Research Article
Surgical Site Infection Rates in Breast Cancer Surgery at a University Hospital in Nairobi, Kenya
Nyaoncha A1*, Wasike R1, Ahmed M2 and Njihia B1
1Department of Surgery, Aga Khan University Hospital, Kenya
2Department of Surgery Aga Khan University Hospital, Tanzania
*Corresponding author: Andrew Nyaoncha, Department of Surgery, Consolata Mathari Mission Hospital, PO Box 30270-00100, Nyeri, Kenya
Published: 02 Sep, 2016
Cite this article as: Nyaoncha A, Wasike R, Ahmed M,
Njihia B. Surgical Site Infection Rates in
Breast Cancer Surgery at a University
Hospital in Nairobi, Kenya. Clin Oncol.
2016; 1: 1069.
Abstract
Background: Surgery is the mainstay of treatment for breast cancer and with it the attendant
risk of surgical site infection (SSI). Breast cancer surgery, though classified as a clean procedure
presents with a relatively higher rate of infection than similar operations and it remains unclear
whether extended antibiotic regimes improve this outcome. This study sought to evaluate SSIs using
validated detection protocols.
Methods: A prospective surveillance study for patients undergoing breast cancer surgery using the
National Nosocomial Infections Surveillance (NNIS) system.
Results: Sixty nine patients who underwent breast cancer related surgery at Aga Khan University
Hospital-Nairobi were recruited over the period from September 2013 to April 2014, with 2 lost to
follow-up. Six percent (n=4) of patients developed SSI, with 1 case of CDC Class I, and 3 cases CDC
Class II. Various risk factors such as obesity, diabetes, age >65 years and prolonged drain duration
were noted to be important contributors increasing the risk of SSI development. Only one patient
required operative management of their infection.
Conclusion: This study demonstrates the successful introduction of an SSI surveillance protocol
at a tertiary facility with an subsequent infection rate lower than reported in other studies that
utilize only pre-operative antibiotics. We recommend a randomized controlled trial to compare
outcomes between pre-operative only and pre- plus post-operative antibiotic use in order to explore
this further.
Keywords: Infection; Breast Surgery; Antibiotic; Prophylaxis
Background
Surgery is the mainstay of treatment for breast cancer, a major cause of morbidity and mortality
in both pre- and post-menopausal women [1-4]. The evolution from debilitating extensive
procedures in the earlier years to the low morbidity procedures currently practised has improved
the adverse events attributable to surgery.
Surgical site infection (SSI) however remains a common complication of breast surgery, with
attendant increase in patient discomfort, cost of care and burden to clinical staff [5-7]. It carries the
further risks of delaying adjuvant chemo-radiation and unsightly scarring [7-9] in women already
traumatized by the diagnosis of cancer and the surgical procedure itself. This situation obtains
despite the recommended use of single dose peri-operative antibiotics, leading to unregulated postoperative
doses in an attempt to reduce infection rates. It is not known whether these extended
antibiotic regimes result in reduced SSI rates. The absence of SSI surveillance protocols further
compounds the scenario.
With paucity of data as regards use of post-operative antibiotics and its effect on SSI rates, the
present study sought to evaluate the SSI outcomes in breast cancer surgery with the use of perioperative
and post-operative antibiotic prophylaxis using internationally validated protocols for SSI
detection and SSI definitions.
Patients and Methods
The study was a prospective surveillance study at the Aga Khan University Hospital. Inclusion
criterion was age above 18 with diagnosis of breast cancer. Exclusion criteria included: failure to
obtain consent for inclusion; patients in whom there was already a suspected infection following
biopsy procedure or where tumor ulceration was present.
Ethical approval was sought from the institutional ethical
committee of the Aga Khan University College of Health Sciences
and confidentiality of patient information was secured.
In addition to standard hospital procedure with regard to patient
care for breast surgery, the following aspects were instituted into the
study protocols:
The patients were assessed pre-operatively for SSI risk factors using
a questionnaire. Patients were scheduled to receive peri-operative
chemoprophylaxis as a single dose of Cefuroxime 750 mg IV at the
time of induction of anesthesia. If surgery was prolonged beyond 4
hours, patients were to receive an additional dose of Cefuroxime 750
mg IV. Where there was known allergy or any contraindication to the
use of Cefuroxime, Augmentin 1.2 g IV was to be used. All patients
were to receive antibiotic prophylaxis of Cefuroxime 750 mg IV 8
hourly for the first two days while in hospital. Patients undergoing
BCT would receive no further treatment. Patients undergoing
mastectomy would receive an additional 3 days of oral antibiotic
(Cefuroxime 750mg 12 hourly) to make the total prophylaxis
duration 5 days. In the event of reported allergy or contraindication
to Cefuroxime use, patients were to receive Augmentin 1.2 g 8 hourly
for IV prophylaxis and Augmentin 1g 12 hourly for oral prophylaxis.
All patients were to be reviewed in the clinic within one week of
discharge from the hospital. If SSI was detected or suspected based on
CDC criteria, an aspirate or a swab would be collected where possible
and sent for microbiological study in a transport medium used for
standard culture and sensitivity. The drain was assessed for amount
of drainage. Criteria for removal were based on volumetric analysis;
removal was advised once the drainage was <30 ml/24hour. Events
such as drain dislodgement and adjustment, drain unblocking or
aspiration of seroma were also collected.
Patients were to be followed up for a minimum 30 days regardless
of the number of clinic visits. This data was captured in the adapted
NCC post-operative SSI detection form.
Primary outcome measures were rates of SSI (detected as per the
protocol), calculated as a percentage of the total number of subjects
analyzed over the study period; occurrence of SSI over the duration of
time to assess for temporal spread; categories/ classes of SSI.
Secondary outcome measures included incidence of common
risk factors for SSI development; sub-analysis to identify risk
factors present in patients who developed SSI; associations between
identified patient and surgical risk factors and development of SSI;
Documentation of other clinical outcomes other than SSI.
Collected data were summarized in the form of tables, graphs and
figures showing the variables of interest and the temporal distribution
of events during the study period. The data were analyzed in form of
rates and proportions.
Results
Sixty nine consenting patients who underwent breast cancer
related surgery at Aga Khan University Hospital-Nairobi were
recruited over a period of 8 months from September 2013 to April
2014. Two (2) patients could not be contacted subsequently and were
excluded from the final analysis. All the remaining 67 patients were
followed up until completion of the surveillance period of 30 days.
Their characteristics are summarized in Table 1.
All mastectomy procedures and 30 BCT procedures were
classified as clean operations while two of the BCT procedures were
classified as clean contaminated operations (with minor breaks in
sterility: contaminated diathermy cord and break of gloves). Surgical
drains were used in all operations except patients who underwent
BCT and sentinel lymph node biopsy (SLNB) without further axillary
dissection (n=55). Only one drain was used per patient. External
compression dressing for 24-48 hours was used in all patients who
underwent mastectomy or had axillary dissection with any other
procedure.
All patients received a stat prophylactic dose at the time of
induction of anesthesia or before surgical incision. Thirty one
patients who underwent mastectomy and a similar number who
underwent BCT received Cefuroxime 1.5 g IV stat while 4 patients
who underwent mastectomy and 1 who underwent BCT received
Augmentin 1.2 IV stat at induction of anesthesia. There were no
antibiotic-related adverse reactions. The actual pattern of antibiotic
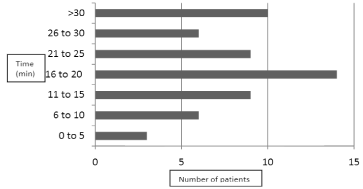
administration is summarized in Figure 1. None of the patients
required a second intra-operative antibiotic dose.
Thirty one (31) post-mastectomy patients received Cefuroxime
500mg orally 12 hourly while 4 patients received Augmentin 1 g
orally 12 hourly. All patients reported completion of the prophylaxis
regimen as prescribed. Of patients who underwent BCT 31 received
Cefuroxime 750 mg 12 hourly while 1 received Augmentin 1g
12 hourly oral medications. All patients reported completion the
prophylaxis regimen as prescribed.
Strict supervision and emphasis of the antibiotic prophylaxis
regimen was done, resulting in no deviation from this protocol. No
patients received additional antibiotics except where SSI had been
detected (4 patients).The rest of the post-operative care remained
standard.
All patients were followed up with at least once-weekly clinic
review as scheduled by the primary surgeon or alternatively by
telephonic interview. A new data entry form was filled for each review
until each patient had received at least four evaluations within the
30-day surveillance period. There was no drop-out or breach in study
protocol.
Primary outcome: Rate of SSI
Four patients (6%) developed surgical site infections. Two had
undergone mastectomy and the other two others breast conservation
surgery. The infections were diagnosed on post-operative days 10, 18,
19 and 27. Three of the infections were CDC class II infections and
one, CDC class I infection. One patient required surgical drainage of
the infection. Wound swabs were taken for all infections. One culture
grew Staphylococcus aureus. No growth was obtained from the other
three cultures. All patients with SSI were treated with a combination
of Amoxicillin-Clavulanic acid for 7 days. There was complete
resolution of the infection during this time.
Secondary outcomes
Risk factors for SSI development: Patients who developed SSI
had the following risk factors:
• Patient 1: Age >65 years, BMI of 44, diabetic, prolonged drain
duration of >19 days.
• Patient 2: 4 cycles of neo-adjuvant chemotherapy, blocked
drain.
• Patient 3: Age >65 years, BMI 41.6, diabetic
• Patient 4: No identifiable risk factors
Other complications: Other complications are as summarized
in Table 2. Only one patient an SSI that delayed administration of
adjuvant chemotherapy.
Table 1
Figure 1
Table 2
Discussion
In this prospective study, a total of 67 patients undergoing breast
cancer surgery were recruited and followed up until completion
of the surveillance period. While there is general agreement in the
literature that prophylactic antibiotics should be administered preoperatively,
there is ongoing debate whether the timing of preoperative
administration matters. Hawn et al. [10] in a large recently
published retrospective cohort study involving 32,459 patients failed
to demonstrate any benefit in the adherence to the recommendations
on the timing of prophylaxis. However, a prospective surveillance
study by Steinberg et al. [11] involving 4,472 patients using the
National Nosocomial Infections Surveillance system methodology
(similar to that used in this study) showed a trend towards reduced
risk for SSI when antibiotics with short infusion times such as
cephalosporins were given within 30 minutes of the surgical incision.
Another similarly designed prospective study by Weber et al. [12]
contradicts this view. We could not demonstrate an association
between the timing of antibiotic prophylaxis and the occurrence of
surgical site infection from our data. The present study was however
not powered to evaluate the effect of this variable. It is nonetheless
good clinical practice despite the ongoing debate to time prophylactic
antibiotics according to their pharmacokinetic properties despite lack
of strong evidence. The development of SSI is multi-factorial, and this
is one of the few modifiable factors that can be addressed.
The principal finding of the study was that only 4 out of 67
patients (6%) undergoing surgery for breast cancer developed SSI;
two of whom underwent mastectomy and two BCT. The rate of SSI
in this study is much less than that elicited in other studies in which
the standard pre- operative dose of antibiotics was used [5-7,9,13]. All
four patients required additional antibiotic treatment and additional
hospital visits. In addition, one patient required an additional surgical
procedure to drain the infection, multiple hospital visits for local
wound care and secondary wound closure.
The cosmetic outcome in this patient was unsatisfactory and there
was a delay of several weeks in starting adjuvant chemotherapy. In
addition patients with SSI suffered additional psychological trauma.
There was also an increase in the cost of treatment. The adverse
impact of SSI has been reported by others [6-9].
Three of the 4 infections were classified as CDC class I; one patient
had CDC class II infection. Vitug et al. [13] and Villar-Compte et al.
[14] also found that superficial infections were commoner than deep
ones. There was only one positive culture which grew Staphylococcus
aureus (S. aureus). In the 5-year prospective surveillance study by
Villar-Compte et al. [15], S. aureus was the most commonly isolated
micro-organism from SSI following breast cancer surgery. In other
studies of SSI following breast cancer surgery S. aureus comprised 48
% of 63 positive cultures [16], and 42% of 24 positive cultures [17].
This finding gives strength to the recommendation to administer
intravenous prophylactic antibiotics with anti-staphylococcal activity.
Several risk factors contribute to SSI development following
breast cancer surgery. Vitug et al. [13] and Xue et al. [6] identified
recent biopsy (<7 days before operation), obesity, smoking, neoadjuvant
chemotherapy or radiotherapy, age >60 years and heavy
alcohol intake as risk factors for SSI development. Olsen et al. [18],
Hall et al. [8] and Villar-Compte et al. [19] reported additional risk
factors including older age, ASA grade III and IV, intra-operative
hypotension, prolonged use of a drain and compromised overall
immune status. Some of these risk factors for SSI development were
prevalent in our study population (Table 1). Smoking was found to be
relatively uncommon (only 1% of the patients smoked) as compared
to rates reported in other studies [15,18]. Obesity was the commonest
risk factor in our study population, occurring in 27% of all patients,
and in 50% of patients who developed SSI. Three patients who
underwent mastectomy and 2 patients who had BCT were diagnosed
with cancer following excision for breast lumps initially thought to be
benign. All other cancers were diagnosed following core biopsy. There
was no association between the biopsy procedure and development of
infection. All biopsy procedures except one were done 7 days or more
before surgery; and so recent biopsy was not an important risk factor.
Neither of the patients who had recognized sterility breaks during
surgery developed an infection. We could not draw any important
conclusions regarding the role of intra-operative hypotension from
this study as this was mostly due to anesthetic factors and was easily
controlled. Hypotension was not due to intra-operative bleeding
requiring transfusion.
Among the patients who developed SSI, the most common risk
factors were diabetes, obesity, age >65 years, blocked drain and
prolonged drainage exceeding 19 days. SSI occurred in 22% of the
severely obese patients (BMI >35); in 25 % diabetic patients; in 25% of
those aged above 65 years; in 20% each of patients who had received
neo-adjuvant chemotherapy; in 13% of patients who had a drain in
situ for longer than 19 days and in 25% of patients with a blocked
drain.
Prolonged use of drains is known to be associated with increased
risk of SSI [6,20,21]. Villar-Compte et al. [19] reported drain use
exceeding 19 days to be associated with increased risk for SSI
development. In the present study, the average duration of drain use
was 12 days. However, out of 5 patients with prolonged use of drain
(>19 days), 1 (20%) developed surgical site infection. Proneness to
SSI following prolonged drain use is an important reason why some
surgeons prefer to use prophylactic antibiotics postoperatively for an
extended period [15,22]. In the present series additional extended
antibiotics were not used for patients with prolonged drain duration,
the maximum duration of drain use being 25 days post-operatively.
Seroma formation is reported to be the most common
complication following breast cancer surgery [13] and occurs in 20-
30% of patients. A 15% seroma formation rate was observed in the
present study. All the seromas followed modified radical mastectomy.
Seromas occurred more commonly when the effluent was 20 ml or
more in the 24 hours prior to drain removal. Seroma formation was
also commoner when drains were removed within 19 days of surgery.
Philips et al. [22] and Barton et al. [23] observed that volumetric
analysis of the drain effluent is a good indicator for drain removal.
Most surgeons remove drains when the 24 hour drainage volume
is <30ml in the previous 24 hours or at 14 days post operatively as
this is associated with reduced risk of seroma formation. Obesity
is associated with increased risk of fluid production and seroma
formation leading to prolonged duration of drain use [20,24]. The
findings in the present study related to seroma formation correlate
well with observations in the contemporary literature.
Shoulder dysfunction and medial upper arm numbness
collectively were reported in 21% of all patients who underwent
axillary dissection, making this the most common complication. This
complication was more common amongst patients undergoing MRM
(32%) compared to those undergoing BCT (17%). This is a higher
incidence than the 13.5% rate of shoulder dysfunction reported by
Roses et al. [25], 14% reported by Box et al. [26] and 17.7% reported
by Hack et al. [27]. Shoulder dysfunction is due to fibrosis during
healing following axillary dissection. As part of the post-operative
care program, all patients in this study were educated to perform
upper arm abduction and range of motion exercises starting from the
first post-operative day to help reduce the incidence and severity of
this complication. These exercises were reinforced during the postoperative
follow-up period to avert long-term immobility and the
development of a frozen shoulder. Other authors have contested this
regime of early mobilization and have reported increased wound
complications and seroma formation with shoulder mobilization
[28,29]. A systematic review by Shamley et al. [30] showed reduced
seroma formation with delayed shoulder exercises while there was no
significant difference in the incidence of shoulder dysfunction when
regimens of early and delayed shoulder exercises were compared.
Clinical evidence therefore supports a regimen of delayed shoulder
exercises calling into review our current practice of early shoulder
mobilization exercises.
Upper medial arm numbness is due to injury to the intercostobrachial
nerve during dissection [25]. Deliberate effort was made
during dissection to visually identify and protect the nerve, unless
it was matted within metastatic lymph nodes in which case it was
sacrificed. Inadvertent minor injury can also occur during tissue
retraction and is often temporary. Compared to the rates reported by
others [25,27] our rate of medial upper-arm numbness is quite low.
This is likely as a result of under-reporting amongst our population
of patients and also lack of deliberate assessment by the clinician
for this complication. It is however a major factor affecting quality
of life following surgical treatment of breast cancer [27], although
the natural history is one of improvement over time [25,27]. A
weakness of the present study was that this complication was not
independently elicited but was combined with elicitation of shoulder
dysfunction. Despite reports of hematoma formation between 2 to
10% in literature [13] following breast cancer surgery, there was
no hematoma formation in this study. This could be attributed to
meticulous surgical technique. In summary, with the antibiotic use
and strict surveillance protocols used in this study an SSI rate of 6%
was observed. This was significantly less than the mean incidence of
15% (5%-30%) reported in other similar studies [5,7,13,14,31]. This
reduction in observed infection rates could be attributed to the use
of peri-operative antibiotics. It could also be attributed to behavioral
change induced by the surveillance for taking preventive measures.
Several risk factors for SSI development were noted to be
prevalent in the study population. However, age >65 years, obesity
and diabetes appeared to be the most common risk factors for SSI
development. By contrast smoking was not a common risk factor.
Seroma formation and shoulder dysfunction/ medial upper arm
numbness were also common complications. We need to review our
current practice of early shoulder mobilization in view of evidence
that it is associated with a higher incidence of seroma formation
without a significant advantage over delayed mobilization in terms of
avoidance of shoulder dysfunction.
In conclusion, there was successful introduction of a surveillance
protocol for assessment of the risk of SSI following breast cancer
surgery at Aga Khan University Hospital, Nairobi. An SSI rate of
6% was obtained following active and prospective surveillance of
patients undergoing breast cancer surgery with the use of perioperative
antibiotics. This rate was lower than that reported in similar
studies using only pre-operative prophylactic antibiotic. Various risk
factors such as obesity, diabetes, age >65 years and prolonged drain
duration were noted to be important contributors increasing the risk
of SSI development. We recommend a randomized controlled trial
comparing SSI outcomes with the use of pre-operative antibiotics
only versus the use of additional post-operative antibiotics is
recommended to establish whether there is any significant reduction
in the rates of SSI development.
References
- Surgical Techniques in Breast Cancer Therapy; Schwartz‘s Principles of Surgery. 8th Edition. Chapter 16.
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009; 59: 225-249.
- El Saghir NS, Adebamowo CA, Anderson BO, Carlson RW, Bird PA, Corbex M, et al. Breast cancer management in low resource countries (LRCs): consensus statement from the Breast Health Global Initiative. Breast. 2011; 20: S3-S11.
- Bhikoo R, Srinivasa S, Yu TC, Moss D, Hill AG. Systematic review of breast cancer biology in developing countries (part 1): Africa, the middle East, eastern europe, Mexico, the Caribbean and South america. Cancers (Basel). 2011; 3: 2358-2381.
- Gupta R, Sinnett D, Carpenter R, Preece PE, Royle GT. Antibiotic prophylaxis for post-operative wound infection in clean elective breast surgery. Eur J Surg Oncol. 2000; 26: 363-366.
- Xue DQ, Qian C, Yang L, Wang XF. Risk factors for surgical site infections after breast surgery: a systematic review and meta-analysis. Eur J Surg Oncol. 2012; 38: 375-381.
- Tejirian T, DiFronzo LA, Haigh PI. Antibiotic prophylaxis for preventing wound infection after breast surgery: a systematic review and metaanalysis. J Am Coll Surg. 2006; 203: 729-734.
- Hall JC, Hall JL. Antibiotic prophylaxis for patients undergoing breast surgery. J Hosp Infect. 2000; 46: 165-170.
- Bold RJ, Mansfield PF, Berger DH, Pollock RE, Singletary SE, Ames FC, et al. Prospective, randomized, double-blind study of prophylactic antibiotics in axillary lymph node dissection. Am J Surg. 1998; 176: 239-243.
- Hawn MT, Richman JS, Vick CC, Deierhoi RJ, Graham LA, Henderson WG, et al. Timing of surgical antibiotic prophylaxis and the risk of surgical site infection. JAMA Surg. 2013; 148: 649-657.
- Steinberg JP, Braun BI, Hellinger WC, Kusek L, Bozikis MR, Bush AJ, et al. Timing of antimicrobial prophylaxis and the risk of surgical site infections: results from the Trial to Reduce Antimicrobial Prophylaxis Errors. Ann Surg. 2009; 250: 10-16.
- Weber WP, Marti WR, Zwahlen M, Misteli H, Rosenthal R, Reck S, et al. The timing of surgical antimicrobial prophylaxis. Ann Surg. 2008; 247: 918-926.
- Vitug A, Newman LA. Complications in breast surgery. Surg Clin North Am. 2007; 87: 431-51, x.
- Vilar-Compte D, Jacquemin B, Robles-Vidal C, Volkow P. Surgical site infections in breast surgery: case-control study. World J Surg. 2004; 28: 242-246.
- Vilar-Compte D, Rosales S, Hernandez-Mello N, Maafs E, Volkow P. Surveillance, control, and prevention of surgical site infections in breast cancer surgery: a 5-year experience. Am J Infect Control. 2009; 37: 674-679.
- Mukhtar RA, Throckmorton AD, Alvarado MD, Ewing CA, Esserman LJ, Chiu C, et al. Bacteriologic features of surgical site infections following breast surgery. Am J Surg. 2009; 198: 529-531.
- Rolston K, Mihu C, Tarrand J. Current Microbiology of Surgical Site Infections Associated With Breast Cancer Surgery. Wounds. 2010; 22: 132-135.
- Olsen MA, Lefta M, Dietz JR, Brandt KE, Aft R, Matthews R, et al. Risk Factors for Surgical Site Infection after Major Breast Operation. J Am Coll Surg. 2008; 207: 326-335.
- Vilar-Compte D, Jacquemin B, Robles-Vidal C, Volkow P. Surgical site infections in breast surgery: case-control study. World J Surg. 2004; 28: 242-246.
- Banerjee D, Williams EV, Ilott J, Monypenny IJ, Webster DJ. Obesity predisposes to increased drainage following axillary node clearance: a prospective audit. Ann R Coll Surg Engl. 2001; 83: 268-271.
- Throckmorton AD, Boughey JC, Boostrom SY, Holifield AC, Stobbs MM, Hoskin T, et al. Postoperative prophylactic antibiotics and surgical site infection rates in breast surgery patients. Ann Surg Oncol. 2009; 16: 2464-2469.
- Phillips BT, Wang ED, Mirrer J, Lanier ST, Khan SU, Dagum AB, et al. Current practice among plastic surgeons of antibiotic prophylaxis and closed-suction drains in breast reconstruction: experience, evidence, and implications for postoperative care. Ann Plast Surg. 2011; 66: 460-465.
- Barton A, Blitz M, Callahan D, Yakimets W, Adams D, Dabbs K. Early removal of postmastectomy drains is not beneficial: results from a halted randomized controlled trial. Am J Surg. 2006; 191: 652-656.
- Bonnema J, van Geel AN, Ligtenstein DA, Schmitz PI, Wiggers T. A prospective randomized trial of high versus low vacuum drainage after axillary dissection for breast cancer. Am J Surg. 1997; 173: 76-79.
- Roses DF, Brooks AD, Harris MN, Shapiro RL, Mitnick J. Complications of level I and II axillary dissection in the treatment of carcinoma of the breast. Ann Surg. 1999; 230: 194-201.
- Box RC, Reul-Hirche HM, Bullock-Saxton JE, Furnival CM. Shoulder movement after breast cancer surgery: results of a randomised controlled study of postoperative physiotherapy. Breast Cancer Res Treat. 2002; 75: 35-50.
- Hack TF, Cohen L, Katz J, Robson LS, Goss P. Physical and psychological morbidity after axillary lymph node dissection for breast cancer. J Clin Oncol. 1999; 17: 143-149.
- Schultz I, Barholm M, Gröndal S. Delayed shoulder exercises in reducing seroma frequency after modified radical mastectomy: a prospective randomized study. Ann Surg Oncol. 1997; 4: 293-297.
- Lotze MT, Duncan MA, Gerber LH, Woltering EA, Rosenberg SA. Early versus delayed shoulder motion following axillary dissection: a randomized prospective study. Ann Surg. 1981; 193: 288-295.
- Shamley DR, Barker K, Simonite V, Beardshaw A. Delayed versus immediate exercises following surgery for breast cancer: a systematic review. Breast Cancer Res Treat. 2005; 90: 263-271.
- Gutteridge M, Holden S, Clarkson A. Guideline for Antibiotic Prophylaxis Within Breast Surgery for Adult Patients: Nottingham Antibiotic Guidelines Committee, November 2009.