5
days) followed by 60 mg/kg etoposide x one dose. Approximately
8.61 x 106 CD34 cells/kg and 7.83 x 106 CD3 cells/kg were infused,
and she achieved 100% engraftment with donor cells. Post-HSCT
immunosuppression consisted of cyclosporine and steroids, which
were weaned off by day 180. Her leukemia remains in remission since
then, and she has had no infectious complications other than shingles.
She has had several health issues since HSCT, including cutaneous
graft versus host disease. Her other problems are likely due in part
to radiation: cataracts, avascular necrosis of both femoral heads,
nonalcoholic steatohepatitis, pancreatic exocrine insufficiency with
diabetes, hypothyroidism, and hypogonadism. She was transplanted
during menarche, stopping further sexual maturation and causing
growth arrest secondary to ovarian failure. She has chronic xerostomia
with chronic dental problems, and she is unable to sweat normally,
interfering with thermal regulation.
Breast cancer history
In February 2015, she felt a mass in the lower inner quadrant of
her right breast. Diagnostic mammography revealed a suspicious 1.3
cm mass, and she was referred to a breast surgeon. At that time, she
had a 2 x 2 cm area of thickening in the right breast about 3 cm from
the nipple. Ultrasound showed a 1.3 x 0.7 x 1.1 cm mass along the
2:30 axis 3 cm from the nipple, with branch extension into a dilated
subareolar lactiferous sinus suspicious for intraductal cancer, and a
4 mm hypoechoic nodule along the 1:00 axis about 1 cm from the
superior margin of the mass. No abnormal axillary nodes were seen.
Ultrasound-guided biopsy showed infiltrating and in situ ductal
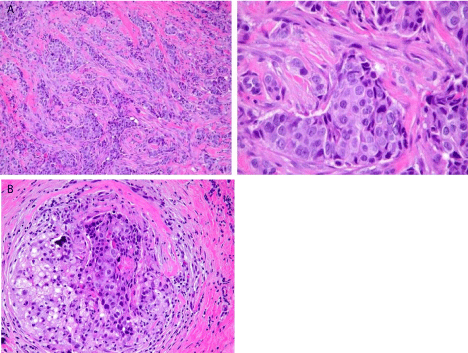
carcinoma of high grade (SBR grade 3) with the in situ component
less than 5% of the tumor volume (Figure 1A and B). The prognostic
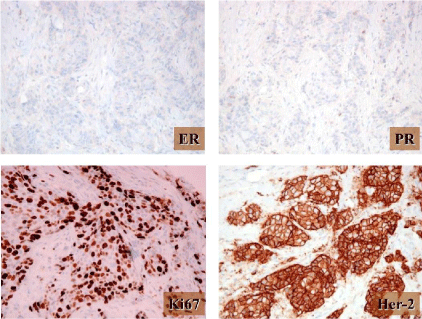
profile showed negative estrogen and progesterone receptors. HER-
2/neu was positive by FISH (HER-2/cep-17 ratio 4.7, average HER-2
spot count 21.0). Ki-67 was high at 54% (Figure 2).
PET/CT scan showed no evidence of metastatic disease. Breast
MRI scan revealed the primary tumor mass, as well as abnormal
linear enhancement extending from the lateral border of the mass
towards the base of the nipple. At least four additional well-defined,
intensely enhancing masses up to 9 mm were located within the
upper, inner, and anterior regions of the breast. These findings were
highly suspicious for extensive multifocal malignancy spanning an
area of 4.4 x 4.5 cm in her relatively small right breast.
Breast conservation was not an option. The extent of disease
within her involved breast and her prior TBI effectively precluded
postoperative breast radiation. In addition, the left breast was
also considered at increased risk for breast cancer due to her prior
radiation, and bilateral mastectomy was recommended. Finally, her
MRI scan strongly suggested multifocal right breast cancer, raising
concern that immediate mastectomy might leave positive surgical
margins requiring post-operative radiation. Accordingly, she was
treated with neoadjuvant chemotherapy prior to her surgery. The
TCHP regimen, consisting of docetaxel, carboplatin, trastuzumab,
and pertuzumab, was chosen for its high rate of pathologic complete
remission in the neoadjuvant setting [16,17].
In August 2015, she underwent bilateral mastectomies following
six cycles of TCHP. The final pathology showed no residual
malignancy in the breast, and a sentinel lymph node was negative.
There was no evidence of disease in the left breast. She did well
postoperatively, completing a full year of adjuvant trastuzumab, and
she remains disease free.
Figure 1A and 1B
Figure 1A and 1B
Ultrasound directed biopsy of breast mass showing
infiltrating and in situ ductal carcinoma, SBR grade 3. The intraductal
component showed a solid growth pattern with comedonecrosis, and
constituted less than 5% of the tumor volume. (Photomicrographs courtesy
of Dr. Jo Ellen Krueger).
Figure 2
Figure 2
Prognostic profile by Immunohistochemical staining of the tumor
tissue. Estrogen Receptor (ER) and Progesterone Receptor (PR) stains are
both completely negative. Ki-67 is high, staining 3+ in 54% of cells, indicating
a high proliferative rate. HER2/neu is strongly positive, staining 3+. HER2
by Fluorescence In Situ Hybridization (FISH) confirmed amplification of the
HER2 gene, with a HER2/cep-17 ratio of 4.7 and HER2 spot count of 21.0.
(Photomicrographs and immunohistochemical stains courtesy of Dr. Jo Ellen
Krueger).
Discussion
Survivors of childhood cancer have long been known to have an
increased risk of second malignancies later in life [2,3,12,14,15,18,19].
Radiation therapy for childhood cancer accounts for a significant
element of this risk, with a particularly common group being patients
treated with mantle field radiation for Hodgkin Lymphoma. Those
treated before age 30 years face an ongoing risk of breast cancer
beginning a few years after radiation, and extending even into the
fifth and sixth decades of life [20]. Recognition of ionizing radiation
in children and adolescents as a risk factor for cancer began with
analytic studies of Japanese atomic bomb survivors, followed by many
epidemiologic studies since that time [1,4,5,18]. With the increasing
use of radiation to treat childhood cancer, the carcinogenic effects of
therapeutic radiation have become progressively more obvious [7,9-
13,14,18,20]. Mantle radiation in girls with Hodgkin lymphoma has
long been accepted a specific risk factor for breast cancer [12-15].
Chemotherapy rather than radiation is the principal treatment
for pediatric cancers. Acute leukemia is the most common cancer
in children, accounting for about 30% of childhood malignancies.
Acute Lymphoblastic Leukemia (ALL) is about five times more
common in children than Acute Myelogenous Leukemia (AML),
with around 2,500-3,500 new cases per year. The five-year survival
rate for childhood ALL is about 85%, and for AML about 60-70%,
and children free of recurrent leukemia for 5 years are probably
cured [21]. For some acute leukemic children, particularly AML, the
prognosis for long-term remission with chemotherapy is poor. For
these patients and for those who relapse after standard chemotherapy,
allogeneic HSCT may offer the best chance for cure. Relatively few
children undergo allogeneic HSCT, however, and only some of
them receive TBI in addition to chemotherapy as their conditioning
regimen [22-24]. Consequently, many fewer girls are treated with
TBI compared with the number treated with mediastinal radiation
for Hodgkin Lymphoma. Additionally, several studies examining
second malignancies after childhood TBI reported a variety of solid
tumors, but did not report an increase in breast cancer [22,23]. For
these reasons, physicians are less aware of the breast cancer risk from
TBI than from mantle field radiation.
Possible contributory factors to our patient developing breast
cancer at age 27 include chemotherapy, exposure to relatively low
dose radiation, and possible immune compromise secondary to
the cord blood transplant. While chemotherapy and radiation are
known carcinogens, there is no reason to believe her immunologic
status was an etiologic factor in her breast cancer. She was only on
immunosuppressive medication for 180 days post-transplant, and her
only significant infection throughout her entire course was an episode
of shingles. In addition, several large studies of immunosuppression
in solid organ transplant patients found no increase in the incidence
of breast cancer, despite an overall increase in secondary malignancy.
The largest of these was a cohort study linking the US Scientific
Registry of Transplant Recipients to 13 state and regional cancer
registries, including 175,732 solid organ transplant recipients. The
incidence of breast cancer in this study was actually lower than
predicted, with a Standardized Incidence Ratio (SIR) of 0.85. As
shown in Table 2, similar results are reported in several other analytic
studies, with SIRs approximating 1.00 in both transplant patients and
HIV/AIDS patients [25-28].
Several recent large-scale investigations of the effects of HSCT
on long-term health have shed considerable light on the problem
of secondary breast cancer [29,30]. In 2008, Friedman et al. [29]
evaluated 3,337 women followed by the Fred Hutchinson Cancer
Research Center or by the European Bone Marrow Transplant
Registry who survived at least 5 years after allogeneic HSCT before
age thirty. The incidence of breast cancer was 26.6 cases per 10,000
patients per year. Three major breast cancer risk factors identified in
this review were younger age at transplantation, disruption of ovarian
function by alkylating agents, and exposure of breast tissue to radiation
with TBI. The hazard ratios were 9.5 with transplantation under
age 18, and 4.0 with TBI. Of these survivors, 1.6% developed breast
cancer (median of 12.5 years after transplantation) with a 25-year
cumulative incidence of 11%. In a 2011 study, investigators examined
long-term health outcomes in childhood cancer survivors treated
with HSCT, compared both with their siblings (N=4,020) and with
control patients treated with conventional cancer therapy (N=7,207)
[30]. The childhood allogeneic HSCT survivors were at highest risk
for severe/life-threatening conditions, including second malignancy,
compared to conventionally treated cancer patients and to the
non-cancer siblings. Nearly half of the female transplanted patients
developed gonadal failure compared to 4% of those conventionally
treated, with more than double the incidence of second malignancies.
Treatment with alkylating agents and anthracyclines may also be risk
factors for breast cancer in childhood cancer survivors, in addition to
the effects of chest radiation, [31]
Moskowitz et al. [32] reported on the cumulative risk of breast
cancer in 1,230 female cancer survivors treated with radiation therapy
[21]. They found treatment with a lower delivered dose of radiation
(2 to 20 Gy) had a much higher risk of subsequent breast cancer
(SIR 43.6), compared to the moderately high risk (SIR 24.2) after the
higher delivered dose of mantle field radiation (40 Gy) for Hodgkin
Lymphoma. The cumulative breast cancer was 30% by age 50, with
breast cancer specific mortalities of 12% at 5 years and 19% at 10
years, showing a substantial risk for both breast cancer incidence and
death among these survivors.
The incidence of secondary malignancy after childhood radiation
continues to increase throughout adult life, extending at least into the
fifth and sixth decades. Turcotte et al. [20] studied 14,364 childhood
cancer survivors diagnosed between 1970 and 1986, of whom 3,171
were age 40 or older at last contact. They found that secondary
malignant neoplasms including new breast cancers continue to
occur after age 40 in these patients, at least to age 55. Among the 196
secondary malignancies (other than non-melanoma skin cancers)
diagnosed after age 40, breast cancer had an SIR of 5.5, mostly in
women who had childhood chest radiation.
These studies substantiate the considerable risk of breast cancer
among women whose breast tissue was exposed to therapeutic
radiation as children. There is a considerable body of evidence showing
the risk with high dose radiation, both therapeutic (e.g., mantle field
radiation) and environmental (e.g., atomic bomb survivors). Much
less data is reported on the risk following low dose childhood radiation
with TBI conditioning for stem cell transplantation. Nonetheless, it is
clear that TBI in female children and adolescents is associated with
a significant risk of breast cancer, continuing into adulthood and
even into old age. Although a retrospective analysis by the Pediatric
Blood and Marrow Transplant Consortium failed to show a survival
advantage, TBI is still used in a small number of pediatric patients
undergoing HSCT for childhood AML [33].
Given the data summarized here, it is apparent that women
treated with childhood TBI need lifelong breast cancer screening. We
do not yet have tools to select those at highest risk for whom even
closer surveillance is appropriate. However, a recent study reported
at the 2016 annual meeting of the American Association for Cancer
Research offers hope that such tools may eventually become available
[34]. Dr. L. M. Morton and colleagues at the National Cancer Institute
looked for genomic variants in female patients who developed breast
cancer after childhood chest radiotherapy. They found two variants at
two genetic loci associated with an increased risk, one with a hazard
ratio of 1.96, and the second with a hazard ratio of 3.71. While these
are preliminary findings, they suggest that these or other genomic
variants may define a population with an increased susceptibility of
breast tissue to the carcinogenic effects of radiation, and in the future
may help select patients for more intense surveillance. However, until
we have such selective tools, all females exposed to chest or breast
radiation in childhood or adolescence will require close surveillance
as adults.
Table 2
Table 2
Compilation of four analytic reports evaluating the risk of breast cancer in immune compromised subjects. These include reports of studies of solid organ
transplant patients on long-term treatment with immunosuppressive drugs, as well as studies of patients with compromised immune systems due to HIV/AIDS. The
studies reviewed in these reports did not demonstrate any substantial increase in breast cancer risk among patients with compromised immune systems.
Conclusions
The studies cited in this report demonstrate clearly that exposure
of female breast tissue to radiation from early menarche to young
adulthood is associated with an increased risk of breast cancer,
extending from young adulthood into at least the sixth decade of
life. Oncologists have long been cognizant of the risk associated with
mantle field and other high levels of radiation exposure. The reports
of the substantial breast cancer risk following low-level total body
radiation used before stem cell transplantation have heightened that
awareness.
When these children become adults, their medical care falls
mainly to primary care providers, who are less likely than oncologists
to be aware of these risks. Because breast cancer surveillance is
not prioritized in the care of young women, the opportunity for
potentially life-saving early detection is diminished. Until such time
as we can identify individuals who are at uniquely high risk based
upon genomic profiling or other as yet unidentified factors, all young
women with a history of chest wall or breast radiation exposure in
childhood or early adulthood should be counseled about their breast
cancer risk, and should undergo lifelong breast cancer surveillance.
Acknowledgement
The author gratefully acknowledges the assistance of Jo Ellen Krueger, M.D., Medical City Hospital, Dallas, Texas, for preparing the photomicrographs and special stains for the Figures and allowing me to use them in this manuscript.
References
- Little MP. Cancer and non-cancer effects in Japanese atomic bomb survivors. J Radiol Prot. 2009; 29: A43-A59.
- Preston DL, Shimizu Y, Pierce DA, Suyama A, Mabuchi K. Studies of mortality of atomic bomb survivors. Report 13: Solid cancer and non-cancer disease mortality: 1950-1997. Radiat Res. 2003; 160: 381-407.
- Preston DL, Ron E, Tokuoka S, Funamoto S, Nishi n, Soda M, et al. Solid cancer incidence in atomic bomb survivors: 1958-1998. Radiat Res. 2007; 168: 1-64.
- Goodman MT, Cologne JB, Moriwaki H, Vaeth M, Mabuchi K. Risk factors for primary breast cancer in Japan: 8-year follow-up of atomic bomb survivors. Preventive Medicine. 1997; 26: 144-153.
- Carmichael A, Sami AS, Dixon JM. Breast cancer risk among survivors of atomic bomb and patients exposed to therapeutic ionising radiation. EJSO. 2003; 29: 475-479.
- Preston DL, Mattsson A, Holmberg E, Shore R, Hildreth NG, Boice JD. Radiation effects on breast cancer risk: A pooled analysis of eight cohorts. Radiat Res. 2002; 158: 220-235.
- Subgroup on Solid Cancer Risk. 3.13 Breast Cancer. In: Independent Advisory Group on Ionising Radiation - Health Protection Agency, editors. Risk of Solid Cancers Following Radiation Exposure: Estimates for the UK Population. Documents of the Health Protection Agency: Radiation, Chemical and Environmental Hazards. August 2011. P. 144-148.
- Walsh L, Zhang W. Radiation risk models for all solid cancers other than those types of cancer requiring individual assessments after a nuclear accident. Radiat Environ Biophys. 2016; 55: 9-17.
- Rheingold SR, Neugut AI, Meadows AT. Chapter 159: Secondary Cancers: Incidence, Risk Factors, and Management - Therapy-Related Secondary Cancers. In: Kufe DW, Pollock RE, Weichselbaum RR, et al., editors. Holland-Frei Cancer Medicine. 6th edition. Hamilton (ON): BC Decker; 2003.
- Ron E. Cancer risks from medical radiation. Health Phys. 2003; 85: 47-59.
- Kamran SC, de Gonzalez AB, Ng A, Haas-Kogan D, Viswanathan AN. Therapeutic radiation and the potential risk of second malignancies. Cancer. 2016; 122: 1809-1821.
- Moskowitz CS, Chou JF, Wolden SL, Bernstein JL, Molkotra J, Friedman DN, et al. Breast cancer after chest radiation therapy for childhood cancer. J Clin Oncol. 2014; 32: 2217-2223.
- Ng AK. Current survivorship recommendations for patients with Hodgkin lymphoma: focus on late effects. Blood. 2014; 124: 3373-3379.
- Koo E, Henderson MA, Dwyer M, Skandarajah AR. Management and prevention of breast cancer after radiation to the chest for childhood, adolescent, and young adulthood malignancy. Ann Surg Oncol. 2015; 22: 545-551.
- Terenziani M, Massimino M, Magazzu D, Gandola L, Capri G, Carcangiu ML, et al. Management of breast cancer after Hodgkin’s lymphoma and pediatric cancer. Eur J Cancer. 2015; 51: 1667-1674.
- Schneeweiss A, Chia S, Hickish T, Harvey V, Eniu A, Hegg R, et al. Pertuzumab plus trastuzumab in combination with standard neoadjuvant anthracycline-containing and anthracycline-free chemotherapy regimens in patients with HER-2 positive early breast cancer: A randomized phase II cardiac safety study (TRYPHAENA). Ann Oncol. 2013; 24: 2278-2284.
- Gianni L, Pienkowski T, Im Y-H, Tseng L-M, Liu M-C, Staroslawska E, et al. 5-year analysis of neoadjuvant pertuzumab and trastuzumab in patients with locally advanced, inflammatory, or early-stage Her-2 positive breast cancer (NeoSphere): A multi-centre open-label phase 2 randomised trial. The Lancet Oncology. 2016; 17: 791-800.
- American Academy of Pediatrics Committee on Environmental Health. Risk of ionizing radiation exposure to children: A subject review. Pediatrics. 1998; 101: 717-719.
- Friedman DL, Whitton J, Leisenring W, Mertens AC, Hammond S, Stovall M, et al. Subsequent neoplasms in 5-year survivors of childhood cancer: The Childhood Cancer Survivor Study. J Natl Cancer Inst. 2010; 102: 1083-1095.
- Turcotte LM, Whitton JA, Friedman DL, Hammond S, Armstrong GT, Leisenring W, et al. Risk of subsequent neoplasms during the fifth and sixth decades of life in the Childhood Cancer Survivor Study Cohort. J Clin Oncol. 2015; 33: 3568-3575.
- American Cancer Society. Cancer Facts & Figures 2016. Atlanta, Ga: American Cancer Society; 2016.
- Ricardi U, Filippi AR, Biasin E, Ciamella P, Botticella A, Franco P, et al. Late toxicity in children undergoing hematopoietic stem cell transplantation with TBI-containing conditioning regimens for hematological malignancies. Strahlenther Onkol. 2009; 185: 17-20.
- Omori M, Yamashita H, Shinohara A, Kurokawa M, Takita J, Hiwatari M, et al. Eleven secondary cancers after hematopoietic stem cell transplantation using a total body irradiation-based regimen in 370 consecutive pediatric and adult patients. Springer Plus. 2013; 2: 424-428.
- Ferry C, Gemayel G, Rocha V, Labopin M, Esperou H, Robin M, et al. Long-term outcomes after allogeneic stem cell transplantation for children with hematologic malignancies. Bone Marrow Transplantation. 2007; 40: 219-224.
- Stewart T, Tsai S-C J, Grayson H, Henderson R, Opelz G. Incidence of de-novo breast cancer in women chronically immunosuppressed after organ transplantation. The Lancet. 1995; 346: 796-798.
- Grulich AE, van Leeuwen MT, Falster MO, Vajdic CM. Incidence of cancer in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. The Lancet. 2007; 370: 59-67.
- Engels EA, Pfeiffer RM, Fraumeni Jr JF, Kasiske BL, Israni AK, Snyder JJ, et al. Spectrum of cancer risk among US solid organ transplant recipients. JAMA. 2011; 306: 1891-1901.
- Chapman JR, Webster AC, Wong G. Cancer in the transplant recipient. Cold Spring Harb Perspect Med. 2013; 3: a015677.
- Friedman DL, Rovo A, Leisenring W, Locasciulli A, Flowers ME, Tichelli A, et al. Increased risk of breast cancer among survivors of allogeneic hematopoietic cell transplantation: A report from the FHCRC and the EBMT-Late Effect Working Party. Blood. 2008; 111: 939–944.
- Armenian SH, Sun CL, Kawashima T, Arora M, Leisenring W, Sklar CA, et al. Long-term health–related outcomes in survivors of childhood cancer treated with HSCT versus conventional therapy: A report from the Bone Marrow Transplant Survivor Study (BMTSS) and Childhood Cancer Survivor Study (CCSS). Blood. 2011; 118: 1413-1420.
- van Leeuwen FE, Ronckers CM. Anthracycline and alkylating agents: New risk factors for breast cancer in childhood cancer survivors? J Clin Oncol. 2016; 34: 891-894.
- Henderson TO, Moskowitz CS, Chou JF, Bradbury AR, Neglia JP, Dang CT, et al. Breast cancer risk in childhood cancer survivors without a history of chest radiotherapy: A report from the Childhood Cancer Survivor Study. J Clin Oncol. 2016; 34: 910-918.
- Sisler IY, Koehler E, Koyama T, Domm JA, Ryan R, Levine JE, et al. Impact of conditioning regimen in allogeneic hematopoietic stem cell transplantation for children with acute myelogenous leukemia beyond first remission: a Pediatric Blood and Marrow Transplant Consortium (PBMTC) study. Biol Blood Marrow Transplant. 2009; 15: 1620-1627.
- Morton LM, Sampson JN, Armstrong GT, Chen T-H, Hudson M, Karlins E, et al. Genome-wide association study identifies two susceptibility loci that modify radiation-related risk for breast cancer after childhood cancer. A report from the Childhood Cancer Survivor Study and St. Jude Lifetime Cohort. Presented at the 107th AACR Annual Meeting (2016), April 16-20, 2016, New Orleans, LA. (Abstr 2691).