Case Report
Pancytopenia and Neuroendocrine Small Cell Carcinoma of the Bladder
Irache Abáigar-Pedraza1*, Juan Pablo Caballero-Romeu1, Estela Costa2 and Juan Francisco Galiano-Baena1
1Department of Urology, General University Alicante's Hospital, Spain
2Department of Anatomo Pathology, General University Alicante's Hospital, Spain
*Corresponding author: Irache Abáigar-Pedraza, Department of Urology, General University Alicante's Hospital, Servicio de Urología, Bloque B, 7ª Planta, Hospital General Universitario de Alicante, Avda. Pintor Baeza, 12, 03010-Alicante, Spain
Published: 18 Oct, 2016
Cite this article as: Abáigar-Pedraza I, Caballero-Romeu
JP, Costa E, Galiano-Baena JF.
Pancytopenia and Neuroendocrine
Small Cell Carcinoma of the Bladder.
Clin Oncol. 2016; 1: 1114.
Abstract
Clinical Practice: Small cell neuroendocrine tumor of the bladder is a rare aggressive tumor with
high metastatic potential. Despite aggressive multi-modal treatment, it has a high mortality rate.
Tumor infiltration of the bone marrow, myelophthisis, is uncommon and has a devastating prognosis.
This is the second case described in scientific literature. In patients with bladder carcinoma and an
alteration in any of the three blood series (red cells, white cells, platelets) that do not respond to
transfusion, infiltration of the bone marrow must be established.
Keywords: Small cell neuroendocrine carcinoma of the bladder and myelophthisic anemia
Introduction
Small cell (oat cell) neuroendocrine tumors were first described in the bronchopulmonary tract
[1]. Later, extra pulmonary locations such as in the urinary tract were published [2,3]. In the latter
case, the urinary organ most commonly affected is the bladder, being the source of 20% of extra
pulmonary neuroendocrine tumors. These have an incidence of 0.3–0.7% of all bladder cancers
[4]. In more than 50% of the cases, it co-exists with other kinds of urinary tract tumors: urothelial carcinoma, in situ carcinoma, adenocarcinoma, squamous cell carcinoma, atypical carcinoid tumor, and sarcomatoid component.
Prognosis is poor. It has a great capacity for local progression and metastasis, even after radical
treatment with neo- or adjuvant therapy.
Infiltration of the bone marrow by a small cell neuroendocrine bladder tumor is rare. This is the
second case reported in the scientific literature.
Case Report
An 85 year old patient, a non-smoker with no surgical or medical history, presented on
admission with a macroscopic hematuria that led to anemia: hemoglobin (Hb) 6g/dl. During his
stay, he suffered a hemorrhagic shock, requiring immediate admission to the intensive care unit and
hemostatic transurethral resection. Cystoscopy revealed a large blood clot and a solid lesion, 7cms in
diameter, with infiltrating appearance, located on the lateral wall of the bladder. The histopathology
study reported a neuroendocrine small cell carcinoma of the bladder that infiltrated muscle layers,
with numerous points of necrosis and a limited urothelial carcinoma component. It was positive for
CD 56 and synaptophysin, with a K167 proliferative index of almost 100%.
After surgery, the patient regained hemodynamic stability, but still had pancytopenia, with
red cells, 3x 106/μL; white cells, 2 x 103/μL; and platelets 13 x 103U/L, which did not improve with
transfusions. His baseline results had been normal a month earlier. A bone marrow aspiration was
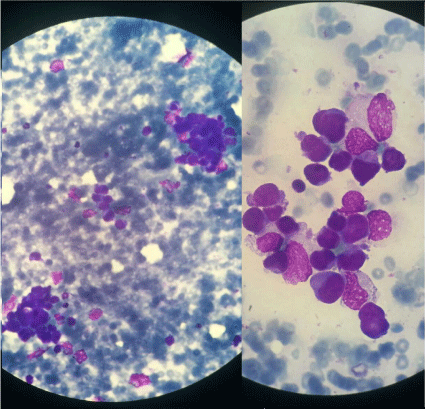
performed that showed infiltration by tumor cells (Figure 1).
Seven days after surgery, the patient had a febrile neutropenia with progressive hemodynamic
instability, managed with palliative measures. The patient died three days later.
A literature search was made in Medline using the keywords “small cell neuroendocrine
carcinoma of the bladder and myelophthisic anemia”. The results of the literature review are
presented.
Figure 1
Figure 2
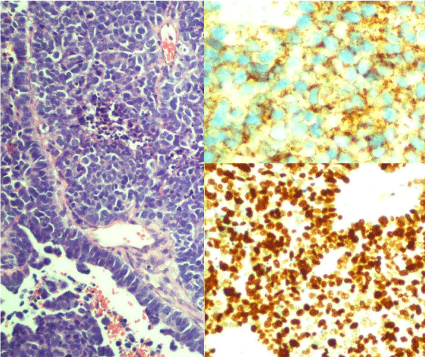
Figure 2
Small cells with a high mitotic index, hyperchromatic nuclei, small
prominent nucleoli and scarce cytoplasm, Ki67.
Discussion
The World Health Organization (WHO) classifies neuroendocrine
tumor as: neuroendocrine tumor grade 1, neuroendocrine tumor
grade 2, neuroendocrine carcinoma (large cell and small cell), and
mixed adenoneuroendocrine carcinoma (MANEC) [5,6]. Large cell
neuroendocrine carcinoma of the bladder is rare, with only ten cases
published in literature [5]. Small cell neuroendocrine carcinoma is
more common and, to date, 400 cases have been reported [5]. The
large series reported by Cheng et al. [7] consisted of 64 patients. The
age of onset, male predilection, symptoms, and debut are similar to
urothelial bladder tumors [2,3,5]. There is a higher prevalence in men
than in women (5:1). It appears around the 7th decade of life, and is
associated with the following risk factors: tobacco (70% of diagnosed
patients are smokers), and exposure to chemical products. It may be
associated, as in the case of squamous cell carcinoma of the bladder,
with a history of kidney lithiasis, chronic cystitis, and urinary catheter
(8,9).
The histogenesis of the neuroendocrine bladder tumor is
controversial. There are three main theories. The first claims that the
tumor originates from Kultschitzsky cells located in the urothelium
(10). The second associates the origin to urothelial cell metaplasia
(11). The third claims that it derives from a pluripotent stem cell of the
bladder mucosa (12). This latter theory is the one supported by most
authors because of the association of this neoplasm with other types
of bladder tumors in up to 50% of the cases, with the most frequent
being carcinoma in situ, followed by squamous cell carcinoma, and
sarcomatoid carcinoma (13).
The clinical picture does not differ from other bladder tumors. As
in the case of the patient being presented, 67 – 100% of the cases have
a mono-symptomatic macroscopic hematuria. It can be accompanied
by lower urinary tract symptoms, constitutional syndrome [5], and
dilation of the upper urinary tract (up to 95% has muscle invasion
at diagnosis). Paraneoplastic syndromes are rare, but may occur,
with metabolic abnormalities (hypercalcemia, hyponatremia,
hypokalemia), endocrine abnormalities (secretion of ADH, Cushing
Syndrome), or neurological abnormalities (myasthenia gravis or
Eaton Lambert Syndrome) (2,14). Many patients have symptoms
caused by metastatic lesions (up to 67% of patients at the time of
diagnosis.) (15,16). The most common parts of the body are: lymph nodes (56%), bone (44%), liver (33%), and lung (20%) (8).
Metastatic infiltration of the bone marrow due to this type
of tumor is extremely rare (17). To the best of our knowledge, the
present case is the second described in literature (18). Myelophthisis,
or bone marrow infiltration by non-hematopoietic cells, was
described by Reich in 1935, and may lead to different degrees of
anemia, thrombocytopenia, and neutropenia (18). Here, a case with
aplasia of the three cell series is presented. It occurs in 30% of solid
tumors, with solid carcinomas being the most common (breast, lung,
prostate, thyroid, kidney (18). It is exceptional in bladder tumors.
The situation that triggers the event is unknown, but excess extratumor
matrix metalloproteinases (MMP3, MMP-7, MMP-13) and
vascular endothelial growth factor are involved. Tumor cells occupy
the space of the trabecular bone, which leads to the destruction of
the bone marrow environment (Figure 1) and a leukoerythroblastic
reaction characterized by the presence of nucleated red blood cells,
dacriocytosis with left shift neutrophilia, and giant platelets on the
peripheral blood smear (18,19). The worst prognostic factors are: an
initial Hb level less than 9g/dl, the presence of febrile neutropenia,
and three or more concomitant visceral metastases. The patient
presented had the first two features. There is no standard treatment
for this condition. Both chemotherapy and radiotherapy show a
similar survival rate (17,18). Half of the patients diagnosed with
myelophthisis have a survival rate of less than 2 months.
As the symptoms and endoscopy findings are very vague,
the diagnosis of neuroendocrine bladder tumor is achieved by
histological and immunohistochemical study. Cryptoscopic findings
in the present case were similar to those described in the literature:
a solid mass a 4 to 10 cm solid mass with calcification and ulcerous
necrotic areas. Up to 54% of the cases were on the bladder side walls,
which suggest a urothelial carcinoma with a high degree of anaplasia
[2,3,5]. The electronic microscope shows small cells with a high
mitotic index, hyperchromatic nuclei, small prominent nucleoli and
scarce cytoplasm. Necrotic areas are seen forming a “lumpy” material
known as “starry sky”. The basophilic material around the blood
vessels is a common phenomenon called “Azzopardi phenomenon”
(8,14,15). The most characteristic data, but not the most specific, is
the presence of neurosecretory granules. At advanced stages, it is not
possible to demonstrate this neuroendocrine difference because the
cells are very difficult to differentiate.
Immunohistochemically, nearly 80% of the patients expressed
positivity or neuron-specific enolase (NSE), but this marker is not
very specific. Other, more specific, neuroendocrine markers include
synaptophysin, which is positive in 64.3% of the cases, and CD 56
in 71.4% of the cases; these were positive in the patient presented
(14,20). Positive results are also found for chromogranin A, in 28% of
the cases; for Nuclear Transcription Factor Thyroid (TTF-1) in 30%
of the cases (21); and epithelial markers (cytokeratin CAM 5.2, MNF
116, and EMA) (Figure 2).
Chromosome anomalies are related to small cell bladder
neuroendocrine tumors, such as the deletions in the short arm of
the chromosomes 4,5,10 and 13; DNA triploidy in the chromosomes
5p, 6p, 8q and alterations of the chromosome p53 and p16. Some of
these abnormalities match with those described in transitional cell
carcinoma (5, 16, 22). which supports the theory of histogenesis of the
pluripotent stem cell (12).
Unlike most small cell tumors of other organs,
immunohistochemical and genetic studies are not very specific for
this type of bladder cancer, and are not pathognomonic. Therefore,
WHO can only base its diagnosis on morphological criteria.
Differential diagnosis must be made with various pathologies.
On the one hand, with bladder metastasis of the lung carcinoma, a
lung focus must be ruled out via a CT scan, as well as a biopsy of
the lesion. On the other hand, it must be differentiated from a
lymphoma, which shows positivity for LCA, CD45, CD20, and also
from a neuroendocrine prostate carcinoma with bladder infiltration,
which is accompanied by an adenocarcinomatous component and
high PSA, as well as from an embryonic rhabdomyosarcoma, more
frequent in childhood, and positive for muscle specific vimentin,
desmin and actin, and finally from the poorly differentiated
transitional tumor, where the existence of small nuclei and positivity
for NSE, synaptophysin, and other neuroendocrine markers point to
small cell carcinoma (3,13).
Due to the low incidence of this disease and the advanced stage
at the time of diagnosis, there are different options for treatment,
but none has proved to be definitive. Most of these tumors have
micrometastases at diagnosis, making surgical treatment on its own
insufficient. Conservative treatment has no place, and is reserved
for palliative therapy (2,3,9). Given the characteristics of the patient
presented, with the hemodynamic instability and the advanced stage
of the tumor, the palliative option was taken.
Treatment with a curative purpose is aggressive. The most
common involves radical surgery plus adjuvant chemotherapy and/
or radiotherapy. The best results obtained combine radical surgery
with adjuvant therapy, as we are dealing with a chemo sensitive tumor
(2,3,5,9,24). As Mackey emphasized, the cisplatin based chemotherapy
was the only factor that improved survival. The most commonly used regimen is the combination of cisplatin and etoposide for a pure
neuroendocrine carcinoma, and the combination of methotrexate,
vinblastine, cisplatin and doxorubicin for a neuroendocrine tumor
associated with transitional cell carcinoma. Neoadjuvant therapy
may be advantageous in comparison to adjuvant therapy because a
large percentage of this patients has tumors with rapid growth rates,
making complete resection difficult or imposible.
The worst prognosis factors are: poor histological differentiation,
tumor size, involvement of bladder fat, the existence of
neurovascularization, and micro metastases at the time of diagnosis.
The latter being the main cause of death.
Despite aggressive treatment and therapy, the mortality rate is
very high. Up to 3 times greater than high-grade urothelial carcinoma,
with 60% of patients dying within 2 years of diagnosis. The average
survival is 3- 35 months. The 5-year survival is 8%. No difference has
been found in survival between pure and mixed tumors (3,15).
Conclusions
The small cell neuroendocrine bladder tumor is rare, very aggressive, and has high metastatic ability.
Despite multi-modal treatment with radical surgery and chemotherapy, with or without radiotherapy, it has an unfavorable evolution, ending the patient’s life around two months from the time it is diagnosed.
References
- Perán Teruel M, Giménez Bachs JM, Ruiz Martínez J. Carcinoma neuroendocrino de vejiga. Análisis retrospectivo de 15 años. Arch Esp Urol. 2012; 65: 237-243.
- Zhao X, Flynn EA. Bladder Small Cell Carcinoma. Arch Pathol Lab Med. 2012; 136: 1451–1459.
- Cuesta JA, Ripa L, Aldave J. Neuroendocrine small cell carcinoma of the bladder. Review of the literature and report of a case. Arch Esp Urol. 2002; 55: 452-456.
- Choong NW, Quevedo JF, Kaur JS. Small cell carcinoma of the urinary bladder: the Mayo Clinic experience. Cancer. 2005; 103: 1172–1178.
- Kickuth R, Laufer U, Pannek J. Magnetic resonance imaging of bone marrow metastasis with fluid-fluid levels from small cell neuroendocrine carcinoma of the urinary bladder Magnetic Resonance Imaging. 2002; 20; 691–694.
- Luján M, Cardona AF, Yepes A. Mieloptisis: viejos aspectos, nuevos conceptos. Acta Méd Colomb. 2009; 34: 169-175.
- Mangar SA, Logue JP, Shanks JH. Small-cell carcinoma of the urinary bladder: 10-year experience. Clin Oncol. 2011; 16: 523–527.